Abstract
Background
Barrett’s esophagus (BE) and esophageal dysplasia (ED) are frequently missed during screening and surveillance esophagoscopy because of sampling error associated with four-quadrant random forceps biopsy (FB).
Aim
The aim of this article is to determine if wide-area transepithelial sampling with three-dimensional computer-assisted analysis (WATS) used adjunctively with FB can increase the detection of BE and ED.
Methods
In this multicenter prospective trial, patients screened for suspected BE and those with known BE undergoing surveillance were enrolled. Patients at 25 community-based practices underwent WATS adjunctively to targeted FB and random four-quadrant FB.
Results
Of 4203 patients, 594 were diagnosed with BE by FB alone, and 493 additional cases were detected by adding WATS, increasing the overall detection of BE by 83% (493/594, 95% CI 74%–93%). Low-grade dysplasia (LGD) was diagnosed in 26 patients by FB alone, and 23 additional cases were detected by adding WATS, increasing the detection of LGD by 88.5% (23/26, 95% CI 48%–160%).
Conclusions
Adjunctive use of WATS to FB significantly improves the detection of both BE and ED. Sampling error, an inherent limitation associated with screening and surveillance, can be improved with WATS allowing better informed decisions to be made about the management and subsequent treatment of these patients.
Background
In Barrett’s esophagus (BE), esophageal squamous mucosa is replaced by metaplastic columnar mucosa predisposed to developing esophageal adenocarcinoma (EAC). 1 BE is the only established precursor to EAC, a cancer with an extremely poor prognosis that resulted in an estimated 15,450 deaths in the United States (US) in 2014. 2 Optimal diagnosis and management of BE is increasingly important as the frequency of EAC in the US is rising at an alarming rate, more than seven-fold over the past several decades, and the incidence of this cancer is accelerating faster than any other solid cancer worldwide. 3 It has been estimated that 5.6% of adult US residents may be at risk for EAC because they have BE. 4
One strategy to decrease the recent rise in esophageal cancer would be an attempt at earlier diagnosis of BE and related esophageal dysplasia (ED). Despite the inability to accurately predict which patients have BE prior to endoscopy, BE is known to develop more frequently in men than women, Caucasians than African Americans, those with chronic heartburn, a hiatal hernia or erosive esophagitis, obese individuals that have a predominantly intra-abdominal fat distribution, tobacco users, those exposed to asbestos, individuals with sedentary occupations, gastroesophageal reflux disease (GERD) patients with frequent symptoms that started under age 30, and those with a family history of GERD.5–8 Higher intake of dark green vegetables, the use of nonsteroidal anti-inflammatory drugs, and gastric infection with Helicobacter pylori are associated with a decreased risk of BE. 9
Endoscopic screening and surveillance guidelines for BE and related ED rely on random four-quadrant FB obtained at 1 cm to 2 cm intervals. Although there is some evidence that patients with BE and EAC diagnosed by surveillance endoscopy have earlier-stage cancers and a better prognosis than those in whom cancers were discovered as a result of symptoms such as dysphagia and weight loss,10,11 the four-quadrant FB protocol has considerable limitations. Intestinal metaplasia and ED are often focally distributed, inconspicuous, and macroscopically indistinguishable from metaplastic tissue. Therefore, FB not infrequently fail to sample the disease being investigated, resulting in false-negative results. 12 Furthermore, even the distribution of goblet cells is patchy within the columnar-lined distal esophagus, and thus, the yield of intestinal metaplasia on biopsies obtained from the columnar-lined esophagus depends on the length of columnar mucosa as well as the number of biopsies obtained. 13 Less than half of US gastroenterologists adhere to the current biopsy recommendations for BE, and failure of protocol adherence is associated with decreased rates of disease detection. 14
A wide array of adjunctive techniques to FB has been developed to improve detection of BE and enhance the detection of ED areas. These include magnification endoscopy, chromoendoscopy, optical coherence tomography, confocal endomicroscopy, narrow band imaging, autofluorescence endoscopy, and wide-area transepithelial sampling with computer-assisted three-dimensional (3D) analysis (WATS). Our paper focuses on one of these adjunctive techniques, the WATS biopsy system, utilized in a screening and surveillance population. WATS utilizes an abrasive brush that samples the full thickness of the epithelium and collects tissue from a wide area of the esophagus. The specimen is then analyzed by a computer image processing system that identifies potentially abnormal cells based on cellular morphology and molecular diagnostics.
WATS is not intended to be a substitute for targeted FB of visible mucosal abnormality, as a forceps can better sample these specific areas with high localization specificity. WATS is instead used to sample entirely different segments of the esophagus that would otherwise remain untested by FB.
In one study involving 1266 patients being screened for BE, the addition of WATS to FB increased the detection of BE by 39.8% (95% CI 32%–48%). 15 In a second multicenter prospective study of 151 patients in a high-risk dysplasia surveillance program at tertiary care centers, the addition of WATS to FB increased the detection of ED by 42% (95% CI 20.7%–72.7%). 16 Together, these studies suggest that WATS may be a valuable adjunct to standard endoscopic screening and surveillance protocols for the identification of BE and ED.
In our study, we sought to investigate the utility of WATS in a community-based setting replicating its use in clinical practice. Our specific goal was to determine if the addition of WATS to four-quadrant FB increases the yield of detecting BE and ED.
Methods
A prospective multicenter, community-based trial involving endoscopists at 25 sites utilizing WATS was conducted during the study period between 2012 and 2014. All patients over age 18 who were scheduled for upper gastrointestinal endoscopy to evaluate symptoms of gastroesophageal reflux and suspected BE were enrolled. Investigators were instructed to sample suspected BE from salmon-colored mucosa in the tubular esophagus. Patients with a highly suspicious lesion requiring resection were excluded from the study. Schulman Associates Institutional Review Board approval was granted October 11, 2012, and approved consent was obtained from all patients prior to study initiation. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a prior approval by the institutions’ human research committees. All the components needed to perform WATS testing were supplied to investigators in kits. A test requisition form that included demographic data including the patient’s age and gender, along with endoscopic findings, was completed.
Investigators were trained in person or by videoconference on how to perform WATS testing and were also provided written instructions. Briefly, the brush, in its enclosed sheath, was passed through the working channel of the endoscope (Figure 1). Sampling was performed by maintaining pressure against the mucosa, and rotating the brush circumferentially along the epithelial surface. Pinkish-red tissue or pinpoint bleeding at the brush-biopsy site was evidence of proper technique. Up to 6 cm of the columnar-lined mucosa was sampled with a single brush.
The cellular material collected on the brush was then transferred to a bar-coded glass slide and rapidly flooded with fixative to avoid air-drying. The bristle portion of the brush was clipped off into the enclosed vial for cell block preparation. A second WATS sample was obtained using an additional brush, and the bristle portion of that brush was placed into the same vial containing the bristle portion of the first brush.
Immediately after obtaining WATS specimens, investigators performed four-quadrant FB of the esophagus at 1 cm to 2 cm intervals and tested any endoscopically visible mucosal abnormality.
All WATS and FB samples were analyzed at CDx Diagnostics (Suffern, NY). One group of pathologists was assigned to analyze FB and a different group analyzed the WATS samples, both blinded to the results of the other technique. To control for potential interobserver variability, an independent pathologist who did not participate in the study and was blinded to all clinical and histologic data confirmed all cases reported as BE or ED. Only the final data from the independent pathologist were used for the analysis. In our study, BE was defined as specialized columnar epithelium, characterized by the presence of goblet cells obtained by WATS or FB from salmon-colored mucosa in the tubular esophagus.
Each WATS sample is composed of a 100 micron-thick disaggregated tissue specimen containing intact microbiopsy tissue fragments, individual cells, and cell clusters, which cannot be effectively visualized by a standard manual microscope with a 3 micron depth of field. Analysis of WATS specimens is therefore aided by a high-speed, computer-assisted, 3D analysis system. Neural networks specifically optimized for esophageal mucosa then examine this synthesized 3D image and prepare a high-resolution video montage of the 80 most suspicious images from each slide for review by a pathologist. The computer does not alter the specimen in any way or provide a diagnosis. Rather, once the computer-assisted microscope identifies and locates goblet cells and dysplasia, these findings are then confirmed by pathologists using a standard microscope utilizing cytopathological criteria for the diagnosis of both BE and ED.
The metric used for evaluating the clinical utility of WATS is “added yield” defined as the (amount of abnormality detected by WATS + FB) – (amount of abnormality detected by FB alone)/(amount of abnormality detected by FB alone). The results of FB cross-classified by WATS were also arrayed as multinomial outcomes of the total number of cases. The proportionate increase in detection yield provided by adding WATS to FB was estimated as the above defined ratio of the number of cases that were negative on FB but positive on WATS, to the total number of cases that were positive on FB. Confidence intervals (CIs) for this added yield ratio were estimated using Fieller’s theorem. Results were calculated with Mathematica software, version 9.
The power analysis used to define the study size was based on the z-test difference between two independent proportions as this was the more conservative statistical approach. In this community-based study composed almost entirely of GERD patients who were being screened and who did not have a history of BE or dysplasia, we assumed FB detection of BE in 15% of these patients, and FB detection of low-grade dysplasia (LGD) in 0.6% of them. The power analysis sought the sample sizes required to show that the added yield of BE provided by adding WATS to FB is at least 70% (0.15 × 0.7 = 0.105) and that the added yield provided by adding WATS to FB for the detection of dysplasia is at least 40% (0.006 × 0.4 = 0.0024). With these assumptions, the sample size required to establish a minimum added yield of 40% for BE detection is 940 cases (alpha 0.05, power 0.9) and sample size required to establish a minimum added yield of 40% for the detection of dysplasia is 3990 cases (alpha 0.05, power 0.8, using Gpower v3.1.4). An independent statistician analyzed all of the data.
Results
Demographic characteristics of enrolled patients (n = 4203).

Average suspected BE length: 1.3 cm (range 0.1–6 cm; standard deviation 1.03; median: 1.1 cm; 25th percentile: 1 cm; 75th percentile: 1.2 cm)
GERD: gastroesophageal reflux disease; BE: Barrett’s esophagus; CA: cornu ammonis.
Increased detection of Barrett’s esophagus (BE) and low-grade dysplasia (LGD) (n = 4203).
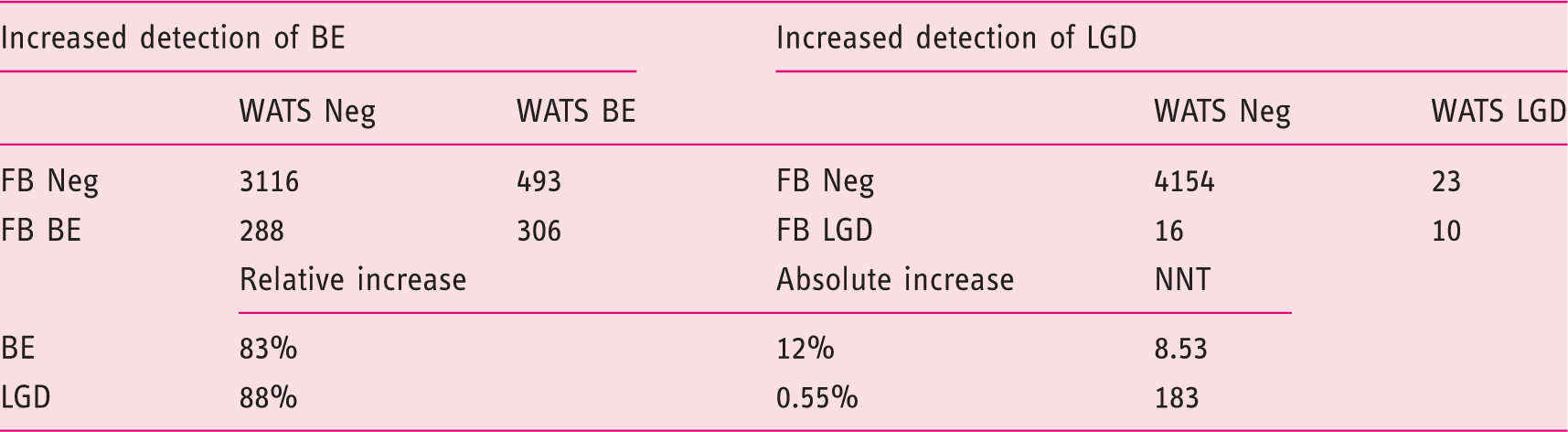
WATS: wide-area transepithelial sampling with three-dimensional computer-assisted analysis; FB: forceps biopsy; Neg: negative; NNT: number of patients needed to test.
While ED was a relatively uncommon finding in this community-based, predominantly screening population, detection rates of LGD by the FB and WATS are also shown in Table 2. LGD was diagnosed in 26 patients by FB and in 33 patients by WATS. Of the 33 patients diagnosed with LGD by WATS (only four had a history of BE and the average suspected length of BE in these patients was 1.4 cm), 23 had negative FB results. Thus, the addition of WATS to FB increased the detection of LGD by 88.5% (23/26; 95% CI 48%–160%). The added detection of LGD in 0.5% of all screening patients tested with WATS (23/4203) results in an NNT to detect one additional case of ED of 182.7. High-grade dysplasia (HGD)/EAC was not detected by FB in this primarily screening population, although WATS detected a single case of EAC.
In the 211 patients with a known history of BE, LGD was diagnosed in three patients by FB alone, with an additional six cases detected as a result of adding WATS. The addition of WATS to FB in this group of patients increased the detection of LGD by 200%.
Discussion
Current endoscopy protocols for detecting BE and ED are hampered by biopsy sampling error using random four-quadrant FB, as only 4%–6% of the BE area is sampled. 17 Sampling error may explain why in a recent Danish study, more than two-thirds of all esophageal carcinomas in newly diagnosed BE patients were identified during the first year of follow-up. 18 Sampling error may also account for the fact that in a study of 68 patients who underwent esophagectomy for BE with high grade esophageal dysplasia, 12 EACs were identified in the resection specimen, eight of which were invasive cancers. 19 Sampling error explains, in part, the results of a study that showed that the frequency of surveillance endoscopy over a three-year period in patients known to have BE who died of EAC was identical to control patients with BE; surveillance was not associated with a decreased risk of death from EAC. 20
In our study, the addition of WATS to FB increased the yield of BE and ED by 83% and 88%, respectively, demonstrating that sampling error can be improved dramatically with use of this adjunctive technique. Although our results with WATS showed a significantly increased yield over what has been reported in studies with much smaller sample sizes,15,16 our results are in complete agreement with another study that included a much larger number of patients. 21 As our study was conducted predominantly on GERD patients who did not have a history of BE, the average BE length diagnosed in our study was less than 1.5 cm, which may result in lower endoscopic diagnostic accuracy. Furthermore, only a small number of patients were diagnosed with LGD in this population and most had short segments of BE; therefore these patients are likely to be at lower risk of progression to EAC than patients with more extensive BE and multifocal LGD.
Another obstacle hindering screening and surveillance is the difficulty in microscopically distinguishing ED from nondysplastic BE. The diagnosis and grading of ED are based on the degree of architectural and cytologic alterations resulting in significant interobserver variability among pathologists, with kappa values reported between 0.36 and 0.78.22,23 The variability is significant even among expert pathologists, especially in the presence of esophageal inflammation. In a recently published study, 149 WATS slides with varying degrees of BE and ED were analyzed by four pathologists trained in the analysis of WATS samples. 24 The overall mean kappa value was calculated at 0.86 (95% CI 0.75%–0.97%), a significant improvement over current FB-based histopathologic assessment.
Our study has several strengths, including a large cohort of patients undergoing screening and surveillance by community-based gastroenterologists nationwide. The demographic information from the patients in our study are representative of the general at-risk population for BE and ED and therefore replicates the utilization of WATS in clinical practice.
Our study also has several limitations. The order of WATS and FB was not randomized. All the FB samples were obtained after WATS sampling. However, the order of performing FB and WATS has been demonstrated in one study to not have any impact on the final results. 21 All specimens in our study were analyzed by two different sets of pathologists (WATS and FB) who were blinded from each other’s findings. Although this analysis was conducted at a single central laboratory, the data are comparable to studies in which WATS was analyzed at one laboratory and FB at another.15,16,21
Although all investigators were instructed to sample only the tubular esophagus, this requirement was not independently monitored. Furthermore, it may be difficult for gastroenterologists to definitively identify where a biopsy specimen may have come from in relation to the gastroesophageal junction (GEJ) as there are no universally accepted validated landmarks that demarcate the distal extent of the esophagus. Studies in BE patients have used the proximal extent of the gastric folds as the landmark for the GEJ despite the fact that it is a dynamic structure whose position changes with respiration, gut motor activity, and the degree of distention of the esophagus and stomach. Thus, given these limitations, it is likely that some patients in our study diagnosed with BE, especially those with ultra-short BE, had in fact intestinal metaplasia of the cardia, which is thought to carry significantly less risk of cancer development than traditional BE. These limitations may be overcome by ensuring gastroenterologists follow published guidelines for sampling suspected BE, and accurately assessing the extent of BE on endoscopy. Measuring both the circumferential extent (C) and the maximum extent (M) (Prague C and M criteria) increases the accuracy in determining the length of intestinal metaplasia in the columnar-lined esophagus and helps identify those at greater risk for cancer. 25
Our study has an important strength. For most diagnostic advances that enhance sensitivity, specificity is decreased. This is not the case with WATS. Although 3D computer imaging is required to assist the pathologist in locating abnormality on the WATS specimen, all diagnoses are made using only standard pathologic criteria. The increased yield we detected in this study thus represents a true increase in the number of patients with BE and ED, and not false-positive cases, as WATS cellular images are interpreted based only on standard pathologic criteria that are considered pathognomonic of disease. Representative images from WATS-positive/FB-negative cases are illustrated in Figures 2 and 3.
(a) and (b) Unlike traditional cytology instruments, which obtain only superficial exfoliated cells, WATS uses an abrasive brush that samples the full thickness epithelium and was designed to consistently sample deeper layers of the more firmly attached glandular epithelium found in Barrett’s esophagus. WATS: wide-area transepithelial sampling with three-dimensional computer-assisted analysis. WATS image of esophageal adenocarcinoma (EAC). Two clusters of EAC detected by WATS (FB-reported nondysplastic Barrett’s esophagus). WATS image of nondysplastic Barrett’s esophagus reported as negative for intestinal metaplasia on FB: Computer-synthesized three-dimensional image of the en face view of the gland.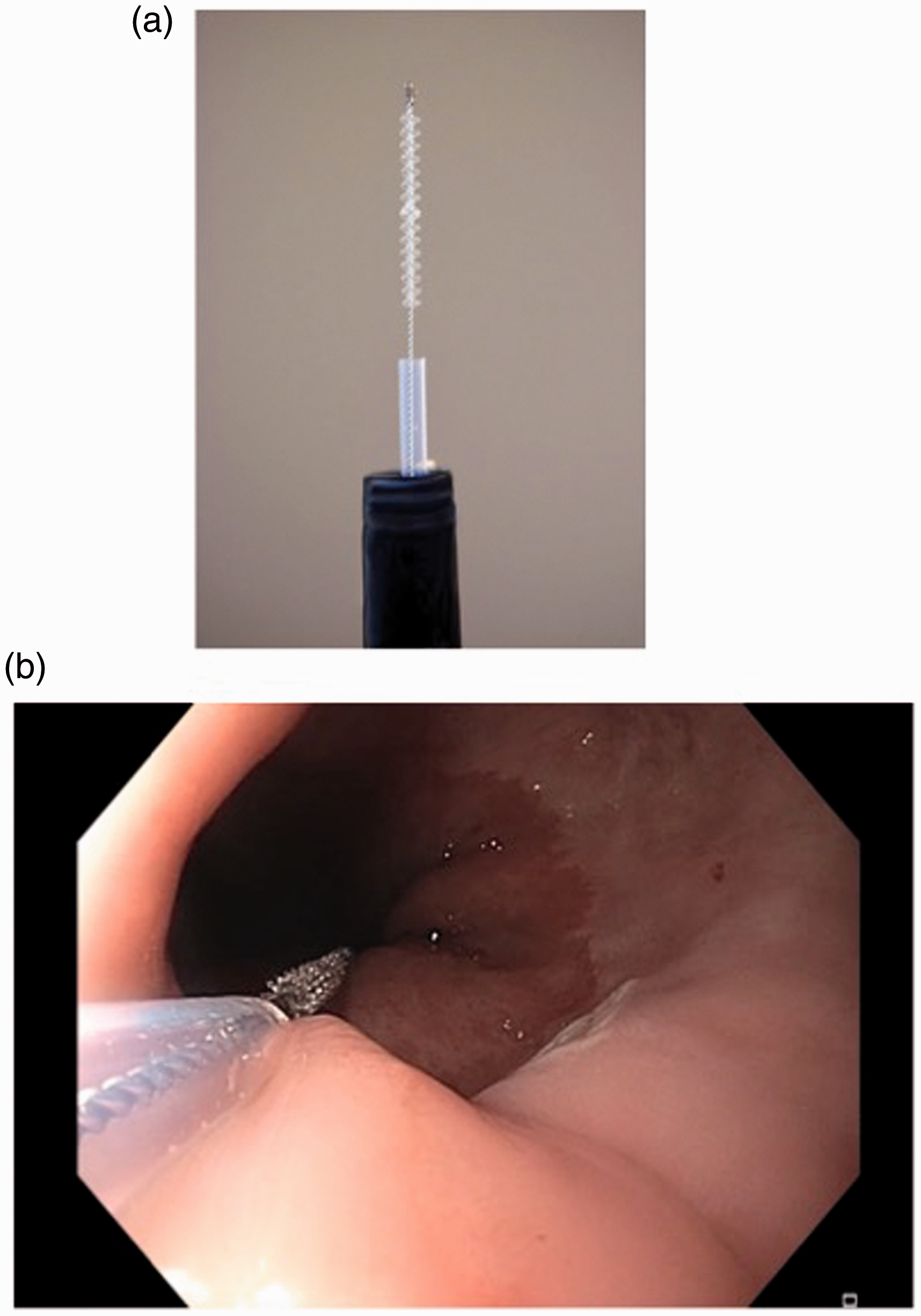
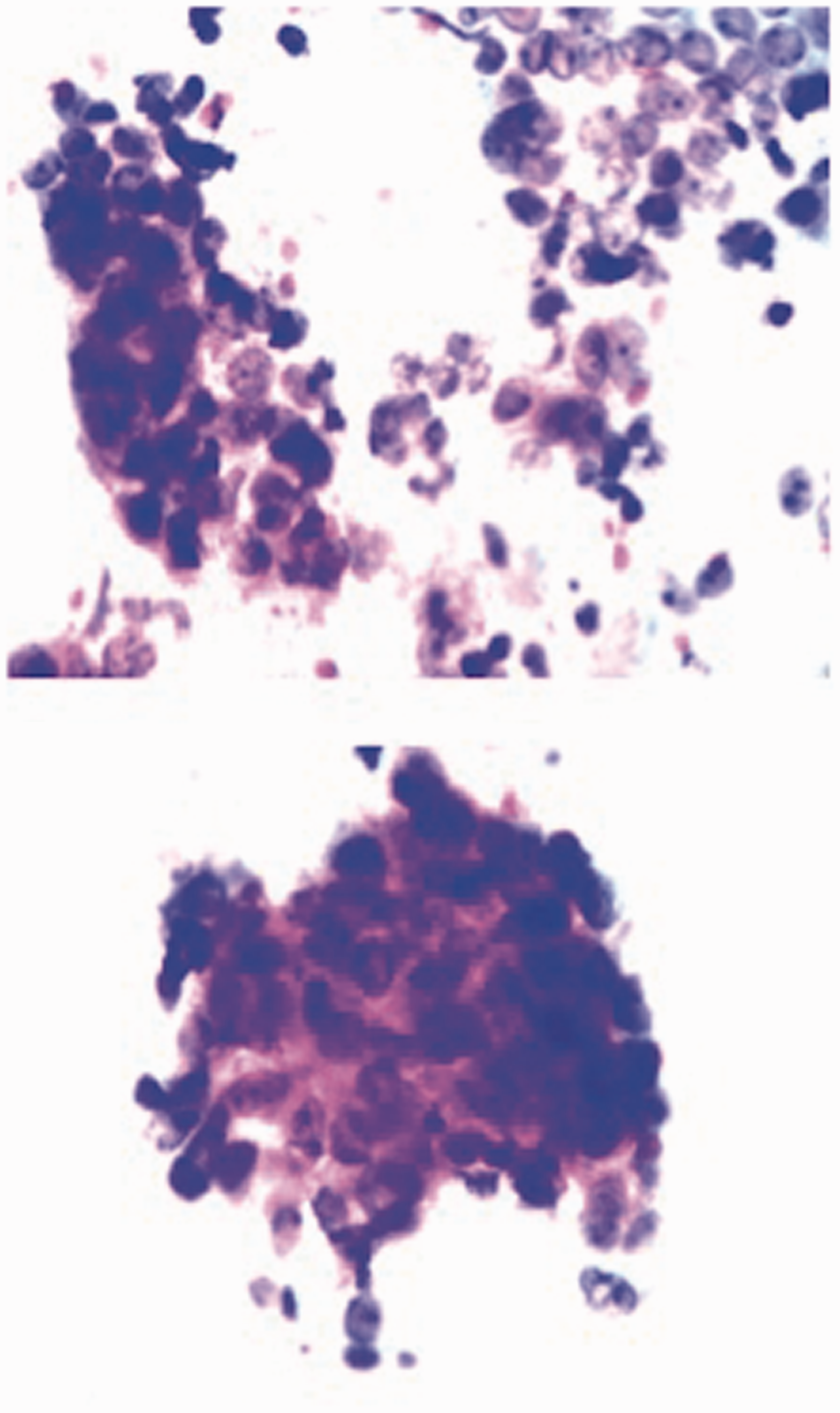
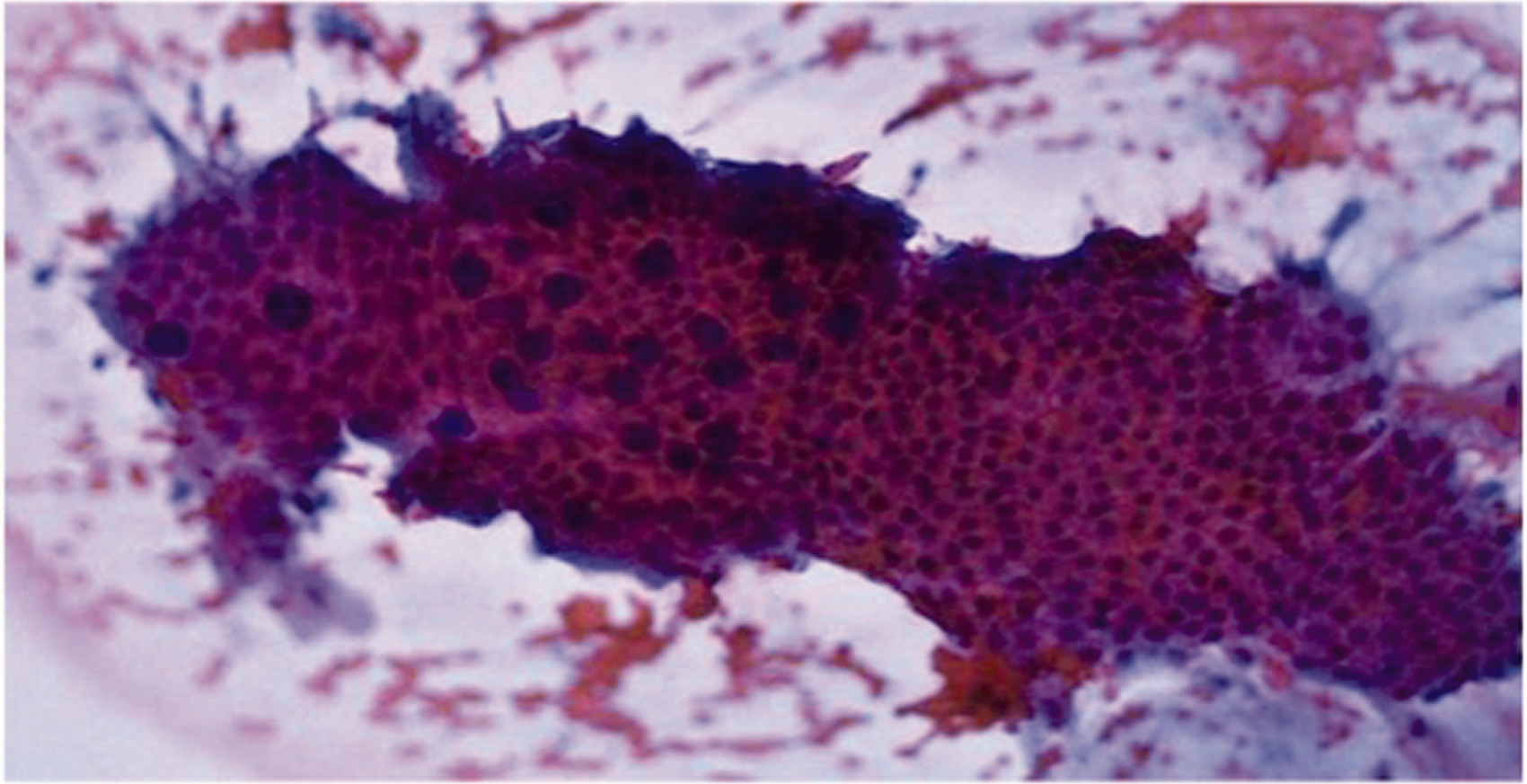
In our study, there were 288 cases of BE and 16 cases of ED identified by FB that were undetected by WATS. These discrepant results are not unexpected as FB was used both to target any visible mucosal abnormality and to obtain random four-quadrant biopsy samples. By contrast, WATS was used only to test large segments of the esophagus that would have remained untested by both targeted and random FB. Our study was not designed to address the question of whether WATS is more or less effective than FB in identifying BE and ED. Additional studies comparing WATS in conjunction with FB vs FB alone would be required. In our study, we clearly demonstrate that the increased adjunctive use of WATS to FB increases the detection of BE and ED, in agreement with previously published studies.
The current limitations of four-quadrant FB in detecting BE and ED demand improved and more accurate techniques to identify patients who will benefit from surveillance or therapy. In this study, we confirm the results from previous clinical trials that showed that WATS, when used adjunctively with FB, significantly increases the detection rate of BE and ED. We believe that the increased detection of disease will allow better informed decisions to be made about the management and subsequent treatment of these patients.
Footnotes
Acknowledgments
This study is registered under clinical trial number NCT03008980.
The US Collaborative WATS3D Study Group includes the following individuals: PA Erachshaw, Queens Surgery Center, Glendale, NY; HN Guss and GD Beri, Advanced Endoscopy and Surgical Center, Eatontown, NJ; M Suchov, Encino Plaza Surgical Center, Encino, CA; RM Gupta, Procare Health and Wellness, Corona, NY; F Ibarra, Gastroenterology Diagnostic Center, Monterey Park, CA; BG Chusid, MI Galica-Vladutiu, LJ Ottaviano and K Sayad, Gramercy Park Digestive Disease Center, New York, NY; KR Feuer, Citrus Ambulatory Surgery Center, Orlando, FL; A Shapsis, Atlantic Gastro, Brooklyn, NY; MA Silverman and DJ Sosnowik, Austin Medical Associates, Forest Hills, NY; M Amadpour, MM Patel, MR Trivedi, RB Moghimi and A Malamud, Simi Surgery Center, Simi Valley, CA; S Tabib, La Peer Surgery Center, Beverly Hills, CA; OS Maat, First Street Hospital, Bellaire, TX; SH Kim and SA Hussain, New York Hospital Queens: Endoscopy Unit, Flushing, NY; SA Desai, Endeavor Surgery Center, Northridge, CA; CA Cox, Garden Grove Surgical Center, Garden Grove, CA; PV Lang, Memorial Hermann Hospital, Houston, TX; I Chakrabarty, Hemet Endoscopy Center, Hemet, CA; AH Schnall, Queens Endoscopy ASC, Fresh Meadows, NY; GJ Ardigo, JT Dinh and LM Hurwitz, Endoscopy Center of Inland Empire, Murrieta, CA; DJ Vargas and RD Lamport, Surgery Center of Lakeland Hills Boulevard, Lakeland, FL; MB Anwer, Celebration Surgery Center, Celebration, FL; JE Weissbluth, Digestive Diseases Diagnostic & Treatment Center, Brooklyn, NY; RB Harooni, Great Neck Gastro, Manhasset, NY; A Shalamov, Advanced Gastroenterology, Flushing, NY; and JS Hixon, Northeast Alabama Regional Medical Center, Anniston, AL.
Declaration of Conflicting Interests
None declared.
Funding
The work was supported by a grant from CDx Diagnostics.
Ethics approval
Schulman Associates Institutional Review Board (IRB) approval was granted October 11, 2012. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a prior approval by the institutions’ human research committees.
Informed consent
Approved consent was obtained from all patients prior to study initiation.
