Abstract
Background
The diagnosis of laryngopharyngeal reflux is currently based on a combination of the patient history of multichannel intraluminal impedance and ambulatory pH (MII-pH); however, none of these findings alone is specific for the diagnosis of laryngopharyngeal reflux. We aimed to compare the baseline characteristics and esophageal baseline impedance values between patients with and without laryngopharyngeal reflux symptoms.
Methods
We retrospectively analyzed data from two groups of patients with laryngopharyngeal reflux according to their reflux finding score (RFS) as scored by ENTs. Control patients were nonerosive reflux disease patients without laryngopharyngeal reflux. All MII-pH parameters and baseline impedance were analyzed from six levels and the proximal and distal baseline impedance and the ratio of the proximal to distal baseline impedance levels was calculated.
Results
Altogether 123 patients with laryngopharyngeal reflux and 49 control patients were included. A total of 81 of 123 patients had RFS ≥ 7, and 42 of 123 patients had RFS < 7. Baseline impedance analysis showed that patients with laryngopharyngeal reflux symptoms had significantly lower proximal baseline impedance values (1997 ± 51 vs 2245 ± 109, p < 0.05) than the control group. Additionally, patients with laryngopharyngeal reflux symptoms had a significantly lower proximal-to-distal ratio (1.28 ± 0.05 vs 1.53 ± 0.09, p < 0.05). In the subgroup analysis, patients with RFS < 7 were found to have a significantly lower acid exposure time than either the patients with RFS ≥ 7 (3.85 ± 0.65 vs 8.2 ± 1.52, p < 0.05) or the control group (3.85 ± 0.65 vs 6.1 ± 0.81, p < 0.05). Additionally, patients with RFS ≥ 7 had significantly lower proximal baseline impedance levels than the control group (1970 ± 63 vs 2245 ± 109, p < 0.05).
Conclusions
Patients with pathologic laryngopharyngeal reflux symptom scores had lower proximal baseline impedance levels and lower proximal-to-distal ratios, which may reflect the proximal mucosal noxious effect of the refluxate. These results may indicate that laryngopharyngeal reflux symptoms may be due to chronic acid exposure in the proximal segments of the esophagus, and the proximal-to-distal ratio may be used as a new metric for diagnosis.
Keywords
Introduction
Gastroesophageal reflux disease (GERD) occurs when the reflux of stomach contents causes troublesome symptoms and/or complications. 1 The prevalence of GERD is between 9–26% in western countries and 1.1–2.5% in eastern countries. 2 In addition to typical manifestations such as heartburn and regurgitation, GERD can also cause atypical symptoms such as the development of a cough, asthma, and reflux-induced laryngitis which may be defined as laryngopharyngeal reflux (LPR). Interestingly, the incidence of LPR has dramatically increased over time. 3 It is well known that LPR is significantly associated with chronic cough, chronic laryngitis, and asthma, but is not believed to be the sole cause of these symptoms.
Due to the lack of a gold standard diagnostic test, the diagnosis of LPR is currently based on a combination of patient history of LPR complaints (hoarseness, excessive laryngeal mucus, throat clearing, globus sensation, cough) and either laryngoscopic findings (posterior laryngeal edema, true vocal fold edema, and pseudosulcus) or multichannel intraluminal impedance and ambulatory pH (MII-pH) monitoring findings associated with GERD. 4 However, laryngoscopy, esophagogastroduodenoscopy, and pH monitoring have been demonstrated to be unreliable tests for the diagnosis of LPR.5–7 There are studies that aimed to determine the diagnostic accuracy of laryngoscopic findings or MII-pH in LPR.4,8–10 However, the clinical signs attributable to LPR detected by laryngoscopy have been shown to be non-specific and may also be seen in healthy patients.3,8,11 Thus, it has been shown that there is an overall lack of correlation between the reflux finding score (RFS) and MII-pH and that RFS is not specific for reflux in untreated volunteers.
Recently, baseline impedance (BI) has been shown to reflect the exposure of the esophagus to gastroduodenal ingredients and to reflect the mucosal injury of long-term esophageal exposure to injurious reflux contents. 12 Consistently, distal esophageal BI values are shown to be lower in GERD patients than in healthy controls.13–15 However, to the best of our knowledge, the diagnostic value of BI values in patients with LPR symptoms and its relationship with LPR symptom severity detected with RFS has never been studied. In our study, we aimed to compare the baseline characteristics and esophageal BI values between patients with and without LPR in comparison with RFS. We hypothesized that, similarly to distal BI values in GERD diagnosis, the proximal BI values could improve discrimination of patients with and without LPR. As the inter-individual variability of BI is considerable we also tested the ratio of proximal: distal BI as a marker of disease.
Patients and methods
Data collection
We retrospectively analyzed the database of the Ege University Reflux Outpatient Clinic between 2008–2015. Patients who had undergone esophagogastro-duodenoscopy, esophageal manometry, and MII-pH were accepted for the analysis and only nonerosive reflux disease (NERD) patients with typical symptoms (the control group) and patients with NERD with LPR symptoms and with RFS analysis were included in the study. For the selection of LPR patients, patients with other causes of reflux and patients with a history of endoscopically documented hiatal hernia (>2 cm), Barrett's esophagus, or erosive esophagitis, or with manometrically suspected esophageal motility disorders or other ENT pathologies were excluded from analysis (Figure 1). In the final analysis, the patients were divided into two groups: patients with LPR symptoms formed the study group, and patients without LPR symptoms and with typical symptoms (regurgitation and heartburn) formed the control group. We manually analyzed all data from patients with acid exposure (pathology was defined as time >4.2, DeMeeester score >14.7, number of reflux episodes >73).
16
Although this study was a retrospective analysis, we evaluated the proton pump inhibitor (PPI) response of patients by telephone follow-up of patients that completed physiological studies. We defined PPI responsiveness as higher than 50% positive response to typical symptoms of GERD and classified patients according to LPR symptoms.
Schematic presentation of the data analysis. LPR: laryngopharyngeal reflux; MII-pH: multichannel intraluminal impedance and ambulatory pH; RFS: reflux finding score; Sx: symptoms; UGE: upper gastrointestinal endoscopy.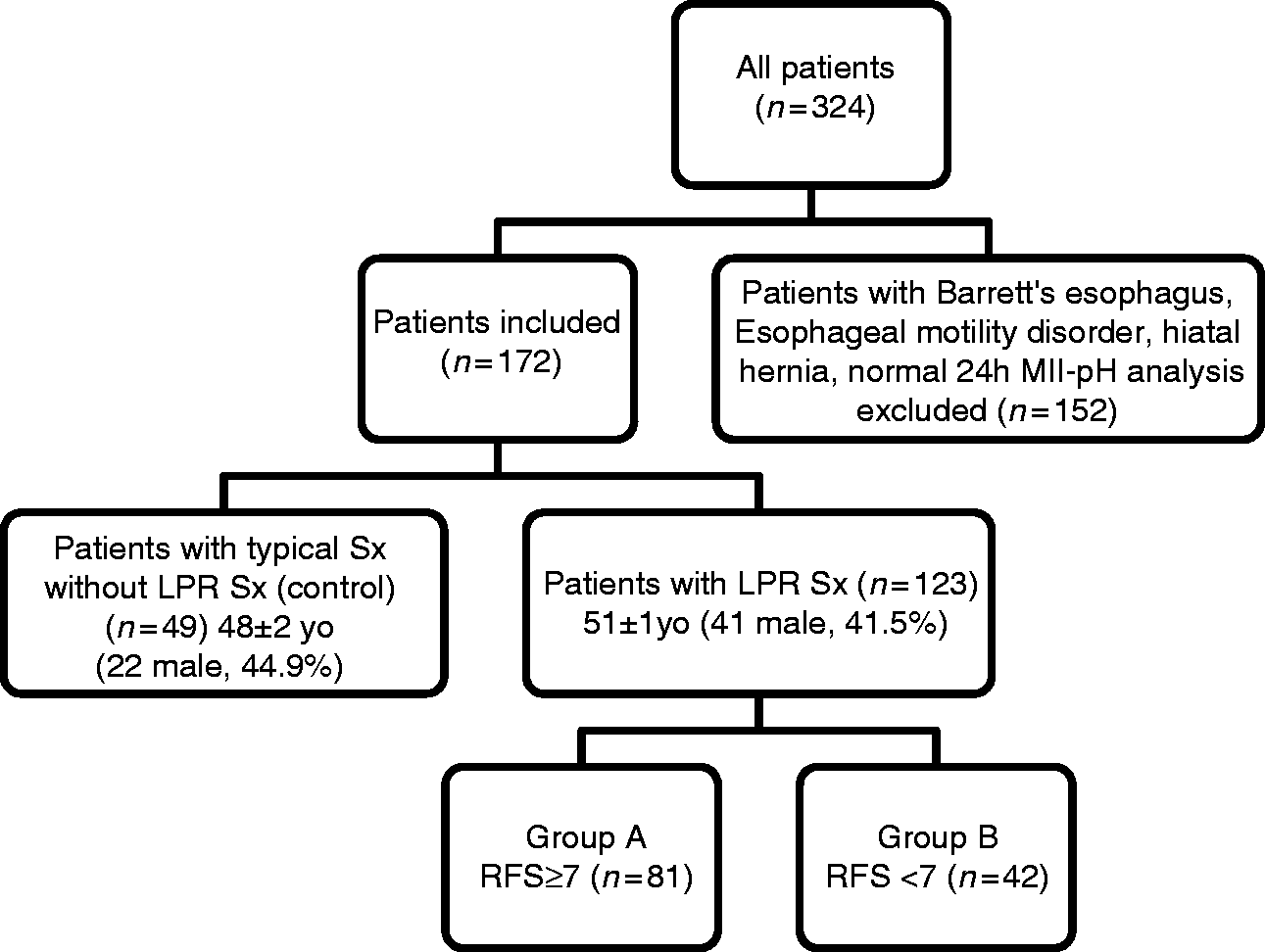
RFS
Two expert physicians performed routine videolaryngoscopic and Ear-Nose-Throat (ENT) examinations with a 90° rigid telescope (Karl Storz Laryngostrobe, Tuttlingen, Germany) without any knowledge of the patients’ other findings. Each patient’s findings were scored by RFS, a system proposed by Belafsky et al. 17 RFS is defined by subglottic edema, erythema/hyperemia, granuloma/granulation tissue, vocal fold edema, diffuse laryngeal edema, posterior commissure hypertrophy, ventricular obliteration, and excessive endolaryngeal mucus. The score ranges from normal laryngoscopic findings at 0 to a maximum score of 26. After patients with concurrent otolaryngological pathologies were excluded, patients who had RFS ≥ 7 were accepted as pathologic.
MII and 24-hour probe analysis
After at least six hours of fasting, all patients were subjected to conventional esophageal manometry with an eight-channel water-perfusion method, in which an infusion pump supplied distilled water to the capillary channels at a speed of 0.5 ml/min (circumferential eight-channel probe, MMS, Greenfield, The Netherlands) to evaluate the location of the lower esophageal sphincter (LES) and to differentiate motility disorders. Nitrates, anticholinergics, antacids, calcium channel blockers, prokinetics, and sedatives were discontinued 24 h prior to this process. Proton-pump inhibitors and H2 blockers were stopped at least seven days before the procedure. The MII-pH catheter had one antimony electrode placed such that the pH sensor was positioned 5 cm above the high-pressure zone of the LES. Impedance measurements were made 3, 5, 7, 9, 15, and 17 cm above the LES. Data were transmitted and stored on a portable data recorder (Ohmega, MMS Measurement systems, The Netherlands). Patients were monitored for at least 20 h and encouraged to eat regular meals and participate in routine activities. Changes in position (between supine and upright) and symptomatic events, including heartburn and regurgitation, were documented using buttons on the data recorder. Data were analyzed using available software (MMS Database software Ver 9.1w, Medical Measurement Systems B.V., The Netherlands). The 24-hour pH analysis, total acid exposure time (AET) (%), and DeMeester Score were analyzed with the software.
MII data analysis
In all MII data analysis, meal periods were excluded. Reflux was defined as having a 50% fall in at least two distal impedance sites. Proximal reflux was defined as when the reflux reached the sensor at 15 cm above the LES. All reflux episodes were analyzed manually and episodes not suitable for reflux were excluded. Reflux was categorized as either acid reflux (pH < 4) or weak/non-acid (pH ≥ 4) reflux
BI analysis
BI measurements were obtained for all segments of the esophagus (3, 5, 7, 9, 15, and 17 cm above the LES) over a period of six hours. Each period exceeding 30 s without swallowing or an episode of gastroesophageal reflux without artifact was selected, and the average value of the entire segment was analyzed within the sleeping period as described by Kessing et al. 14 We describe the measurement places as Z1 (17 cm above the LES), Z2 (15 cm above the LES), Z3 (9 cm above the LES), Z4 (7 cm above the LES), Z5 (5 cm above the LES), and Z6 (3 cm above the LES). Mean BI values of proximal segments (Z1 and Z2) and distal segments (Z5 and Z6) were compared between the groups. To determine the exposure levels of different esophageal segments, we analyzed the ratio of BI measurements between the proximal (Z1 and Z2) and distal (Z5 and Z6) segments of the esophagus as the proximal to distal ratio ((mean Z1 + Z2)/(mean Z5 + Z6)).
Statistical analysis
All parameters were expressed as the mean ± standard error of the mean (SEM). Variables were investigated using visual (histograms, probability plots) and analytical methods (Kolmogorov-Smirnov test) to determine if they were normally distributed. The parameters that were normally distributed were analyzed with the Student’s t test; other parameters that were not normally distributed were analyzed with the Mann Whitney U test. A value of p < 0.05 was considered to be significant. Welch analysis of variance (ANOVA) was used to compare parameters in the subgroup analysis. When overall significance was observed, pairwise post-hoc tests were performed using the Tamhane T2 test. All statistical analyses were performed using commercially available computer software (IBM SPSS Statistics for Windows, Version 22.0., IBM Corp., Armonk, New York, USA).
Results
Of the 324 eligible patients, 152 failed to meet the inclusion criteria (eight had Barrett’s esophagus, 81 had erosive esophagitis, 43 had hiatal hernia and 20 had both hiatal hernia and esophagitis) and were excluded (Figure 1). Therefore, a total of 123 patients with LPR symptoms and 49 patients with typical symptoms were included in this study and completed the GERD questionnaire (Figure 1). 18 Body mass index (BMI) was similar between groups (27.12 ± 4.4 vs 26.6 ± 5.5, p = 0.474). Seven of 49 (14%) patients with typical symptoms and 23 of 123 (18%) patients with LPR symptoms had BMI ≥ 30. A total of 53 of 123 (43%) patients with LPR and three of 49 (6%) patients with typical symptoms were smokers.
MII-pH analysis
The multichannel intraluminal impedance and pH monitorization analysis of patients.
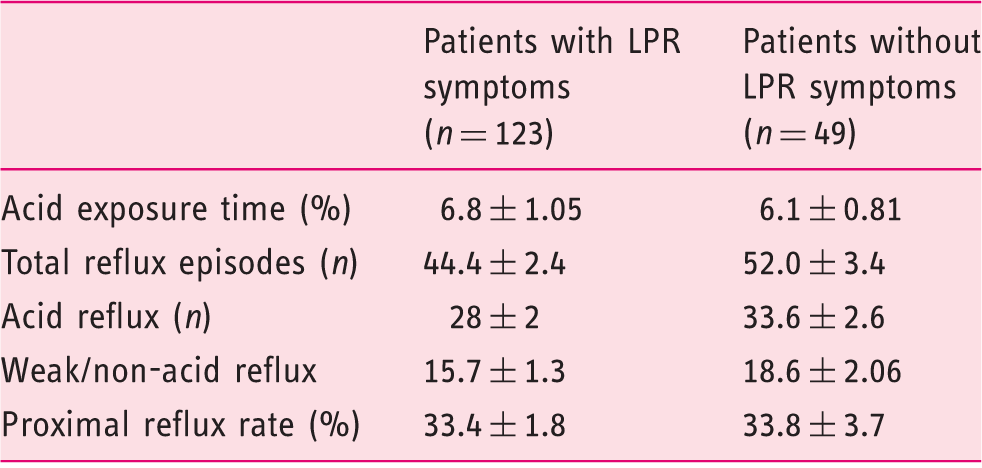
LPR: laryngopharyngeal reflux. The number of reflux episodes and amount of acid reflux were lower but not significant in patients with LPR symptoms; data are expressed as mean ± standard error of the mean (SEM); p > 0.05 for all comparisons.
Mean baseline impedance (BI) values and ratio of proximal vs distal BI in the different levels of the esophagus of patients according to the reflux finding score between groups.

GERD: gastroesophageal reflux disease; LPR: laryngopharyngeal reflux symptoms; RFS: reflux finding score. The proximal-to-distal ratio in patients with LPR was significantly lower than GERD patients without LPR; data are expressed as mean ± standard error of the mean (SEM); ap < 0.05 vs patients without LPR; proximal BI: mean of 15 and 17 cm above lower esophageal sphincter (LES); distal BI: mean of 3 and 5 cm above LES; proximal to distal ratio: proximal BI/distal BI.
Baseline impedance analysis
The mean baseline impedance (BI) values of patients and the ratio of proximal vs distal BI in the different levels of the esophagus.
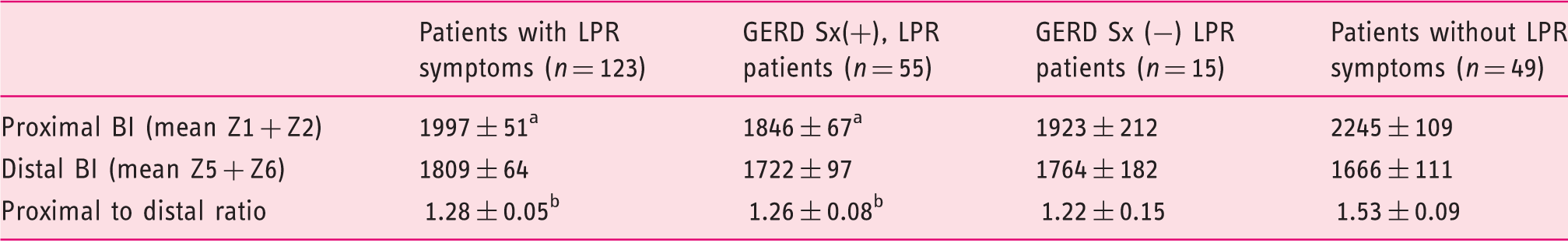
GERD: gastroesophageal reflux disease; LPR: laryngopharyngeal reflux; Sx: symptom. The proximal BI values were lower in patients with LPR symptoms. Additionally, the ratio of proximal BI to distal BI values was lower than the controls; data are expressed as the mean ± standard error of the mean (SEM); ap < 0.05 vs patients without LPR symptoms; bp < 0.01 vs patients without LPR symptoms; proximal BI: mean of 15 and 17 cm above lower esophageal sphincter (LES); distal BI: mean of 3 and 5 cm above LES; proximal to distal ratio: proximal BI/distal BI).
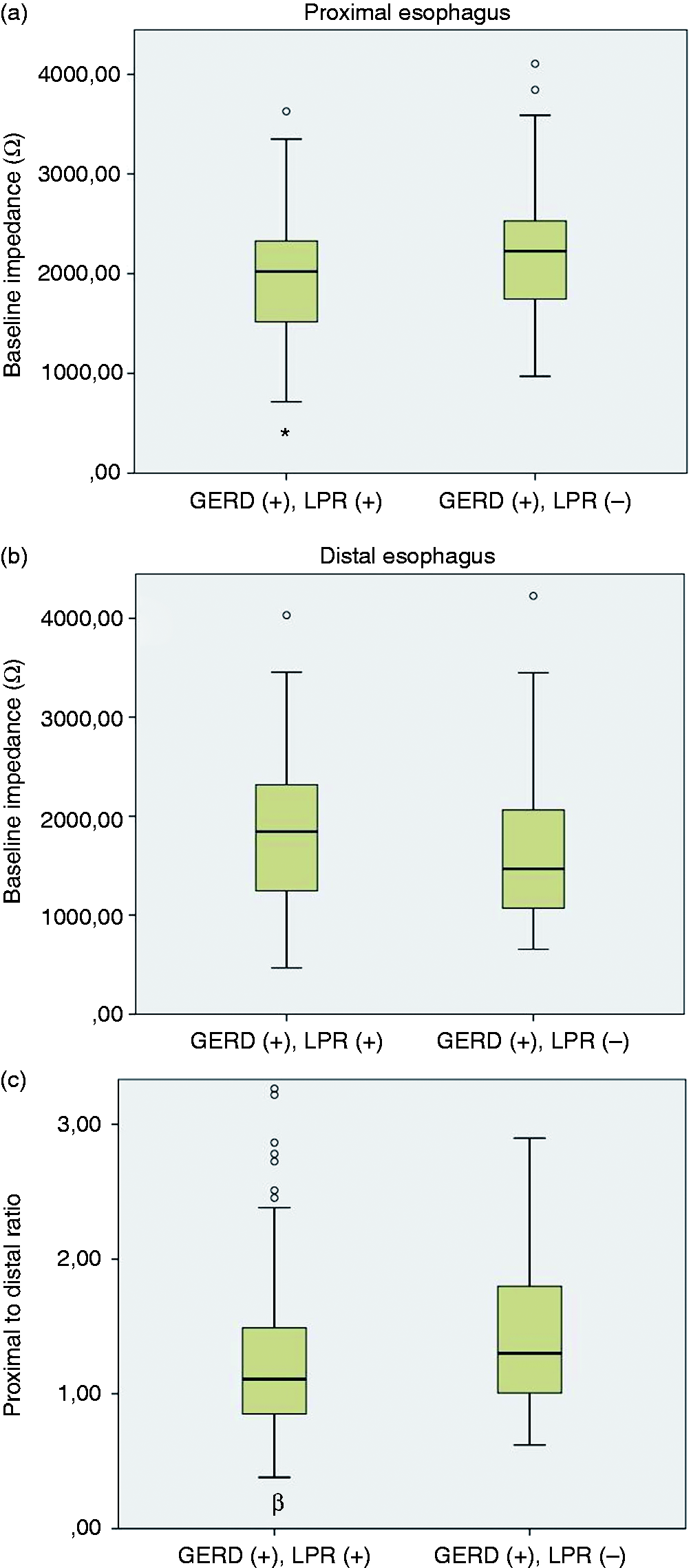
The comparison of baseline impedance (BI) values of (a) proximal esophagus, (b) distal esophagus and (c) proximal to distal ratio between patients with and without laryngopharyngeal reflux (LPR). The BI of proximal esophagus and the proximal-to-distal ratio were significantly lower in patients with LPR symptoms (data are expressed as the mean ± standard error of the mean (SEM)). *p < 0.05, βp < 0.01; proximal esophagus: mean BI of 15 and 17 cm above lower esophageal sphincter (LES); distal esophagus: mean BI of 3 and 5 cm above LES; proximal to distal ratio: proximal BI/distal BI. GERD: gastroesophageal reflux disease symptoms.
According to the RFS, the proximal and distal BI value were not different between groups (Table 2). But, similar to our previous findings, if patients with LPR symptoms were divided according to their RFS, LPR patients both with and without pathologic RFS showed significantly lower proximal to distal ratios than patients without LPR (Table 2).
To control the diagnostic accuracy of these results, we excluded LPR patients without typical symptoms (heartburn and/or acid regurgitation) and compared LPR patients with typical symptoms to the control group (Table 3). Data were collected from 70 of 123 (53%) patients and 55 of these patients had typical symptoms. When we compared these groups, we found similar results as previous findings that in patients without typical GERD symptoms, the proximal to distal ratio was lower but not significantly different in post-hoc analysis (1.22 ± 0.15 vs 1.53 ± 0.09, p = 0.056).
The comparison of the mean baseline impedance (BI) values in proton pump inhibitor (PPI) responsive patients.

LPR: laryngopharyngeal reflux; Sx: symptom. Note that the proximal BI value and proximal to distal ratio were significantly lower in the LPR patients. Data are expressed as the mean ± standard error of the mean (SEM); ap < 0.05, bp < 0.01 vs patients without LPR; proximal esophagus: mean BI of 15 and 17 cm above lower esophageal sphincter (LES); distal esophagus: mean BI of 3 and 5 cm above LES; proximal to distal ratio: proximal BI/distal BI.
Discussion
In this study, we evaluated differences in impedance analysis with the presence of LPR symptoms with normal or pathologic RFS and compared these differences to those in patients with typical reflux symptoms without LPR. Overall, the important key results of this study were the following: (a) the RFS and the endoscopic changes scored by ENTs were also correlated with acid exposure; (b) patients with LPR symptoms had lower proximal mucosal impedance compared to GERD patients without LPR symptoms; (c) the ratio of proximal BI value to distal BI was lower in patients with LPR; (d) the results were independent of the existence of typical symptoms in the LPR group; and (e) proximal to distal ratio seems to be more reliable than RFS.
Diagnosing GERD in patients with LPR remains a serious problem in clinical practice. 19 Several studies have aimed to classify reflux-related cough.4,10,17 However, no technical examination is available to unequivocally establish the cause-effect relationship between extraesophageal symptoms and LPR. However, currently existing diagnostic tests as well as the PPI response frequently fail to demonstrate whether reflux episodes are responsible for LPR. BI values have been considered to be a marker of transepithelial electrical resistance (TEER) which indicates esophageal epithelial integrity. It has been shown that distal BI values are correlated with esophageal acid exposure in GERD patients, and treatment with PPIs significantly increases their BI levels, suggesting that the baseline values are affected by acid exposure.14,15,20
Currently, the most reliable method for the diagnosis of reflux-related cough is the 24-hour intraesophageal MII-pH test,9,20 but the total number of reflux events and/or the exposure of the esophageal mucosa to esophageal contents required to help classify patients as NERD remains to be determined, 21 because there are controversial results.9,22 Although weak acid reflux seems to increase in LPR, there is no consensus about the predictive value of MII-pH findings in LPR. Unlike these studies, we did not find any significant difference between patients with LPR and those with typical symptoms according to their number of reflux episodes.
Current tests for determining the presence of reflux are inadequate for measuring the long-term consequences on the esophageal mucosa. Thus, new techniques for the measurement of the health of the esophageal mucosa are required. It has been suggested that the impaired integrity of the esophageal epithelium may be a key finding in the pathogenesis of heartburn in patients with NERD. 23 As a result of gastric content contact time and frequency of contact between noxious gastric content and esophageal epithelium, permeability to luminal markers increases, and the TEER decreases with the development of dilated intercellular spaces.24–26 BI analysis has been shown to be a useful tool for evaluating esophageal mucosal integrity 13 and is suitable to reflect the mucosal injury of long-term esophageal exposure to injurious reflux contents.
Previous studies have shown that BI values are strongly correlated with acid exposure time.14,15,20,27 Consistently, we found significantly lower BI values in patients with positive AET values. Differently, we found significantly lower BI values at all segments of the esophagus in patients with LPR symptoms but only at distal segments of the esophagus in patients without LPR (up to 7 cm above the LES, data not shown). This result could be caused by a total exposure that is only distributed in the distal esophagus in patients with typical symptoms but is instead distributed along the whole esophagus in patients with LPR. Thus, this difference between groups adds more evidence to the hypothesis that BI clearly demonstrates the esophageal segments affected by acid exposure and may reflect the distribution of gastric acid content reflux in the esophagus.
Previous studies with esophageal intraluminal impedance measurements have shown that GERD patients have lower intraluminal BI values than control patients.14,28 These results suggested that lower esophageal BI represents a sensitive marker of NERD, which could possibly be related to ultra-structural changes of the esophageal epithelium.13,29,30
The BI of the proximal esophagus in patients with LPR symptoms has not been well described. Several studies have shown lower BI levels in the distal esophagus but similar levels in the proximal esophagus in patients with GERD when compared with healthy controls.13–15,29 Saritas Yuksel et al. found that impedance values were similar at 10 cm above the LES for both GERD patients and healthy controls. 28 Consistently, the distal, but not the proximal BI values, were found to be lower in patients with reflux in a recent study. 31 These results could be interpreted as evidence that chronic exposure of the esophageal mucosa is less in the proximal esophagus in typical GERD patients. In our study, we found that the proximal BI in patients with LPR symptoms was lower than in patients with typical symptoms without differences in their AET values. This could be because patients with LPR may have more chronic exposure to gastric contents in the proximal esophagus regardless of the reflux episodes of the MII-pH findings. To our knowledge, this is the first study to determine proximal esophagus BI changes in patients with LPR.
As described before, BI values in the distal esophagus were significantly lower in GERD patients compared to healthy controls. In contrast, we found similar distal BI values in patients with and without LPR. This result suggested that the patients with LPR would have significantly lower BI values than healthy controls. Comparison with healthy controls is needed to confirm this result.
Impedance values have been shown to reflect the resistance of the surrounding mucosa during periods without swallowing or reflux. 12 Savarino et al. showed a gradual increase in the BI values from the most distal to the proximal impedance esophageal segments, which may reflect the distribution of acid exposure in the esophagus. 32 Therefore, when we analyzed this increase according to symptoms with the proximal-to-distal ratio, we found a significantly lower ratio in patients with LPR symptoms compared to patients without LPR symptoms. These results may indicate that the difference in the BI value between the proximal and distal esophagus may reflect a reduction in the resistance of the proximal esophagus, which may be due to an exposure to gastric content. Thus, we think that this ratio may be useful for assessing the long-term exposure of the proximal esophagus.
Due to the inability of existing diagnostic tests to identify LPR symptoms, empirical therapy with PPIs might be a diagnostic test and a therapeutic option for LPR.5,19,33–35 However, the response to acid suppressive therapy is unpredictable in these patients. 36 Since, in patients with LPR symptoms, response to PPI therapy facilitates the diagnosis of LPR, we aimed to analyze the BI in patients with PPI-responsive and non-responsive cases in both groups. We also found similar results in the PPI-responsive NERD patients with the pathologic 24 h MII-pH study. These results indicate that patients with LPR symptoms have lower proximal esophagus BI values and proximal-to-distal ratios. In our opinion, these results support the idea that this metric (the proximal-to-distal BI ratio) can be a new diagnostic tool in patients with LPR if confirmed in further prospective studies.
Our study has some limitations. First of all, it was a retrospective design. While two separate ENT specialists evaluated the patients independently, further studies should be performed according to a prospective design and should reach a consensus between ENTs for the RFS. Secondly, we could not perform RFS analysis in GERD patients without LPR symptoms. Thirdly, there was a higher ratio of smoker patients in the LPR group. However, we found similar results after excluding smoker patients (data not shown). Fourthly, we could not compare the results with healthy controls. Finally, we could reach only about half of the patients to determine their PPI response.
In conclusion, we have shown that patients with LPR symptoms have lower BI values in their proximal esophagus, which may reflect longer exposure times to gastric content in the proximal esophagus. Additionally, the gradual proximal increase in BI is lower in patients with LPR, and we think that documenting this difference as a new metric (the proximal-to-distal BI ratio) may be useful for differential diagnosis. Indeed, in “borderline” cases the presence of low BI or a high proximal: distal BI ratio could help decide whether a patient had reflux-related symptoms. More studies are needed to confirm or refute the value of this metric.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial or non-for-profit sectors.
