Abstract
It is well known that triple therapy for Helicobacter pylori is losing efficacy worldwide. A regimen containing proton pump inhibitor and multiple-dose capsules of bismuth, metronidazole, and tetracycline has proven efficacy. In addition, a literature review on dosage of previous regimens shows that half-dose clarithromycin-based regimens are equally effective to full-dose regimens. However, the applicability of dose reduction to bismuth-based therapy is unknown. This communication shows that a reduced-dose bismuth-based regimen fails to achieve acceptable eradication rates.
Keywords
Dear Editor
Although some reports indicate a recent decrease in the prevalence of Helicobacter pylori, 1 infection with this organism remains a leading cause of peptic ulcer disease, gastric adenocarcinomas and MALT lymphomas. The American College of Gastroenterology guidelines and the Maastricht III consensus continue to recommend triple therapy as first-line treatment, 2 despite staggering evidence indicating a decrease in efficacy worldwide. 3 Bismuth-based quadruple therapy has consequently reemerged and is currently considered the preferred regimen in areas where clarithromycin resistance is high. Recently, in a large multi-centered trial across Europe, 4 a 10-day therapy consisting of proton pump inhibitor (PPI) and a multiple-dose capsule containing bismuth, metronidazole and tetracycline (Pylera®, Aptalis Pharma, Bridgewater, NJ, USA) showed an eradication rate of 80% on intent-to-treat (ITT) with an acceptable safety profile. The relative success of this combination therapy was somewhat offset by the large number of pills (14 tablets daily × 10 days) and four-daily dosing. A number of randomized studies have validated the equal efficacy of half-dose clarithromycin-based regimens compared with their full-dose counterparts. 5 Appealing as it may be, the applicability of this concept of dose reduction to bismuth-based therapy is unknown. This study was conducted to evaluate the efficacy of a capsule containing bismuth (B), tetracycline (T), and metronidazole (M) given with amoxicillin (A) and esomeprazole (E), all in reduced-doses, in H. pylori therapy. Secondary outcomes included tolerability, adverse events (AE), and cost.
Patient characteristics

Values are mean ± SD
PPI: proton pump inhibitor.
Study results
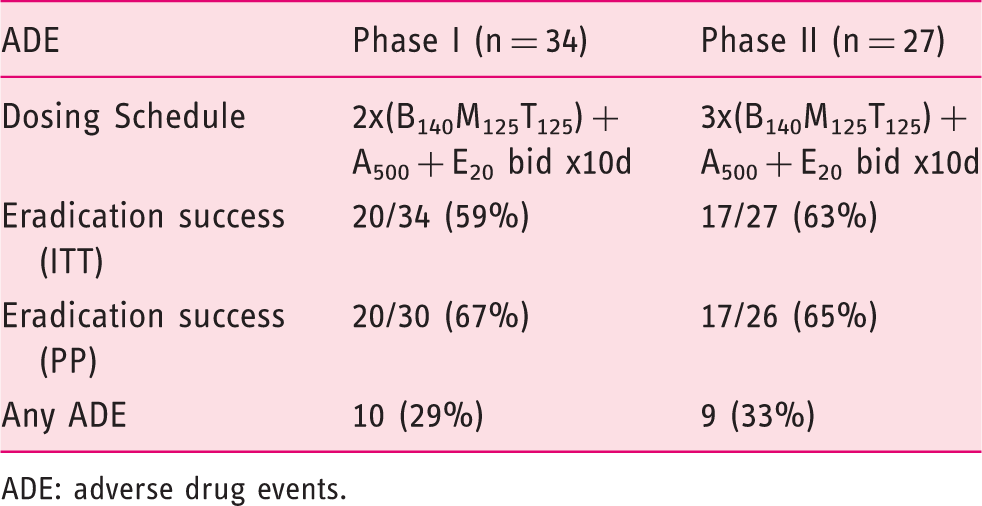
ADE: adverse drug events.
Footnotes
Conflict of interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
