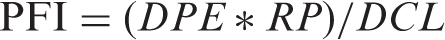Abstract
Background
The role of high-resolution esophageal impedance manometry (HRIM) for establishing risk for dysphagia after anti-reflux surgery is unclear. We conducted a prospective study of children with primary gastroesophageal reflux (GER) disease, for whom symptoms of dysphagia were determined pre-operatively and then post-operatively and we examined for features that may predict post-operative dysphagia.
Methods
Thirteen children (aged 6.8–15.5 years) undergoing work-up prior to 360o Nissen fundoplication were included in the study. A dysphagia score assessed symptoms at pre-operative study and post-operatively (mean 1.4 years). A HRIM procedure recorded 5-ml liquid, 5-ml viscous and 2-cm solid boluses. We assessed esophageal motility, esophago-gastric junction (EGJ) morphology, EGJ contractility and pressure-flow variables indicative of bolus distension pressures and bolus clearance pressures. A composite pressure-flow index score was also derived.
Results
Pre-operative pressure-flow index was positively correlated with post-operative dysphagia score (viscous bolus r = 0.771, p < 0.005). Of three variables that comprise the pressure-flow index, the ramp pressure measured during bolus clearance was the main driver of the effect seen (viscous bolus r = 0.819, p < 0.005).
Conclusions
In order to mitigate symptoms in relation to anti-reflux surgery, dysphagia symptoms and esophageal function need to be pre-operatively assessed. In patients with normal motility, an elevated pressure-flow index may predict post-operative dysphagia.
Key summary
The established knowledge on this subject:
Dysphagia can mar otherwise successful gastroesophageal reflux (GER) treatment. The ability to accurately predict dysphagia risk would inform the choice of fundoplication over other treatments. ‘Pressure-flow’ anomalies may be predictors of dysphagia symptoms following anti-reflux surgery. What are the significant and/or new findings of this study?
Dysphagia symptoms were common in pediatric GER disease patients prior to anti-reflux surgery. In order to avoid symptom recurrence, re-investigations and further interventions, dysphagia symptoms should be attributed to GER. In patients with normal motility, elevated clearance pressures and/or pressure-flow index may predict post-operative dysphagia.
Introduction
High-resolution esophageal impedance manometry (HRIM) derives biomechanical swallow measures which describe flow resistance and muscle contractility. These characterizations may guide clinical decisions around esophageal dysmotility.1–3
Clinical guidelines now recommend pre-operative assessment of children with gastroesophageal reflux (GER) disease referred for anti-reflux surgery. 4 HRIM can exclude a primary motor disorder, most importantly achalasia, which, while rare, causes symptoms of regurgitation, heartburn and chest pain in addition to dysphagia.5,6 HRIM can determine hypomotility,7–10 weak esophago-gastric junction (EGJ) contractility10–13 and/or hiatus hernia subtype morphology,3,14 which are features of primary GER disease.
The role of HRIM for establishing ‘risk’ of post-operative dysphagia is unclear. In pediatric series, dysphagia ranges from 12% to 40%.15,16 Dysphagia can mar otherwise successful GER treatment. Prediction of dysphagia risk would inform the choice of other treatments over fundoplication.
We hypothesized that esophageal ‘pressure-flow’ anomalies may predict dysphagia.17,18 We therefore investigated dysphagia and esophageal motor function in children with GER disease undergoing fundoplication.
Materials and methods
Children who had been referred for Nissen fundoplication surgery were included in the study. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the Women's and Children's Hospital Human Research Ethical Committee (approval number REC 2345, February 2015). Informed consent was obtained.
Assessment of dysphagia symptoms
A dysphagia questionnaire modeled on the score of Dakkak and Bennett was used. 19 This modified Dakkak score assessed dysphagia for nine different food consistencies (water to meat; scale 0–45; score > 10 = dysphagia) 20 and was mostly completed by patients with parental assistance.
Esophageal function testing
Esophageal function was assessed by HRIM catheter (25 1-cm-spaced pressure sensors and 12 impedance; Unisensor USA Inc, Portsmouth, NH). Data were recorded at 20 samples/s (Medical Measurement Systems, The Netherlands).
Patients were studied off proton pump inhibitors (PPI) (minimum 72 h) and in the semi-reclined position. The protocol was 5–10 × 5 ml boluses of liquid (saline) and viscous (‘EFT Viscous’, Sandhill Scientific, USA) and 3–5 × 2 cm solid (bread with saline topically added to increase conductivity).
Pressure topography
Motility disorders were diagnosed by Chicago Classification (V3.0). 3 Esophageal pressure topography metrics were derived, namely: (i) 4s integrated relaxation pressure of the EGJ (IRP4s, mmHg); (ii) contractile front velocity of the distal esophagus (CFV, cm/s); (iii) distal contractile integral (DCI, mmHg cm/s); and (iv) distal latency (DL, s). EGJ barrier function was assessed using the EGJ contractile index (EGJ-CI).12,13 EGJ morphology was defined as Type I (no lower esophageal sphincter (LES) to crural diaphragm (CD) separation), Type II (partial separation) or Type III (full separation). 3
Pressure-flow analysis
Pressure-flow analysis (Figure 1) was applied using purpose designed software (AIMplot v8, copyright T Omari) programmed in MATLAB (version 7.9.0.529 R2009b, The MathWorks Inc., Natick, MA, USA).17,20–22 The analyst selected key temporal and anatomical landmarks including: (i) swallow onset; (ii) esophageal proximal margin; (iii) transition zone; (iv) crural diaphragm and (v) stomach.
Derivation of pressure-flow metrics. Automated analysis was applied to each swallow within a region of interest (see inset Clouse plot, lower right). (a) A pressure topography iso-contour plot with superimposed lines showing the position of the nadir impedance (thick purple line; indicating peak distension) and contractile peak (thick red line) over time. The analyst fine-adjusted the landmarks, paying particular attention to the transition zone (TZ), the contractile deceleration point (CDP; yellow star) and crural diaphragm (CD). (b) Intrabolus distension pressure during bolus transport was determined by pressure at nadir impedance which was measured along the esophagus. The average distension pressure (DP) was determined within three anatomical regions approximating the different phases of bolus transport. These were accommodation (DPA, pressures proximal to TZ), compartmentalized transport (DPCT, pressures TZ to CDP) and esophageal emptying (DPE, pressures from CDP to CD). (c) Effectiveness of bolus clearance was determined from TZ to CDP based on the impedance ratio (IR = nadir impedance divided by impedance at contractile peak). A higher IR equates to less effective bolus clearance. (c) Bolus flow latencies and clearance pressures were determined based on the pressure and impedance recordings at the CDP level. Swallow to distension latency (SDL) was measured from swallow onset to nadir impedance and distension to contraction latency (DCL) from NI to luminal clearance/closure corresponding to recovery of impedance to 50% from baseline (see plot lower left). The ramp pressure (RP) was determined within the distal esophagus (sensors within distal 25% of the TZ to CDP length) and defined by the mean gradient of pressure change over time from maximum distension (NI) to luminal closure (see plot lower middle, note impedance presented relative to baseline and reversed in direction). Variables DCL, DPE and RP were combined to derive the pressure-flow index (PFI) composite score.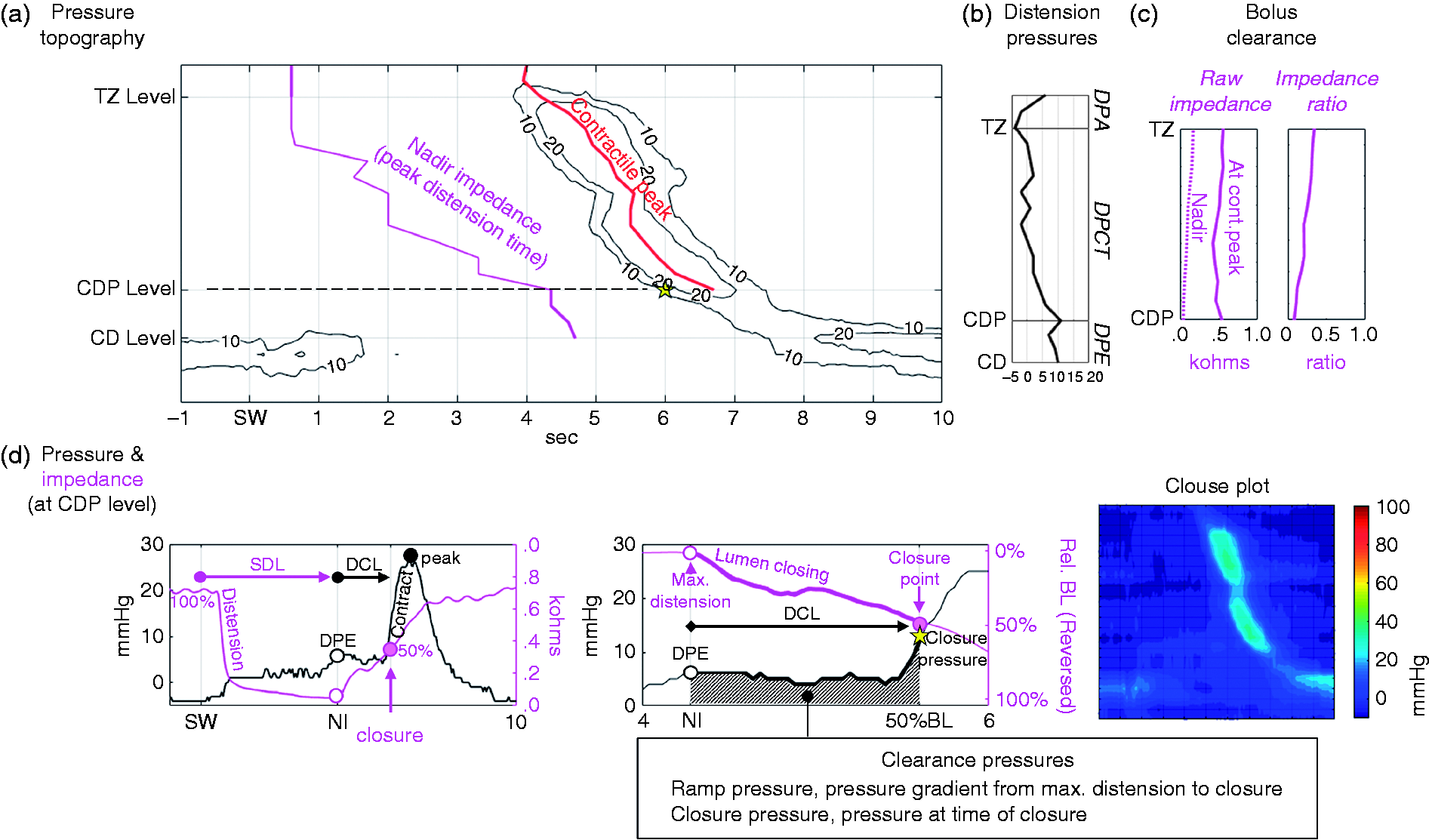
Five classes of variable were then derived as described below (see Figure 1):
Intrabolus distension pressures (DP) during bolus transport were determined by pressure at nadir impedance. These were DP bolus accommodation (DPA, pressures UES to transition zone (TZ)), DP compartmentalized transport (DPCT, pressures TZ to contractile deceleration point (CDP)) and DP esophageal emptying (DPE, pressures CDP to CD; see Figure 1(b)). Bolus clearance was determined by impedance ratio (IR = nadir impedance divided by contractile peak impedance). When bolus residual is present, then the impedance at the contractile peak has a lower value, hence IR becomes higher indicating less effective bolus clearance (see Figure 1(c)).
23
Bolus flow latencies were determined using pressure and impedance at the CDP level. These were swallow to distension latency (SDL) and distension to contraction latency (DCL) (Figure 1(c)). Pressure generation during bolus clearance (or clearance pressures) was measured within the distal esophagus. The closure pressure (CP) was the pressure at luminal closure and the ramp pressure (RP) was the mean gradient of pressure during closure (Figure 1(c)). Timing of luminal closure was defined by the 50% impedance recovery, a validated criterion in widespread use.
24
A pressure-flow index (PFI) composite score was derived using the following formula:
Trans EGJ bolus flow time (BFT) was estimated based on the method of Lin.
25
A shorter BFT indicates reduced esophageal emptying period.
Other assessments
Information regarding clinical history was garnered from clinical notes and correspondence. Patients typically underwent 24-hour reflux monitoring (pH only, pH-MII probe), barium swallow radiology study and upper GI endoscopy. Available biopsy specimens were scored for presence of eosinophils. Eosinophilic esophagitis (EoE) was defined as ≥ 15 eosinophils per high power field (eos/hpf). 26
Post-operative assessments
The families of patients were contacted post-operatively and invited to complete a Dakkak questionnaire. Those who had not already undergone manometry investigation during the post-operative period were invited to repeat a HRIM. A medical record review was undertaken to determine post-operative therapy and investigations indicative of symptom recurrence.
Statistical analysis
Analysis was performed using SPSS (IBM Corporation, USA). Data are means (standard deviation) if normally distributed (passed Shapiro–Wilk normality testing) or median (interquartile range). Spearman's correlation rho investigated relationships between continuous variables and dysphagia scores. Group comparisons were performed using t-test or Mann–Whitney U-test. To compare pre- versus post-operative findings across consistencies, repeated measures ANOVA with logarithmic transformation was performed on data failing normality testing. Receiver operator curve analysis determined if larger PFI scores indicated evidence for a dysphagia using curve area, sensitivity and specificity. Only p-values ≤ 0.099 are reported.
Results
Patients
Twenty-seven children with GER disease (aged 0.8–16.1 years) were originally identified. However, 11 children were excluded due to missing assessments or they did not undergo surgery. Three had a poor quality HRIM; two, both aged one year, were unsettled rendering the measurement uninterpretable and one, aged 16 years, was not able to complete the protocol.
The final dataset included 13 children who were aged 6.8–15.5 years (average 12.5 years old) at initial investigation. The patients were between four months and 4.7 years (average 1.4 years) post-surgery when followed up symptomatically by Dakkak questionnaire; six patients were over one year post-op at follow-up.
Pre-operative clinical findings
Summary of clinical findings from 13 children who received gastroesophageal reflux (GER) diagnostic work-up and complete high-resolution esophageal impedance manometry (HRIM) protocol prior to receiving anti-reflux surgery and in whom followed up to assess bolus hold-up symptoms was achieved.
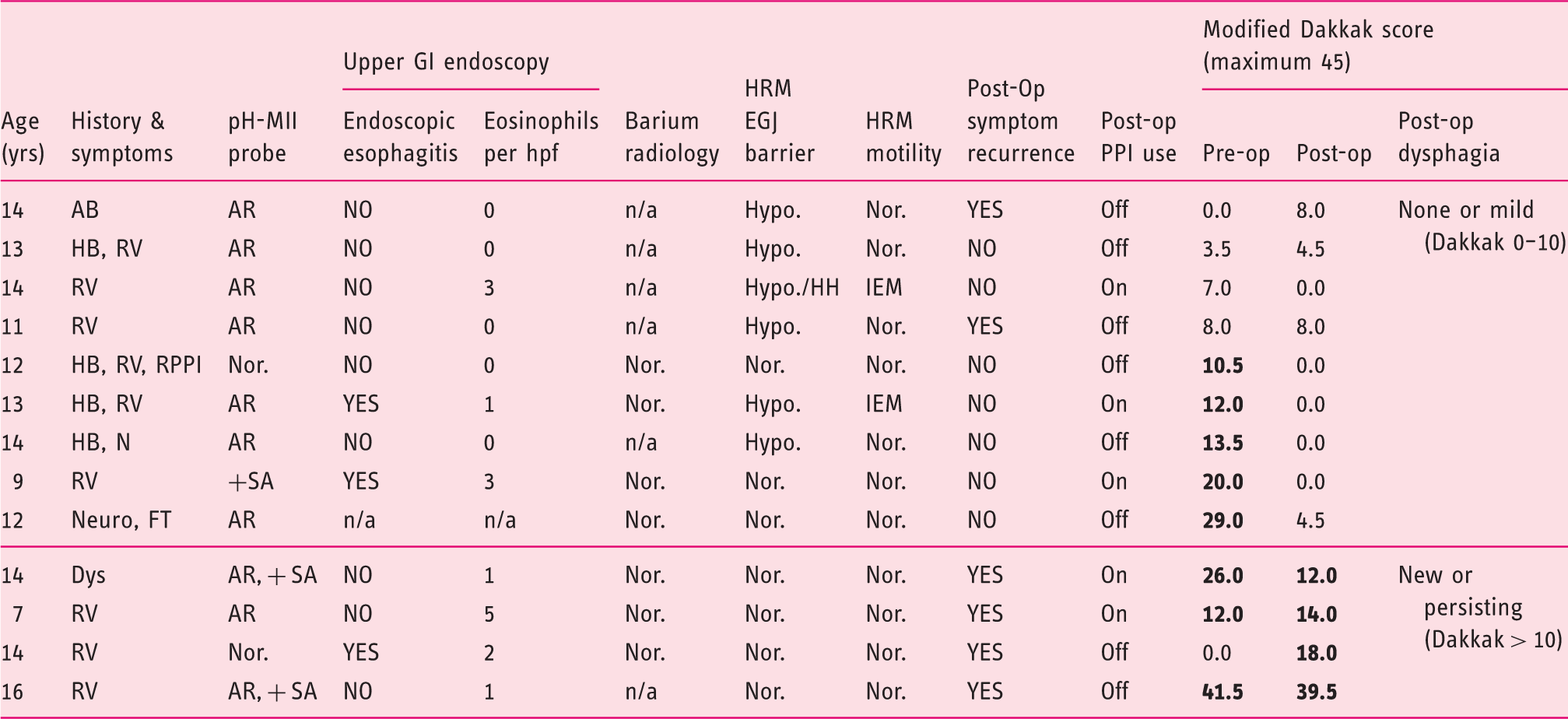
Modified Dakkak scores, which define bolus hold-up perception, are shown in the far-right columns. The four patients reporting new or ongoing swallowing difficulties are ranked by their post-operative Dakkak score, other patients ranked by pre-operative Dakkak score.
Abbreviations: n/a: not available; Nor.: normal findings.
History: Neuro, neurological patient; HB, heartburn; RV, regurgitation and/or vomiting; N, nausea; Dys, dysphagia; FT, failure to thrive; IC, irritability and crying; AB, acid brash; RPPI, refractory to PPI.
pH-MII GER: AR, abnormal acid GER; NAR, abnormal non-acid GER; + SA, positive symptom association probability.
HRM: Hypo, hypotensive EGJ; HH, hiatus hernia morphology; IEM, ineffective esophageal motility.
Pre-operative dysphagia questionnaire
Correlation esophageal function variables recorded pre-operatively with dysphagia symptoms reported pre-operatively and post-operatively.
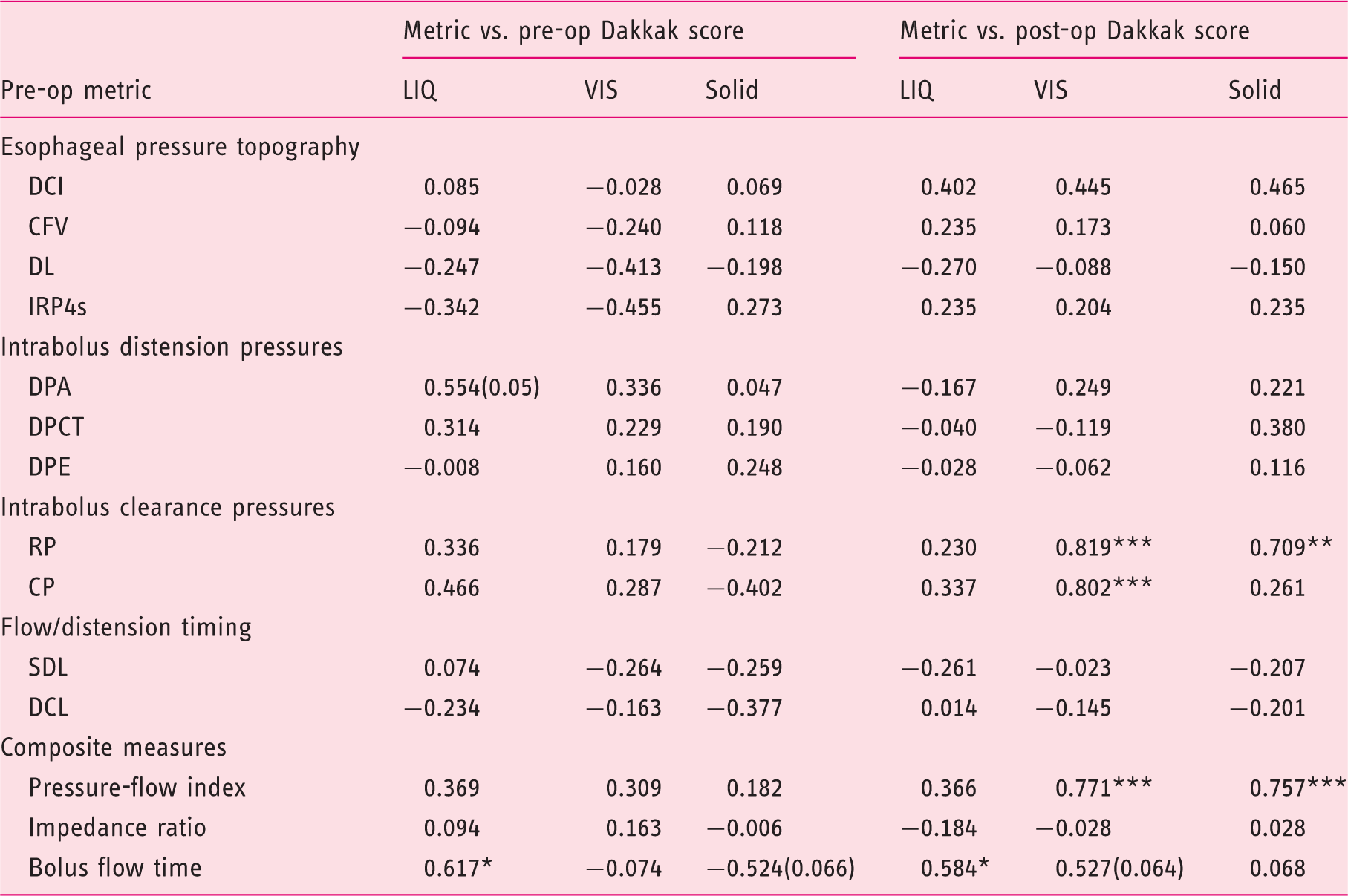
Spearman's correlation rho based on data from 13 patients with complete HRIM studies for all consistencies.
CFV: contractile front velocity; CP: clearance pressure; DCI: distal contractile integral; DCL: distension to contraction latency; DL: distal latency; DPA: DP bolus accommodation; DPCT: DP compartmentalized transport; DPE: DP esophageal emptying; IRP4s: 4s integrated relaxation pressure; RP: ramp pressure; SDL: swallow to distension latency.
*Indicates significant correlation (two-tail; *p < 0.05, **p < 0.01, ***p < 0.005).
Post-operative dysphagia questionnaire
The severity of post-operative dysphagia symptoms was not significantly correlated with the time of follow-up (r = –0.084). Six patients were assessed >1 year post-operatively and their dysphagia scores were not different from those of patients surveyed at earlier time points (Dakkak 2 [0, 23] > 1 year vs. 8 [0, 12] < 1 year, t = –0.221, p = n.s.).
Five of eight patients with pre-operative dysphagia improved post-operatively decreasing their Dakkak scores to <10 (Table 1). However, four patients reported significant dysphagia post-operatively; three with persisting dysphagia and one with ‘new’ dysphagia (a 14-year-old boy with Dakkak 0 pre-operatively increasing to Dakkak 18 post-operatively).
Surgery reduced esophageal acid exposure in six patients investigated by pH-probe after surgery on grounds of symptom recurrence (Figure 2). These patients had greater dysphagia post-operatively than other patients (Dakkak 13[8, 23] in refractory patients vs. 0[0, 2.2] in others, t = 3.095, p = 0.001), suggesting that post-operative dysphagia may have contributed to post-operative investigation.
Reflux monitoring findings in six patients investigated by pH or pH-MII probe pre- and post-operatively due to symptom recurrence. a) Total number of acid GER episodes, b) reflux index (%time pH<4) and c) total number of impedance detected liquid GER episodes Wilcoxon Signed Rank Test standardized t- and p-value shown. Total liquid GER data (in c) are incomplete for four patients due to pH-MII not being performed pre-operatively.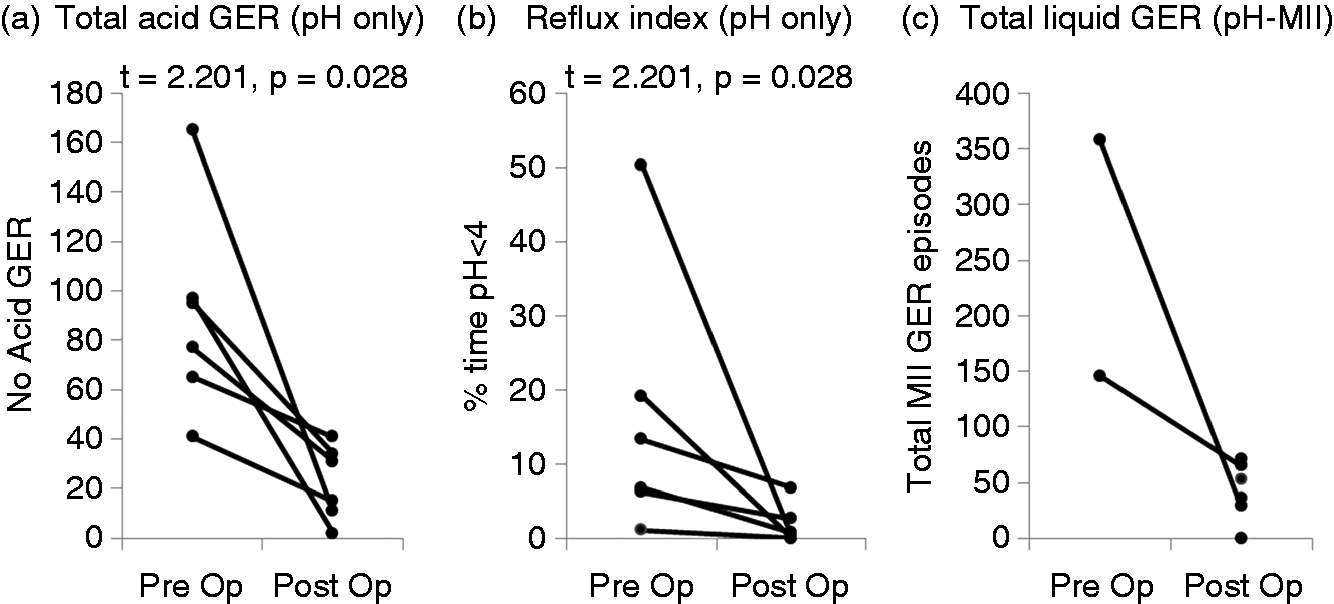
Pre-operative findings associated with symptoms of post-operative dysphagia
Routine reflux investigations did not discriminate post-operative dysphagia. However, manometric features that would normally corroborate a diagnosis of primary GER disease, such as evidence of IEM or EGJ barrier dysfunction, were never seen in the patients reporting dysphagia (Table 1). No patients raised clinical suspicion of rumination syndrome.
Of the parameters measured pre-operatively, higher RP, CP and PFI were correlated with post-operative Dakkak score and, for these variables, correlations using viscous consistency were superior (Table 2). Viscous bolus correlations were similarly apparent within patient sub-groups that were assessed < 1year post-op (RP: r = 0.764, p = 0.046; CP: r = 0.764, p = 0.046; PFI: r = 0.837, p = 0.019) and >1 year post-op (RP: r = 0.820, p = 0.046; CP: r = 0.942, p = 0.005; PFI: r = 0.880, p = 0.021). Grouped analysis showed that pre-operative viscous bolus clearance pressures were higher in those who had post-operative dysphagia compared with those who did not have post-operative dysphagia (RP: 47 [27, 71] mmHg/s vs. 13 [10, 18] mmHg/s, respectively, p = 0.002; CP: 72 [48, 100] mmHg vs. 31 [25, 38] mmHg, respectively, p = 0.002). BFT was longer pre-operatively in patients with post-operative dysphagia (Table 2).
To clarify and explain our main results we provide two case examples that are illustrative of our findings.
Case 1: A child with pre-operative dysphagia that resolved (Figure 3(a) to (d))
A nine-year-old female with regurgitation, esophagitis and a positive SAP to heartburn. Pre-operative HRIM was considered normal (no hiatus hernia, EGJ-CI 43 mmHg.cm, mean liquid DCI 1960 mmHg.cm.s and IRP4s 12 mmHg). The patient reported dysphagia pre-operatively (Dakkak 20), which resolved following fundoplication (Dakkak 0). The patient was successfully treated.
Pre-operative recordings and pressure-flow data from example cases: Case 1 with pre-operative dysphagia that resolved post-operatively and Case 2 with significant persisting dysphagia. Tracings are based on recordings of 5-ml viscous bolus swallows. (a–e) Esophageal pressure topography of the distal esophagus with axial location of contractile deceleration point (CDP) marked. (b and f) Pressure (black) and impedance (purple) profiles over time at the level of the CDP. (c and g) Pressure (black) and the impedance relative to baseline (purple) for the period 1 s before to 0.5 s after the 30-mmHg iso-contour (the direction of impedance change is reversed compared to b and f; 100% is pre-swallow baseline, 0% is maximum distension and 50% defines luminal closure). Note that the impedance-defined luminal closure (50%) occurs relatively later in time for Case 2, resulting in greater clearance pressures being generated as the lumen closes. (d and h) The relationship between relative impedance (x axis) and pressure (y axis) over time. As seen in d, from Case 1, the lumen opens and then closes without any change in clearance pressure (known as isotonic contraction). As seen in h, from Case 2, the lumen opens and then closes with clearance pressures increasing (known as auxotonic contraction). (i) Bar charts of patient Dakkak scores and the average data for the relevant pressure-flow metrics derived for liquid and viscous bolus swallows. Note, markedly higher clearance pressures (RP and CP) for Case 2.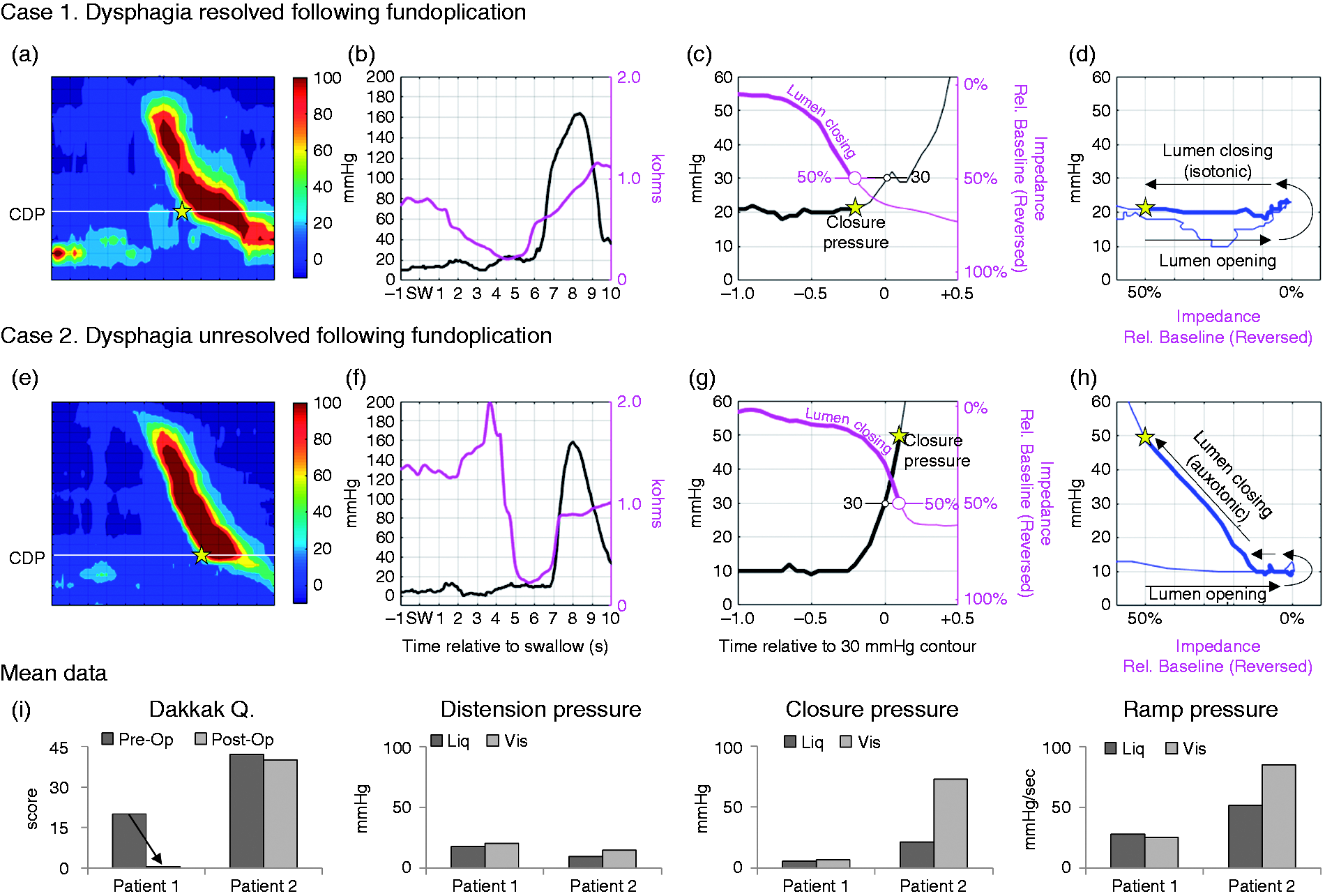
Case 2: A child with pre-operative dysphagia that did not resolve (Figure 3(e) to (h))
A 16-year-old female with regurgitation, abnormal reflux index (19.2%) and positive SAP to regurgitation. Pre-operative HRIM was considered normal (no hiatus hernia, EGJ-CI 32 mmHg.cm, mean liquid DCI 2541 mmHg.cm.s, IRP4s 10 mmHg). The patient reported significant dysphagia pre-operatively (Dakkak 41.5), which did not resolve following fundoplication (Dakkak 39.5). Repeat pH-probe demonstrated normalization of reflux parameters.
When Case 1 and Case 2 were compared (Figure 3), clearance pressures (CP and RP) were much higher pre-operatively for the viscous swallows in Case 2 (Figure 3(i)). In Case 2, luminal closure occurred later in time (compare Figure 3(b) vs. Figure 3(f)) and auxotonic contraction was observed (Figure 3(h)).
Effects of surgery on biomechanical measures
Effect of fundoplication surgery on esophageal function variables. Data from seven patients participating in repeat study. Data are estimated marginal mean (95% confidence interval of difference). Statistics for RM-ANOVA shown.
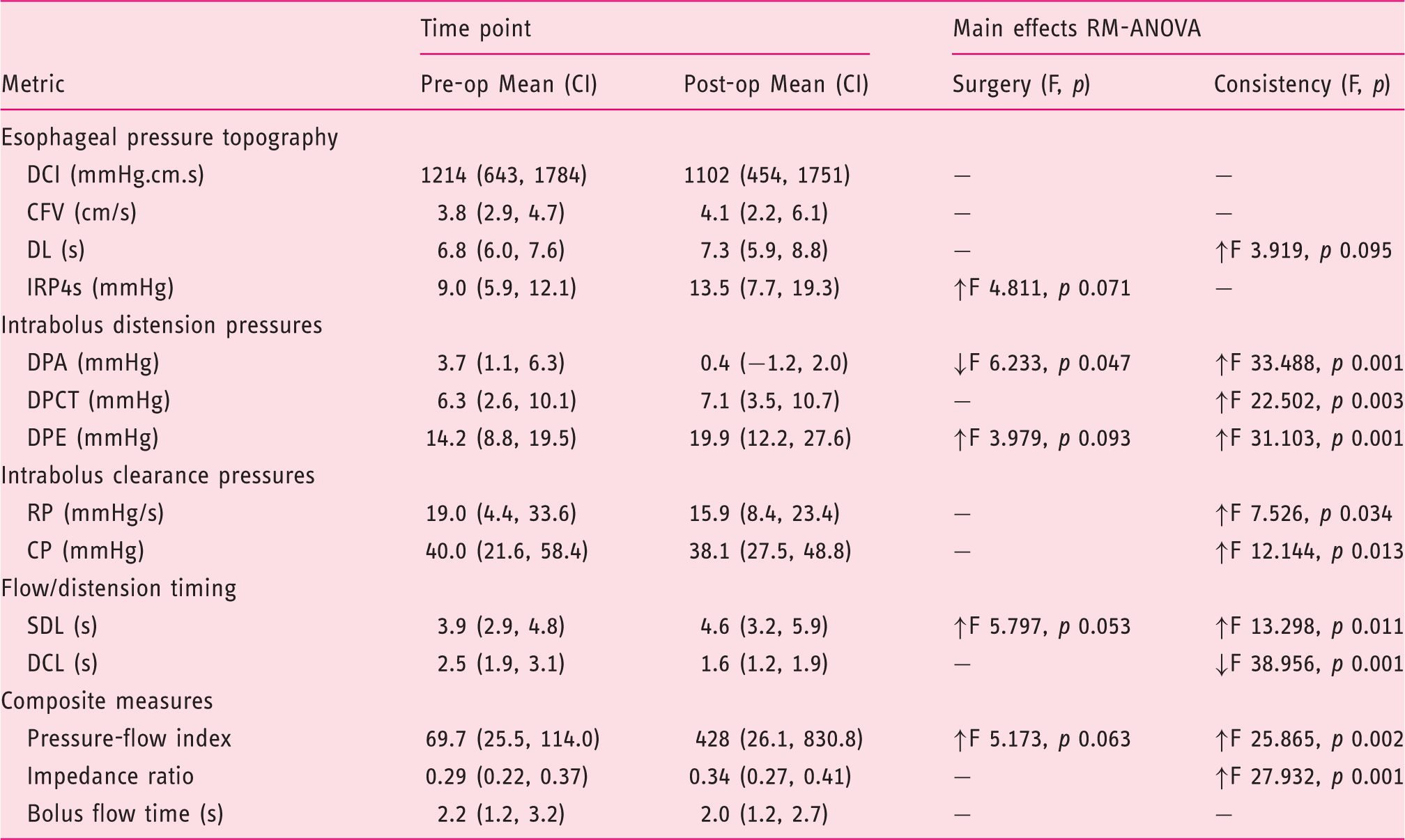
Main effects in relation to surgery and bolus type on swallow function variables are indicated; – indicates no effects; ↑↓ indicates the directionality of the effects.
CFV: contractile front velocity; CP: clearance pressure; DCI: distal contractile integral; DCL: distension to contraction latency; DL: distal latency; DPA: DP bolus accommodation; DPCT: DP compartmentalized transport; DPE: DP esophageal emptying; IRP4s: 4s integrated relaxation pressure; RP: ramp pressure; SDL: swallow to distension latency.
Discussion
Dysphagia and esophageal function were investigated in children undergoing fundoplication surgery. Our findings were: (i) patients reported symptoms of dysphagia pre-operatively; (ii) fundoplication surgery decreased dysphagia in most; however (iii) some had post-operative dysphagia and were distinguishable by elevated bolus clearing pressures.
When assessed pre-operatively, dysphagia was common. Improvement after surgery suggests GER was a causal factor. Few parameters were linked to pre-operative dysphagia before reflux, as an influencer of symptoms, was mitigated. Patients with manometry evidence of primary GER disease (low EGJ-CI, hiatus hernia EGJ morphology and/or an IEM subtype) did not have post-operative dysphagia. While those with a normal motility diagnosis and higher clearing pressures had higher post-operative dysphagia scores. This association has been previously reported in post-operative17,18 and non-obstructive dysphagia2,27,28 (note, defined by the parameter ‘intrabolus pressure slope’).
Prognostic value of the pressure-flow index score to predict levels of dysphagia.
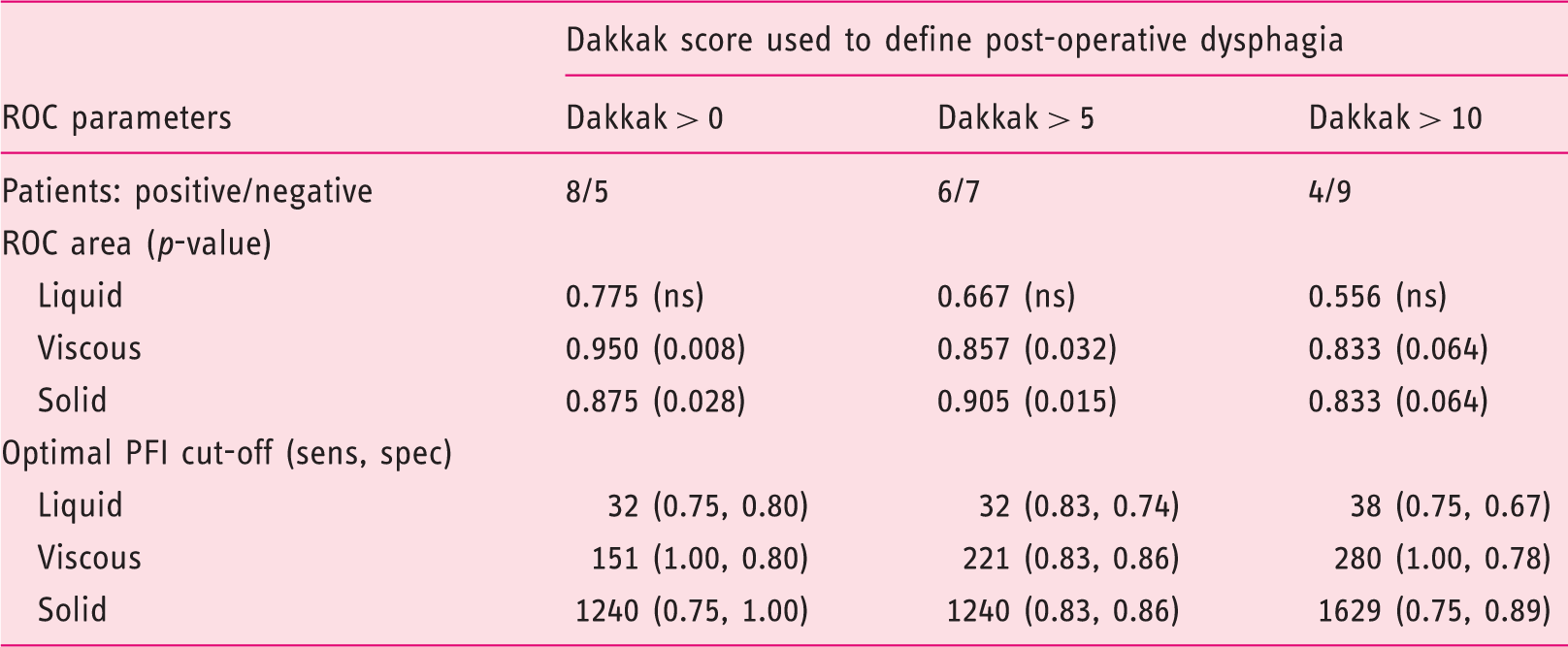
Receiver operator curve analysis determining if larger values of the PFI result indicate stronger evidence for a dysphagia.
Esophageal resistance to bolus flow during bolus transport occurs on a continuum from unimpeded flow at one extreme, to totally obstructed flow at the other extreme. Obstructed luminal flow can be deduced by the presence of high compartmentalized DPs (for example, in relation to focal strictures and EGJ outflow obstruction). However, if bolus flow of a viscous or solid bolus is impeded rather than obstructed, then clearance pressures that quantify bolus pressurization superior to the bolus midpoint (i.e. toward and at the bolus tail) are important because they drive the bolus movement. A high clearance pressure suggests that the contraction, rather than being isometric (pressure rise occurs with a closed lumen after the bolus has passed), is instead auxotonic (pressure increases with a closing lumen as the bolus passes, see Figure 3(h)).29,30 The recording of auxotonic pressure-flow phenomena demonstrates that the demands placed on the esophageal muscles to do mechanical work (expend energy) during bolus clearance are greater because the bolus is, for one or more reasons, less easily transportable. Various intrinsic and extrinsic factors to the bolus material influence this. Bolus rheology (resistance to flow) is important; solids resist flow. Additionally, other superimposed factors may be relevant, namely: (i) the size of luminal aperture; (ii) the stiffness of the esophageal wall; and (iii) the surface ‘tackiness’ of the mucosal lining due to the presence/absence of luminal secretions which influence surface interaction and lubrication.
The mechanism(s) by which higher clearance pressures may directly lead to bolus perception are unclear. Sensory afferent mechanisms are largely insensitive to common isotonic and isometric wall states. 29 However, the active tension produced in relation to a prolonged auxotonic state may generate wall tension sufficient to produce a noxious stimulus. In other words, a bolus being transported that has been orally processed to be soft, moist and slippery (via saliva coating and mucosal secretions lining the esophagus all the way down) should not be felt. Conversely, a bolus that is hard, dry and resistive should be felt in order to generate alarm sensations that will alter eating behavior. However, in certain pathophysiological situations, a soft, moist and slippery bolus may be rendered less easy to transport. If so, this will lead to the bolus being felt because its transport is being impeded changing the wall state.
Elevated clearance pressures may indicate dysregulated bolus transport, due to changes to the enteric nervous system (ENS) and/or the muscle layers. Muscle relaxation ahead and then contraction behind the moving bolus are dependent upon normal functioning of the ENS.31,32 Rapid swallow testing reveals aberrant ENS inhibition and may predict dysphagia risk. 33 Esophageal distension in-vivo can reveal abnormal intramural motor patterns in dysphagic patients with otherwise normal primary peristalsis. 34 Contractile decoupling and selective contractile dysfunction(s) of the circular and longitudinal muscle layers are reported in various disorders 35 and may perturb the normal ability of the esophagus to biomechanically distend. 35 Hence, contractile decoupling can produce a motor pattern suggestive of impaired inhibition, even though the inhibitory apparatus is functioning normally. Irrespective of the underlying mechanism(s), the lack of ability of the lumen to distend would impede normal bolus flow leading to high clearance pressures.
Strengths of our study are the detailed characterizations of symptoms, reflux, and motility and pressure-flow phenomena. Our data support the findings from previous studies;17,18 however, the variation of follow-up time is a significant limitation. A larger prospective study, including longitudinal measurements at fixed early and long-term post-operative time points, is clearly needed. Data from very young patients were not included limiting generalizability to all pediatric GER disease. There are challenges for generating quality HRIM data in the very young because patients must tolerate the procedure and repeat-swallow boluses on command.
In conclusion, dysphagia symptoms were common in pediatric GER disease patients prior to anti-reflux surgery. In order to avoid symptom recurrence, re-investigations and further interventions, dysphagia symptoms should be attributed to GER. In patients with normal motility, elevated clearance pressures and/or PFI may predict post-operative dysphagia.
Footnotes
Acknowledgements
We thank Mrs G Seiboth, Mrs K Lowe and Ms S Kritas for assistance with performance of HRIM studies, Dr Kyra Sierakowski for assistance with obtaining patient histories and Dr Junko Fujino for assistance with reviewing endoscopy images.
Declaration of conflicting interests
Omari holds inventorship of patents that cover some of the analytical methods described. The other authors have no conflicts of interest relevant to this article to disclose.
Funding
Supported by The Women’s & Children’s Hospital Foundation.
Ethics approval
The study protocol was approved by the Women s and Children’s Hospital Human Research Ethical Committee (approval number REC 2345, February 2015).
Informed consent
Informed consent was obtained from each patient’s parent or guardian.