Abstract
Background
Endoscopic radiofrequency ablation (RFA) is a rapidly evolving therapeutic modality for early flat esophageal squamous cell neoplasms (ESCNs). However, the in vivo tissue effects of RFA on the esophageal wall are uncertain.
Methods
We prospectively enrolled eight patients with flat-type early ESCNs who were treated with balloon-based RFA. We evaluated the in vivo tissue effect on the esophagus using endoscopic ultrasound (EUS) and the histology of retrieved coagulum.
Results
The mean tumor length was 6.1 cm, and six of the eight patients achieved a complete response after primary RFA. Real-time evaluation of the tissue effect showed that the mucosa and submucosal layer were more edematous and thicker after RFA than before the procedure (mean 4.89 vs. 2.04 mm, p<.001), suggesting that the thermal effect of RFA may injure the submucosa. Histological evaluation of retrieved coagulum showed a severe cauterization (burning) effect with extensive cell necrosis; however, four cases had some residual viable neoplastic cells. Even though there were viable cells in the sloughed coagulum, half of the patients still achieved complete remission after RFA.
Conclusions
Our findings suggest that the thermal effect of RFA may injure the submucosal layer and enable neoplastic epithelium to slough off without “burning.”
Keywords
Introduction
Recent advances in image-enhanced techniques such as Lugol chromoendoscopy and narrow band imaging1–3 have resulted in a marked increase in the detection of patients with early esophageal squamous cell neoplasms (ESCNs). Endoscopic radiofrequency ablation (RFA) is a rapidly evolving therapeutic modality, and recent studies have shown its efficacy and safety in eradicating dysplastic Barrett’s esophagus4–6 and flat type early ESCNs.7–11 However, postoperative stricture (14–28%) is a major concern with regard to the quality of life after balloon-based RFA, especially when treating squamous neoplasias.7–12 Previous studies have demonstrated that the maximum ablation depth (∼1000µm) of RFA in the human esophagus is around the muscularis mucosa layer;13,14 however, why the effect of ablation of such a thin layer without damaging the submucosa causes stenosis is unknown. In vivo tissue effects such as a thermal effect and effects on the submucosa or deeper layer of the esophagus have not been reported. In addition, even though the ablated tissue sloughs off during the cleaning step of the RFA procedure, the histology of the retrieved coagulum has never been investigated. Therefore, in this study, we aimed to evaluate the tissue effect of RFA on the esophageal wall in vivo using real-time endoscopic ultrasound (EUS). In addition, we analyzed the histology of retrieved coagulum to determine whether it can serve as a predictive marker for treatment response.
Methods
Patients
We prospectively recruited eight adult patients with newly diagnosed histologically proven early-stage ESCNs who received balloon-based RFA as the initial treatment at E-Da Hospital, Taiwan. All of the enrolled subjects had completely flat (type 0–IIb) ESCNs. EUS revealed no submucosal invasion or lymphadenopathy in any of the tumors, and computed tomography revealed no metastasis or lymphadenopathy. All patients provided informed consent following a full explanation of the use of RFA and alternative treatment options. The Institutional Review Board of E-Da Hospital approved this study on 5 May 2014. Every patient signed the informed consent for each procedure, and the study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a prior approval by the institution's human research committee.
Radiofrequency ablation
All of the enrolled patients received balloon-based RFA treatment as previously described.9,11 Prior to the RFA procedure, Lugol’s staining was performed to determine the location and size of the lesions. All RFA procedures were performed by a single endoscopist (Dr WL Wang) using a HALO360 System (Covidien GI Solutions, Sunnyvale, California, USA). We used a 12 J/cm2 – clean – 12 J/cm2 regimen for all of the patients. After the first pass of the ablation, the coagulum was collected during the cleaning step of the procedure and then sent for histological evaluation. To monitor post-ablation symptoms, the patients were asked to complete a diary using a 0–10-point scale to record chest pain, throat pain, odynophagia, and dysphagia. The patients received follow-up endoscopy with Lugol chromoendoscopy at 1, 3, and 6 months after the procedure and every 6 months thereafter.
Endoscopic ultrasound
To evaluate the in vivo real-time tissue effect of RFA in the treatment of ESCNs, we performed EUS before and immediately after the RFA procedure. EUS was performed as previously reported.9,11 A miniprobe ultrasound catheter (20 MHz, Olympus Corp., Tokyo, Japan) was inserted to measure the sum of the thicknesses of the esophageal mucosa and submucosa. 15 Thickness was measured where the tumor was located before treatment and at the post-treatment mucosal defect after RFA. The catheter was propagated to the distal margin of the lesions and then pulled back at 1-cm intervals to measure the thickness of the esophageal wall pre- and post-treatment. The maximum thicknesses of the esophageal mucosa and submucosal layer were recorded.
Results
Clinical and endoscopic characteristics of the patients
The clinical and endoscopic characteristics of the eight patients who received radiofrequency ablation.
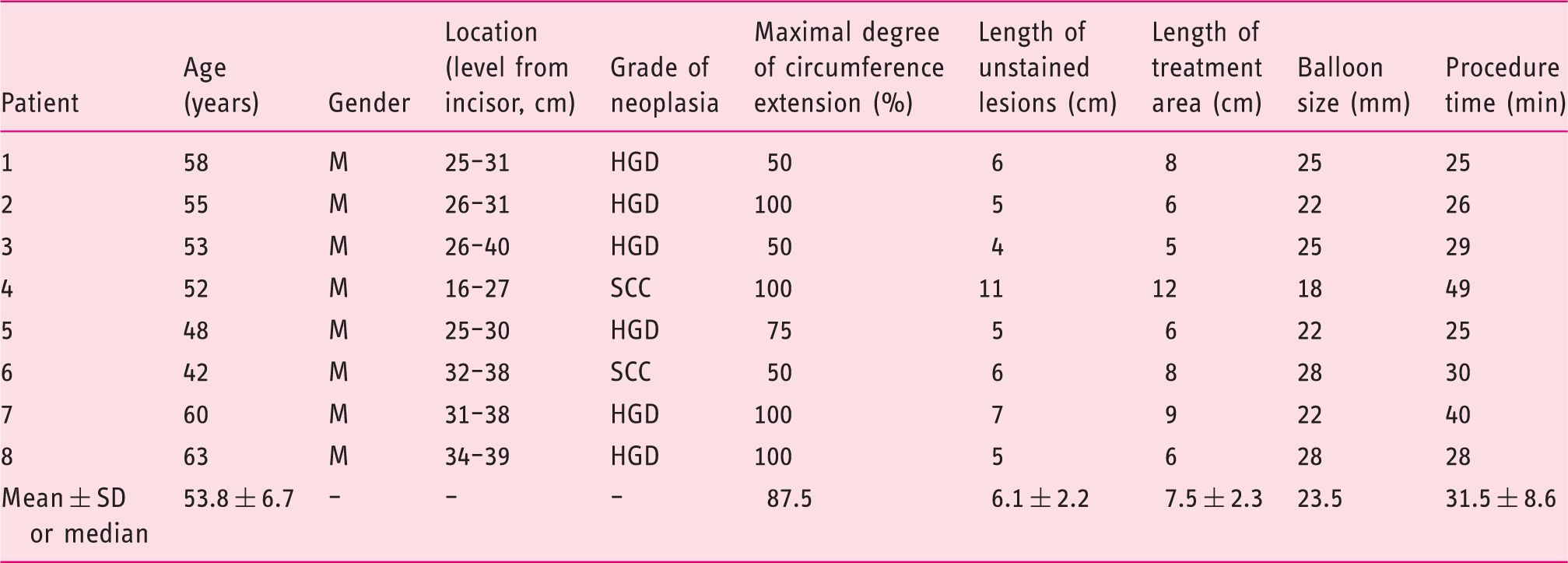
HGD: high-grade dysplasia; SCC: squamous cell carcinoma.
Evaluation of the in vivo tissue effect and outcomes after radiofrequency ablation.
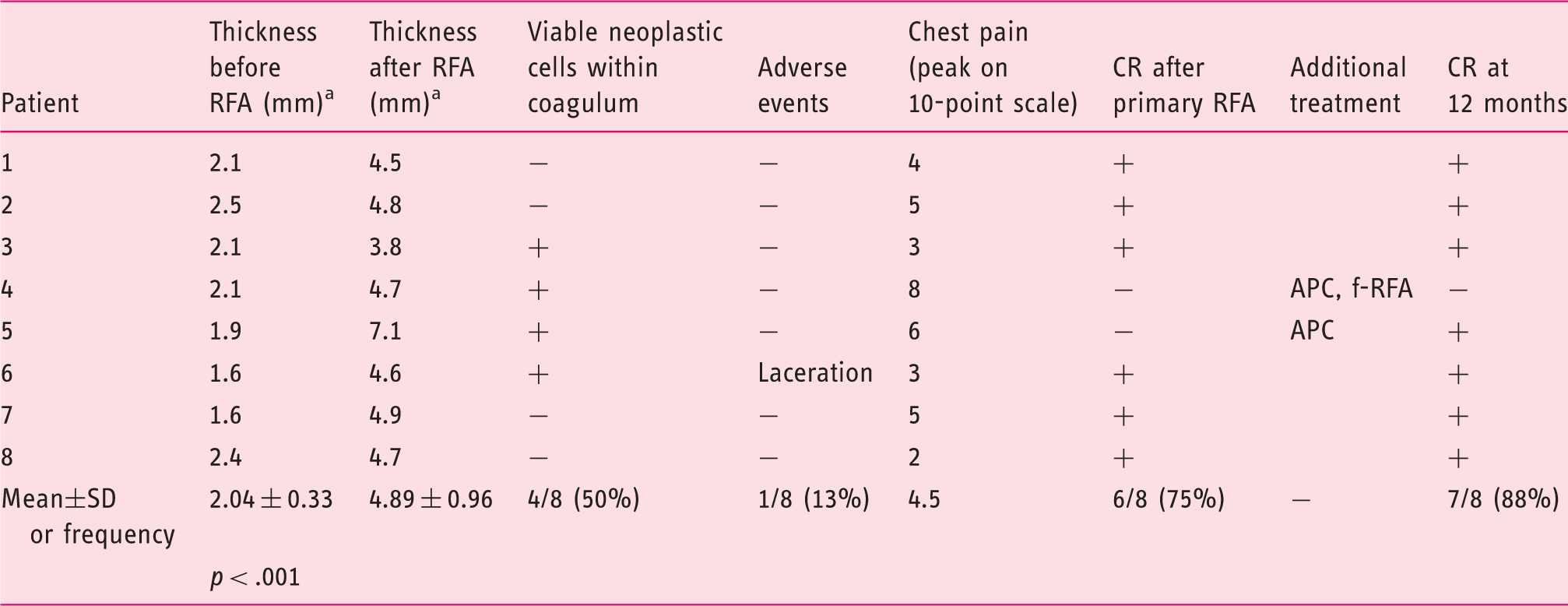
APC: argon plasma coagulation; CR: complete response; f-RFA: focal-type RFA (Halo 90).
EUS evaluation for the sum of the mucosa and submucosa thickness before and after RFA.
Real-time evaluation of the in vivo tissue effect by endoscopic ultrasound
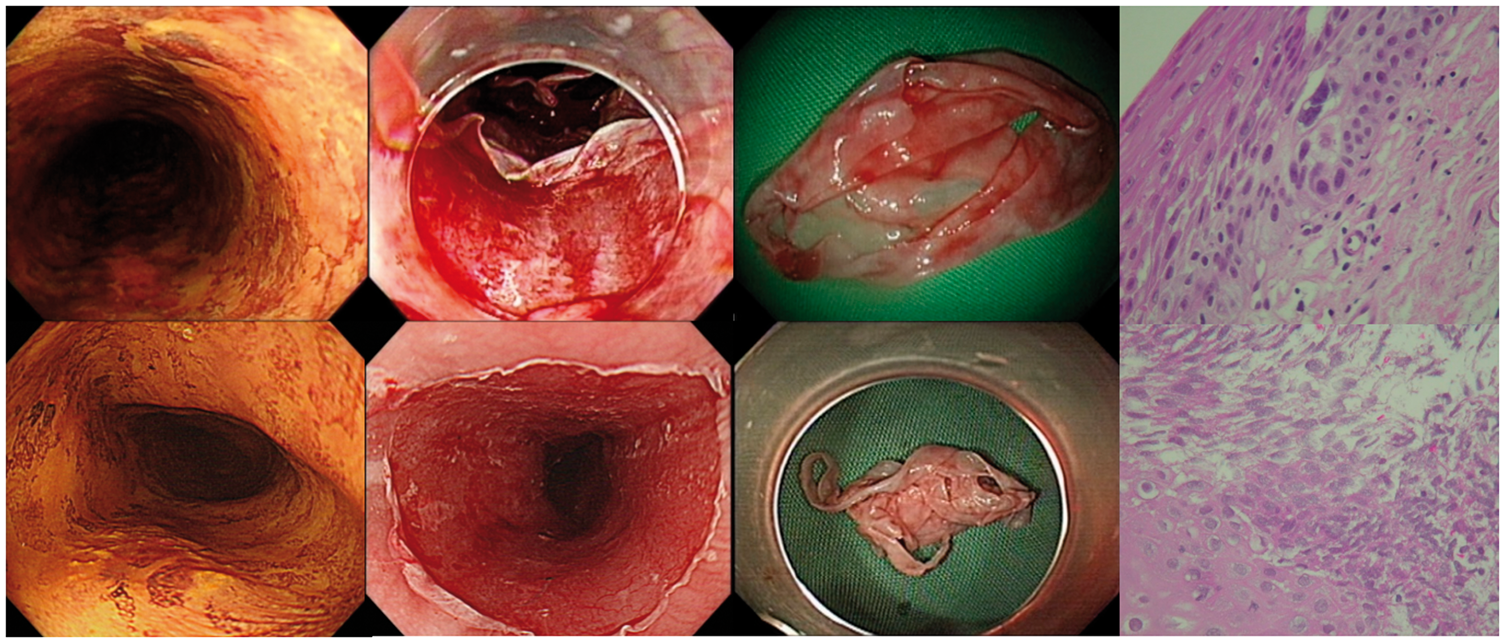
We evaluated the in vivo tissue effect using EUS immediately after RFA. The mucosal and submucosal thicknesses before and after the procedures are shown in Figure 1. After balloon-based RFA, swelling of the esophageal submucosal layer could be detected by EUS in each case. Quantitatively, the mucosa and submucosal layer were more edematous and thicker after RFA than before the procedure (mean 4.89 vs. 2.04 mm, p < .001, Table 2), suggesting that the thermal effect of RFA may injure the submucosa. We also evaluated the histology of retrieved coagulum. As shown in Figure 2, most of the ablated epithelium showed a severe cauterization effect with extensive cell necrosis. However, in four of the eight cases, some viable neoplastic cells were detected within the retrieved coagulum (Figure 2), suggesting incomplete contact between the epithelium and the catheter during ablation. Even though viable cells were seen in the coagulum, two of the four patients in whom this observation was made still achieved complete remission after the second ablation of RFA. The finding of viable cells in the sloughed epithelium suggests that the thermal effect of RFA can cause sloughing without complete epithelial cautery.
Real-time evaluation of the in vivo tissue effect by endoscopic ultrasound. Endoscopic ultrasound of two representative cases (Case 1 and Case 7) with ESCNs before (a, b) and immediately after RFA (c, d) showed that the mucosa and submucosal layer were more edematous and thicker after RFA than before the procedure. Histological evaluation of retrieved coagulum. Representative cases showed some residual viable dysplastic cells (upper panel) and extensive cauterization (lower panel) within the coagulum.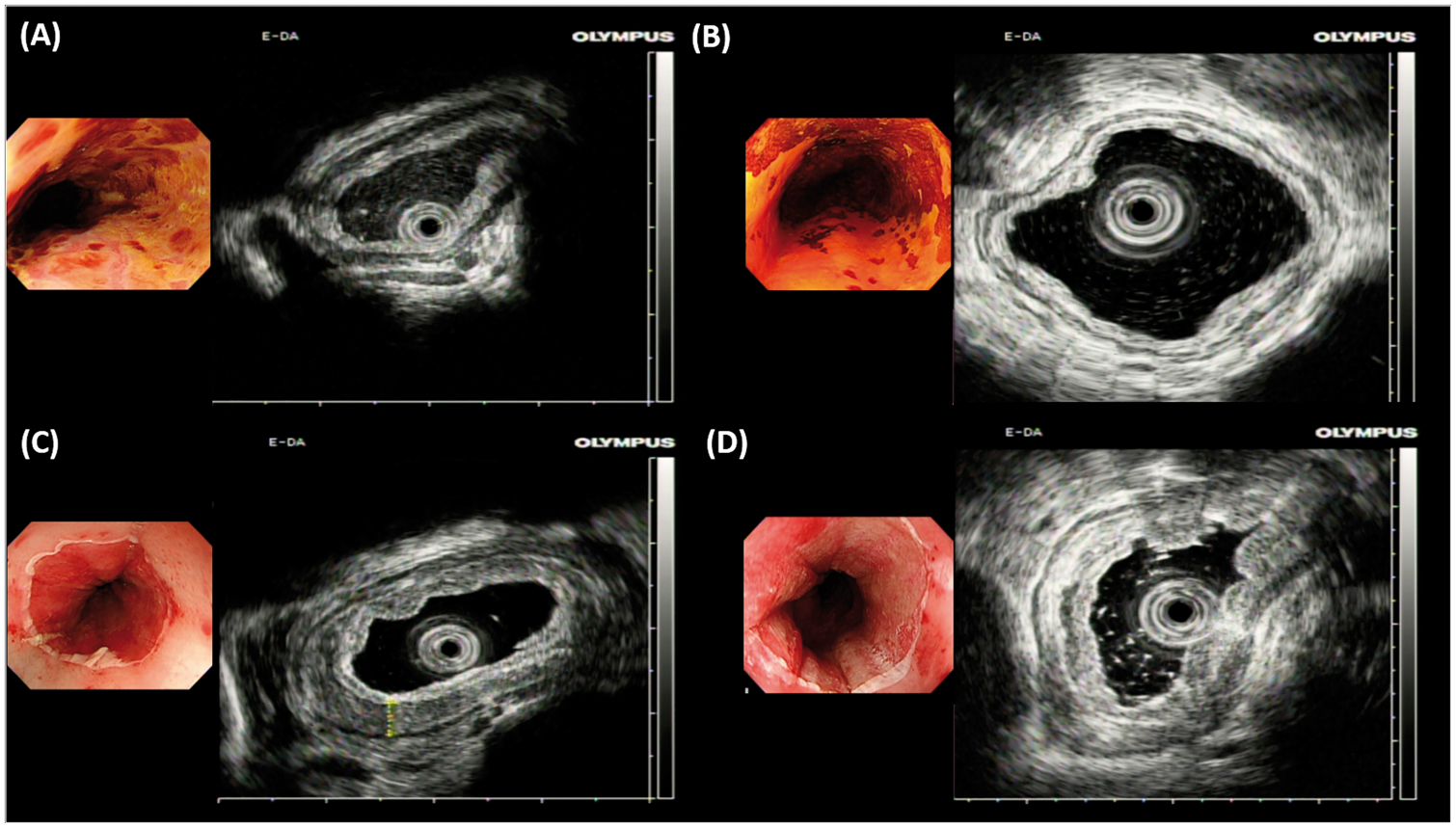
Discussion
Endoscopic RFA is a rapidly evolving therapeutic modality; however, the actual tissue effect on the esophageal wall and the potential mechanisms of post-operative stenosis are still uncertain. To the best of our knowledge, this study is the first to investigate the real-time tissue effect of RFA in vivo using EUS, which revealed obvious edematous changes over the submucosal layer, suggesting that RFA may injure the submucosal layer, which may partly account for the occurrence of stenosis.
The mechanisms of post-RFA stenosis are still uncertain. Some studies have evaluated the effect of Lugol staining on stenosis formation, but the results are controversial.10,16 The tissue effect of RFA on the esophageal wall may explain the mechanism of post-RFA stricture. Most previous studies on pigs and humans have used the histology of surgical specimens to evaluate tissue injuries related to RFA and have shown that the maximum ablation depth (∼1000 µm) is the muscularis mucosa layer, with an ablation energy of 12 J/cm2 in humans.13,14 However, this raises the question of why the effect of ablation of such a thin layer without damaging the submucosa causes stenosis. The post-ablation submucosal edema noted in our study suggests that the thermal injury may have reached the submucosal layer, or even deeper, and therefore not only the muscular mucosa as detected in surgical specimens. As none of our patients developed post-RFA stenosis, we could not analyze whether the severity of submucosal swelling can predict stenosis. Further large-scale studies are required to validate the significance of submucosal injuries caused by a thermal effect. Although EUS is not sensitive enough to show the efficacy of ablation, RFA combined with other optic modalities such as optical coherence tomography 17 or confocal laser endomicroscopy 18 may potentially be practical and informative. Furthermore, previous studies have shown that the use of RFA in treating ESCN is associated with higher risk of post-treatment stenosis than when RFA is used to treat Barrett’s esophagus. 16 Whether or not the reflux-induced injury in Barrett’s esophagus provides a protective effect may require further investigation.
In this study, four of the eight patients had viable tumor cells within the removed epithelium, which may have been due to incomplete contact between the epithelium and the balloon ablation catheter during the RFA procedure. Thus, of the two cases without complete remission after primary RFA, both had residual viable cells in the coagulum. However, the number of cases in this study is limited, and further studies are needed to clarify whether histological evaluation of retrieved coagulum can predict a response to RFA. Of note, even though there may have been incomplete contact between the epithelium and the catheter in the four patients with viable tumor cells within the removed epithelium, two of them achieved complete remission after RFA. This suggests that the thermal effect may enable the dissociation of cell-to- cell contact and subsequently enable the neoplastic epithelium to slough off. Further studies are needed to clarify whether this thermal effect is responsible for the efficacy of treatment on deeper layers of the esophageal wall.
Also, the finding of viable cells in the sloughed epithelium may account for the higher rate of incomplete eradication or even neoplastic progression when a single-ablation regimen was used in a previous study. 19 The use of a double-ablation regimen may potentially decrease the risk of residual lesions.
There are several limitations to this study. First, the number of cases is small, and thus we could not confirm the association between submucosal injuries caused by the thermal effect and stenosis. Second, the study is inevitably limited by the imperfect accuracy of EUS in evaluating submucosal disease or disease permeation.20,21 However, EUS can not only definitively demonstrate the submucosal edema but also show the changes of the thickness of mucosa and submucosa layer before and after RFA in this study. Third, we only demonstrated edematous changes of esophageal submucosa, and further studies are needed to elucidate the mechanisms involved, including a possible immunological response and pre-existing fibrous cells in the submucosal layer.
In conclusion, our findings suggest that the thermal effect of RFA may injure the submucosal layer and enable neoplastic epithelium to slough off without “burning.” Further studies are warranted to elucidate whether this thermal effect is responsible for the efficacy of treatment over deeper tumor tissue or whether it can account for the mechanism of post-RFA stenosis.
Conclusion
RFA is a rapidly evolving therapeutic modality for early flat esophageal neoplasias. Some in vitro studies have demonstrated that the maximum ablation depth of RFA on the esophageal wall is around the muscularis mucosa layer (∼1000µm); however, the in vivo tissue effects are still uncertain. The in vivo tissue effect evaluated by EUS showed that the mucosa and submucosal layer were more edematous and thicker after RFA than before the procedure. The thermal effect of RFA may injure the submucosal layer and enable neoplastic epithelium to slough off without “burning.”
Footnotes
Declaration of conflicting interests
All authors disclosed no potential conflicts (financial, professional, or personal) that are relevant to the manuscript.
Funding
This work was supported by grants (MOST-106-2314-B-650-006) from the Ministry of Science and Technology, Taiwan and E-Da Hospital (EDAHP106003).
Ethics approval
The Institutional Review Board of E-Da Hospital approved this study.
Informed consent
Written informed consent was obtained from all patients for each procedure.
