Abstract
Background and aim
Direct antiviral agents (DAAs) have led to high sustained virological responses (SVR) in hepatitis C virus (HCV) patients. However, genotype 3 patients respond to treatment in a suboptimal way. This study aims to identify which of the several treatment schedules recommended for genotype 3 would constitute the best option.
Methods
Twenty-four Italian centers were involved in this real-life study of HCV genotype 3 patients treated with DAAs. To expand the number of cases, we conducted a systematic review of the literature on the outcome of genotype 3 patients treated with DAAs.
Results
A total of 233 patients with HCV genotype 3 were enrolled. Cirrhotic patients accounted for 83.7%. Overall, the SVR12 rate was achieved by 205 patients (88.0%); the SVR rates were 78.8% after sofosbuvir/ribavirin, 92.5% after sofosbuvir/daclatasvir ± ribavirin, and 100% after sofosbuvir/ledipasvir (seven patients). No difference in rate of SVR was observed in cirrhotic and non-cirrhotic patients (92.2 vs 94.4) using a combination regimen of NS5A and NS5B inhibitors.
The systematic review of the literature provided data of 3311 patients: The mean weighted SVR12 rate was 84.4% (CI: 80.4–87.8); the rates varied from 79.0% (CI: 70.9–85.3) with sofosbuvir/ribavirin, to 83.7% (CI: 66.2–93.1) with sofosbuvir/ledispavir, and to 88.2% (CI: 83.3–91.7) with sofosbuvir/daclatasvir.
Conclusions
Our results reinforce the concept that patients with HCV genotype 3 should no longer be considered difficult-to-treat individuals. The optimal therapeutic regimen for these patients appears to be the combination sofosbuvir/daclatasvir, administered for 12 weeks without the use of RBV in non-cirrhotic patients. In cirrhotics the meta-analytic approach suggests extending therapy to 24 weeks.
Key summary
In the era of direct antiviral agents (DAAs), the schedule of treatment for hepatitis C virus (HCV) genotype 3 is not optimized, with low rates of sustained virological response (SVR), especially in cirrhotic patients. In our cohort (233 genotype 3 patients) the use of a schedule containing a combination of NS5A and NS5B demonstrated high efficacy (>90%) without difference in cirrhotic and non-cirrhotic patients. The meta-analytic approach in 3311 patients confirms the higher efficacy of the combination of sofosbuvir and daclatasvir. The extension of treatment to 24 weeks independently of the use of ribavirin seems to be the best option in difficult-to treat-patients (cirrhotic and treatment-experienced patients).
Introduction
Hepatitis C virus (HCV) genotype 3 is the second most common subtype, most prevalent in the South Asian region. It accounts for about 50 million infected individuals worldwide.1,2
At the time when pegylated-interferon (IFN) in combination with ribavirin (RBV) was the only available treatment for chronic HCV infection, genotype 3 was considered an “easy to treat” genotype, and several studies showed this regimen to be effective in 68% to 90% of patients.3,4
The shift from IFN-based therapy to all-oral IFN-free therapies has been a watershed for the management of chronically HCV-infected patients. The first licensed direct antiviral agent (DAA) was sofosbuvir, a pangenotypic NS5B inhibitor: When given in combination with RBV, the regimen proved sub-optimal for HCV genotype 3, as sustained virological response (SVR) rates were unexpectedly lower than those registered in non-3 genotypes. This notion applies especially to cirrhotic patients with HCV genotype 3 infection. 5 In fact, in registration trials SVR was achieved in 47% of treatment-naïve cirrhotic patients, and in 21% of treatment-experienced patients with cirrhosis, when treatment lasted for only 12 weeks.6,7 Extending sofosbuvir/RBV treatment to 24 weeks resulted in 92% SVR rates for treatment-naïve patients, but for treatment-experienced patients SVR rates remained at an unacceptable 62%. 8 In the DAAs era, HCV-3 genotype has emerged as the new “difficult to treat” genotype.
A partial explanation for the previous different rates may be related to the fact that most of the available DAAs have been designed to fit with the catalytic/functional sites of HCV genotype 1 protease, polymerase and NS5A proteins. Consequently, many of the currently approved DAAs are HCV-genotype restricted with robust in vitro efficacy toward genotype 1, and less so toward genotype 3. 9 Moreover, the impact of baseline and on-treatment appearance of resistant antiviral variants in genotype 3 treatment failure has not yet been systematically investigated. 10 When pangenotypic NS5A inhibitors like daclatasvir or velpatasvir were employed in combination with sofosbuvir, the therapeutic outcome remarkably improved.11,12 Indeed, in registration trials the sofosbuvir/daclatasvir combination for non-cirrhotic genotype 3 patients achieved SVR rates above 95%, both in naïve and experienced patients. 11 However, this same therapeutic regimen proved less effective in cirrhotic patients, with SVR rates dropping to 57% and 63% in naïve and treatment-experienced cohorts, respectively. 11 For the latter subgroup of patients, SVR rates attained a value of 86% when this regimen was complemented by the inclusion of RBV and prolonged to 16 weeks. 13 Several real-world studies using sofosbuvir and daclatasvir, with or without RBV, reported SVR rates >80% in cirrhotic patients with genotype 3, including those with decompensation.14,15 Therefore, the current therapeutic strategies for genotype 3 patients seem to be optimal only for those without cirrhosis, while for cirrhotic patients the optimization of the schedule with current DAAs requires further investigation.
The present report focuses on HCV genotype 3 cirrhotic patients treated with second-generation DAAs and aims to identify the best treatment option in relation to the type of combination of administered drugs, duration of treatment and the need to include RBV in the treatment schedule. In addition, with the intent to ensure the robustness of our project, we enlarged the number of genotype 3-infected patients treated with the new DAAs by scrutinizing the available studies in the recent literature, and pooled the data obtained from our cohort of patients with those retrieved by our search in a meta-analysis.
Material and methods
Study design and patients population
From May 2015 to September 2016, a consortium of 23 community and academic Italian centers (the ITAL-C consortium) conducted an observational real-life study collecting consecutive patients infected by all HCV genotypes treated with any of the new DAA regimens. 16 From this large database, we extracted data on all patients infected by the HCV genotype-3. Patients with any of the following features were excluded: infection with HCV genotypes other than 3, active hepatocellular carcinoma (HCC) on imaging, human immunodeficiency virus (HIV) and/or hepatitis B virus (HBV) co-infection, liver-transplant recipients, active alcohol consumption; patients with an estimated glomerular filtration rate <30 ml/min. All patients had no history of prior alcohol abuse. Eligible patients were>18 years old with chronic HCV infection, either naïve or treatment experienced. Treatment-experienced patients may have received prior therapy with peg-IFN and RBV in combination or not with first-generation DAAs. In Italy antiviral therapy with DAAs is reimbursed by the national health system only in patients who fulfill specific criteria: patients with advanced fibrosis (F3 sec. Metavir); patient with cirrhosis; patients with previous HCC with a complete response to curative therapies; patients with significant fibrosis (≥F2) and recipients of solid organ transplantation (other than liver) or bone marrow transplantation; patients with any degree of fibrosis with severe extrahepatic manifestations (cryoglobulinemic syndrome with organ damage, B-cell lymphoproliferative syndromes). Therefore all the patients in the cohort fit one of these criteria.
The treatment regimen was chosen individually by the prescribing clinician in accordance with national and international guidelines,17–19 although in seven cases a non-recommended therapy was prescribed (namely ledipasvir and sofosbuvir) because of its use as individual compassionate therapy. Treatment duration (12/24 weeks) was dictated by the severity of liver disease, with longer treatment reserved for cirrhotic patients. All patients who received at least one dose of the new DAAs were included in the study. The study protocol was approved July 27, 2016 by the Ethics Committee of the Casa Sollievo della Sofferenza Hospital, in San Giovanni Rotondo, Italy. Each patient included in the study provided a written informed consent. All the study’s procedures were conducted according with the provisions of the Declaration of Helsinki and Good Clinical Practice Guidelines. For each enrolled participant, baseline characteristics were acquired: gender, body mass index (BMI), age, prior treatment, cirrhotic status, decompensating events (ascites, esophageal bleeding, encephalopathy, HCC), enlisting for orthotopic liver transplantation (OLT). The following laboratory values were registered: alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin, creatinine, international normalized ratio (INR), blood count and albumin, alpha-1-fetoprotein. Comorbidities were classified according to Harboun and Ankri, 20 and the possible interaction of co-administered drugs with the DAA to be prescribed were checked by the Liverpool HEP interactions. 21
Measurement of fibrosis and classification of cirrhosis
Evaluation of liver fibrosis was made by histology or elastometry (FibroScan©); one of the two evaluations was carried out within two months before starting therapy. Advanced fibrosis was defined at histology by a Metavir score of F3 or a FibroScan ≥ 10 kPa but < 14 kPa. Cirrhosis was defined by a Metavir score of F4, or by a FibroScan ≥ 14 kPa or in presence of clinical, laboratory and ultrasound (US) parameters of clinical apparent cirrhosis. Portal hypertension was defined by the presence of esophago-gastro varices at endoscopy and/or detection of collateral veins at ultrasound.
For cirrhotic patients, Child-Pugh-Turcotte (CPT) classes and Model for End-Stage Liver Disease (MELD) scores were calculated. Cirrhotic patients were further stratified into five classes according to D’Amico classification. 22 They were classified as follows: compensated cirrhotics without or with endoscopic evidence of varices were included in class 1 or 2, respectively; decompensated cirrhotics were those with a history of variceal bleeding (stage 3), a previous, single episode of ascites or encephalopathy in the absence of esophageal bleeding (stage 4), and those with multiple, recurrent episodes of decompensation, including a bleeding event (stage 5).
Efficacy assessment and outcomes
Patients were reviewed monthly during the antiviral treatment and virological response was assessed by quantitative HCV-RNA at week 4, at the end of treatment, and at four and 12 weeks following the end of treatment. The SVR, defined as the persistent absence of detectable HCV-RNA in serum 12 weeks after the end of treatment (SVR12), was used as the primary outcome in this study. The secondary outcome was the identification of baseline characteristics that could predict treatment failure.
Meta-analysis
With the intent to delineate a treatment schedule with the new DAAs that would offer the higher chance of SVR to patients with HCV genotype 3, a systematic search of the literature data was implemented and the retrieved information was pooled and evaluated by a meta-analytical approach.
Selection of primary studies
Electronic, systematic review of the available evidence in the published literature, conducted in line with the Cochrane Handbook for Systematic Reviews of Interventions, 23 was undertaken to identify all studies. Secondly, the proceedings of the 2015 and 2016 Annual Meetings of the American Association for the Study of Liver Diseases and of the European Association for the Study of the Liver were hand-searched. The systematic literature review was performed via Medline from 2012 to 2016 by the following search keys: HCV Genotype 3 AND [(DAA)OR (sofosbuvir)OR(daclatasvir)OR(Ledipasvir)OR(Velpatasvir)]. The last search was on December 2016. Manual searching included reading through reference lists of relevant papers to capture missing studies that met our inclusion criteria. Three independent reviewers (FM, ML, RG) scanned every identified study to determine eligibility. Studies were excluded if they were in languages other than English, or reported data on HIV- or HBV-co-infected patients. Phase II and dose-finding studies were also excluded, as were duplicate publications, narrative reviews, and editorials. The studies may be focused only on genotype 3 patients or be studies with patients infected with all HCV genotypes; in this event, only data regarding for HCV genotype 3-infected patients were extracted and pooled into the analysis.
Data extraction and management
All information was extracted using a standardized data abstraction form. Retrieved data included: study characteristics (clinical trials or interventional studies); baseline patient characteristics, treatment history (naïve or treatment experienced); treatment regimens, including dose and duration; use of RBV; efficacy of therapy as ascertained by SVR12. Data were extracted by a single review author and checked by a second independent reviewer. Any disagreement was resolved through discussion.
Data synthesis
The primary efficacy outcome was SVR12 (undetectable HCV-RNA levels 12 weeks after the end of therapy). Separate analyses were performed in non-cirrhotic and cirrhotic patients, for each treatment regimen, and for subpopulations based on prior treatment experience (whether naïve or previously exposed to peg-IFN and RBV), on the concomitant use or not of RBV, and duration of therapy (12 or 16 or 24 weeks).
Statistical analysis
Baseline characteristics of patients were assessed with standard descriptive statistics. Categorical variables were reported as percentages and compared using chi-square (or Fisher’s exact test, when needed). Variables with a p < 0.05 were included in a multivariate stepwise logistic regression model. Regarding meta-analysis, proportions were the measure of interest and subgroup analyses were carried out. Random rather than fixed-effects models were used to estimate pooled proportions in order to account for heterogeneity. All analyses were performed using SPSS software, version 13.0 (SPSS Inc, Chicago, IL, USA) and Comprehensive Meta-analysis software (Version 2.2064, Biostat, Englewood, NJ, USA).
Results
Patients
A total of 233 HCV genotype 3 patients who met the inclusion criteria were extracted from the database of the collaborative ITAL-C consortium. Enrolled patients were adults with a median age of 52 years. The great majority of patients were males (82.0%) with liver disease progressed to the stage of cirrhosis (83.7%). Two-thirds of them were naïve to antiviral treatment. Eighty percent (157/195) of patients were in CPT class A, 15.4% (30/195) in class B, and the remaining eight patients (4.1%) in class C. On the basis of MELD scoring, 188 patients (96.4%) had a score <16, and seven patients (3.6%) a score ≥16. According to cirrhosis stage 98/195 (50.2%) patients were classified as class 1 (no signs of portal hypertension or previous decompensating events), while the remaining cirrhotic patients had clinically apparent cirrhosis.
Univariate analysis of factors affecting treatment outcome to DAAs in 233 HCV genotype 3-infected patients.
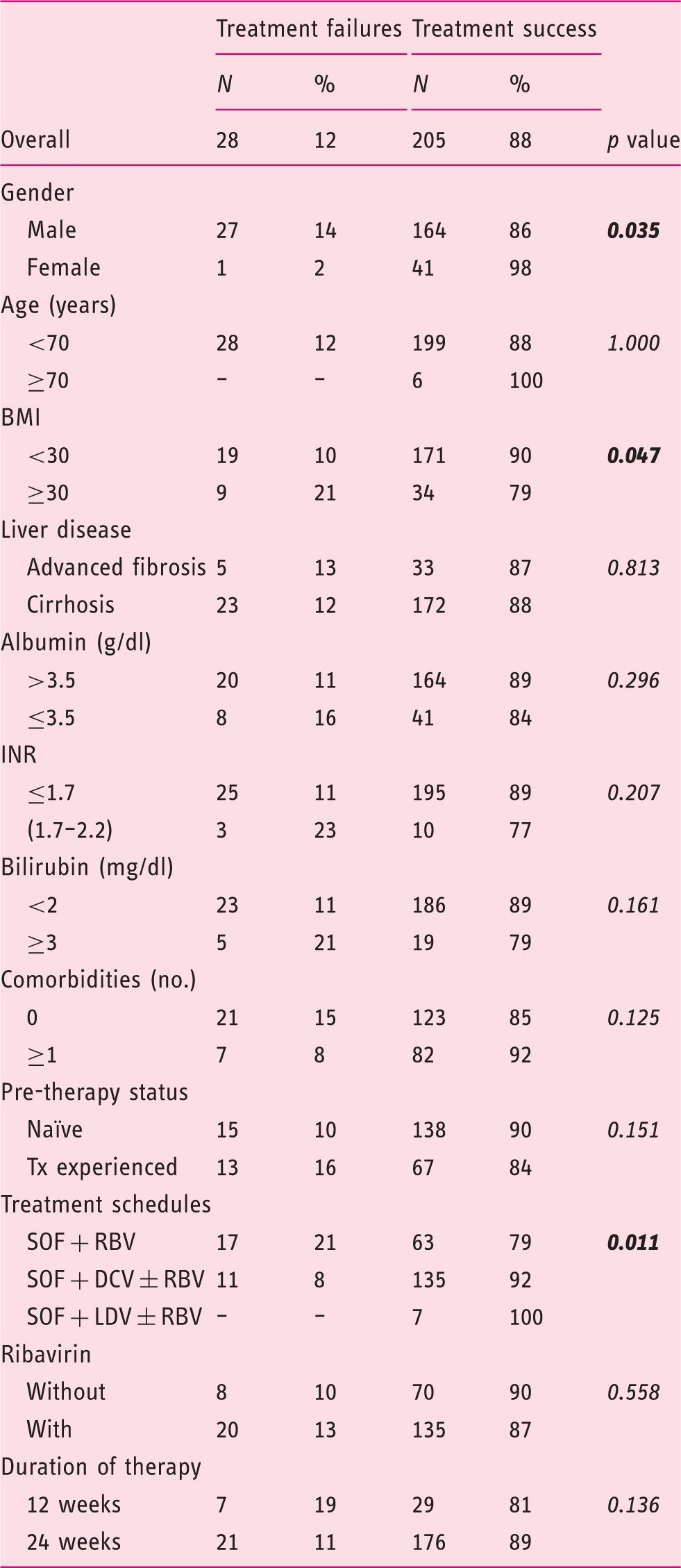
DAAs: direct-acting agents; HCV: hepatitis C virus; BMI: body mass index; INR: international normalized ratio; Tx: therapy; SOF: sofosbuvir; RBV: ribavirin; DCV: daclatasvir; LDV: ledipasvir.
Univariate analysis of factors affecting treatment outcome to DAAs in 195 HCV genotype 3-infected cirrhotic patients.
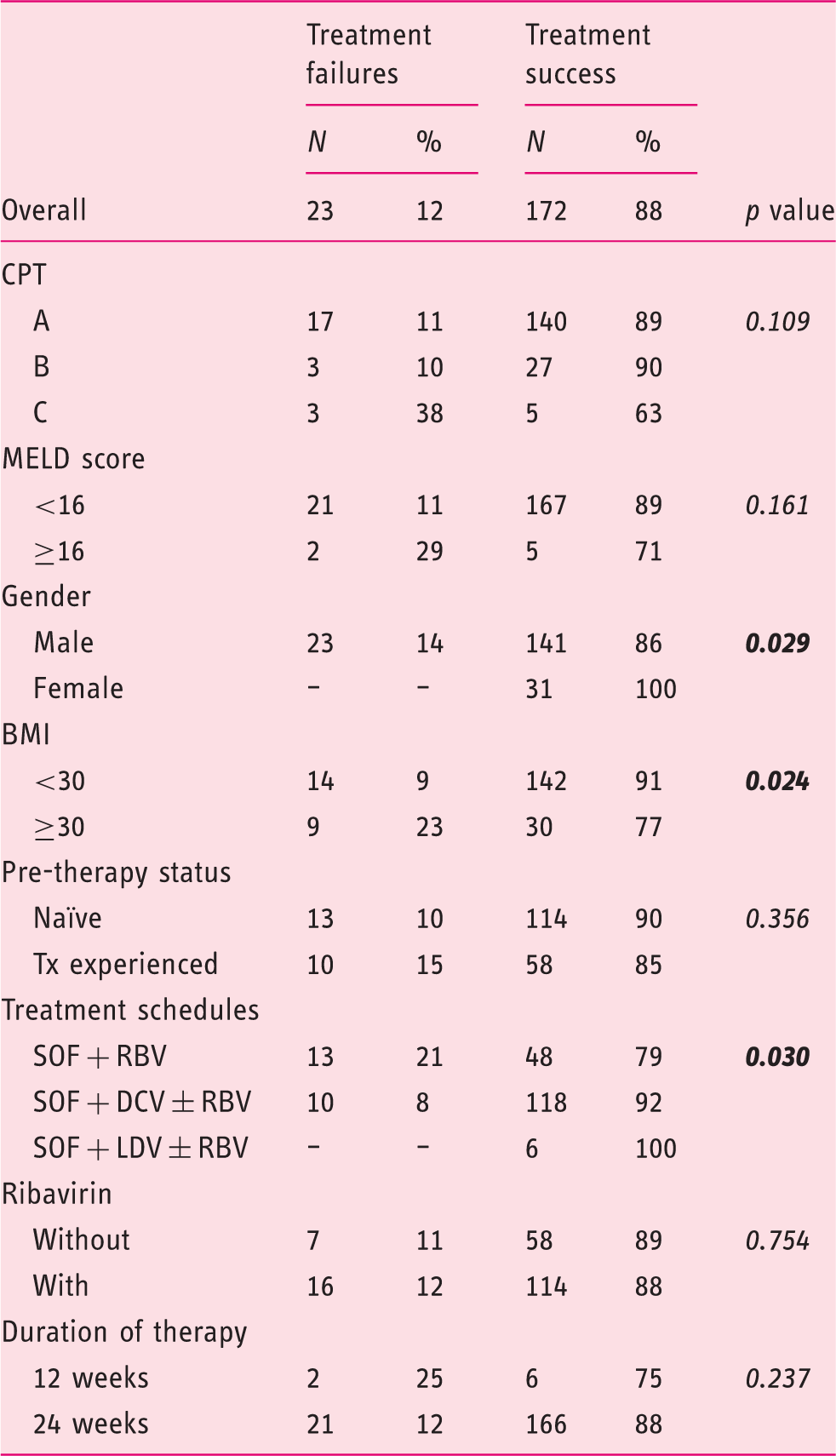
DAAs: direct antiviral agents; HCV: hepatitis C virus; CPT: Child-Pugh-Turcotte; MELD: Model for End-Stage Liver Disease; BMI: body mass index; Tx: therapy; SOF: sofosbuvir; RBV: ribavirin; DCV: daclatasvir; LDV: ledipasvir.
Meta-analysis
Figure 1 presents the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram for the selection of primary studies. Seventeen full text records (11 randomized controlled trials (RCTs) and six observational cohorts),6–8,11,13,14,24–34 the present investigation and two abstracts15,35 were retained for analysis. Main clinical features of the 3311 patients treated with different schedules of the DAAs are shown in Table 3, and the meta-analytical data in Figure 2. Overall, the mean weighted SVR12 rate was 84.4% (95% CI: 80.4–87.8). The three schedules of antiviral treatment yielded numerically different SVR12 rates, which varied from 79.0% (95% CI: 70.9–85.3) in the regimen comprising sofosbuvir with RBV, to 83.7% (95% CI: 66.2–93.1) when sofosbuvir was given in combination with ledispavir, and 88.2% (95% CI 83.3–91.7) with a combination of sofosbuvir with daclatasvir.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) diagram for the processing of literature data. Outcomes following treatments in 3316 HCV genotype 3-infected patients. HCV: hepatitis C virus; SVR: sustained virological response; SOF: sofosbuvir; RBV: ribavirin; DCV: daclatasvir; LDV: ledipasvir; TOT: total. Weighted estimates of SVR12 rates in all clinical trials evaluating the efficacy of the different antiviral regimens with the new direct-acting agents in patients with HCV genotype 3 infection. The results are broken down according to administered regimen.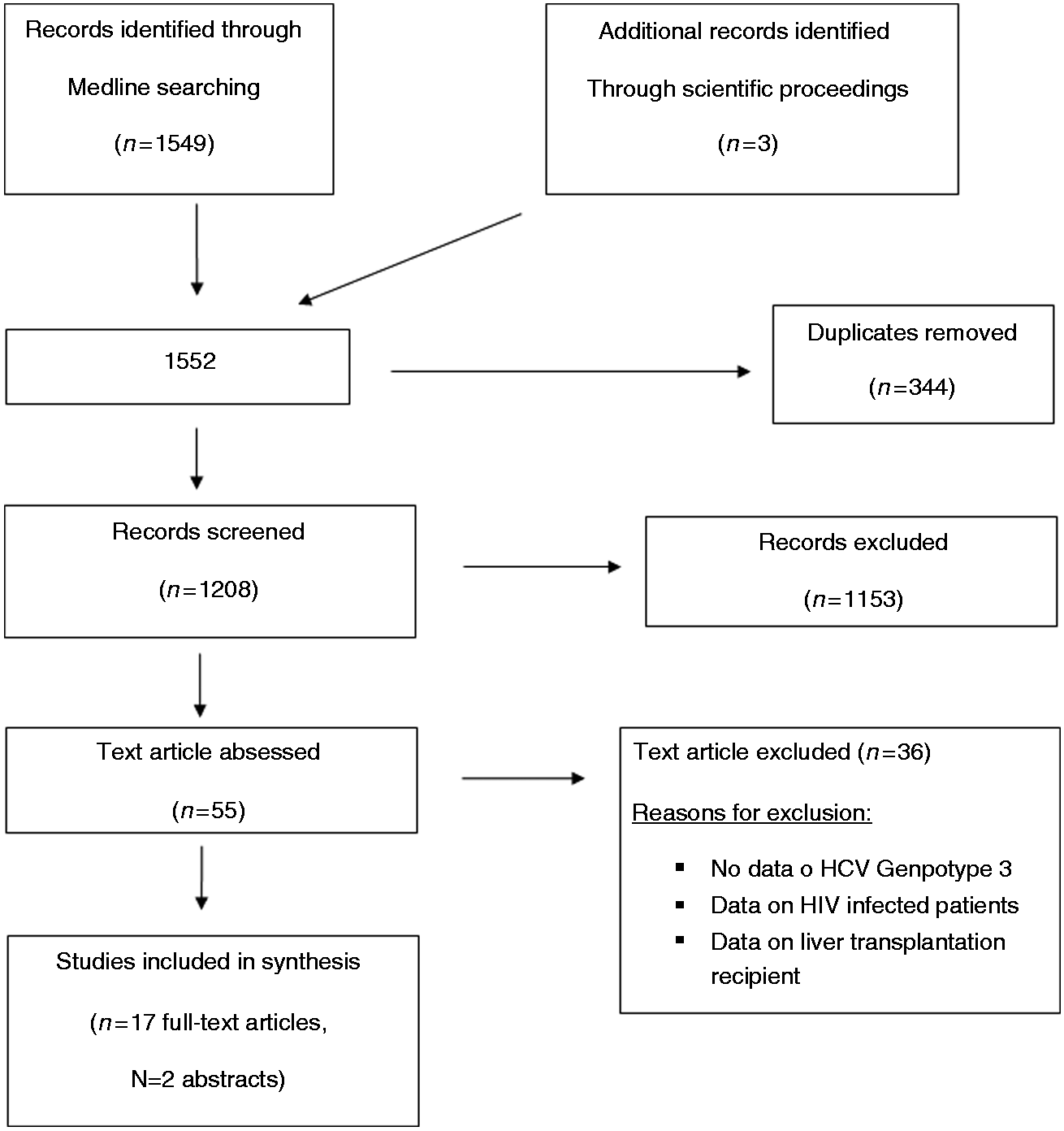
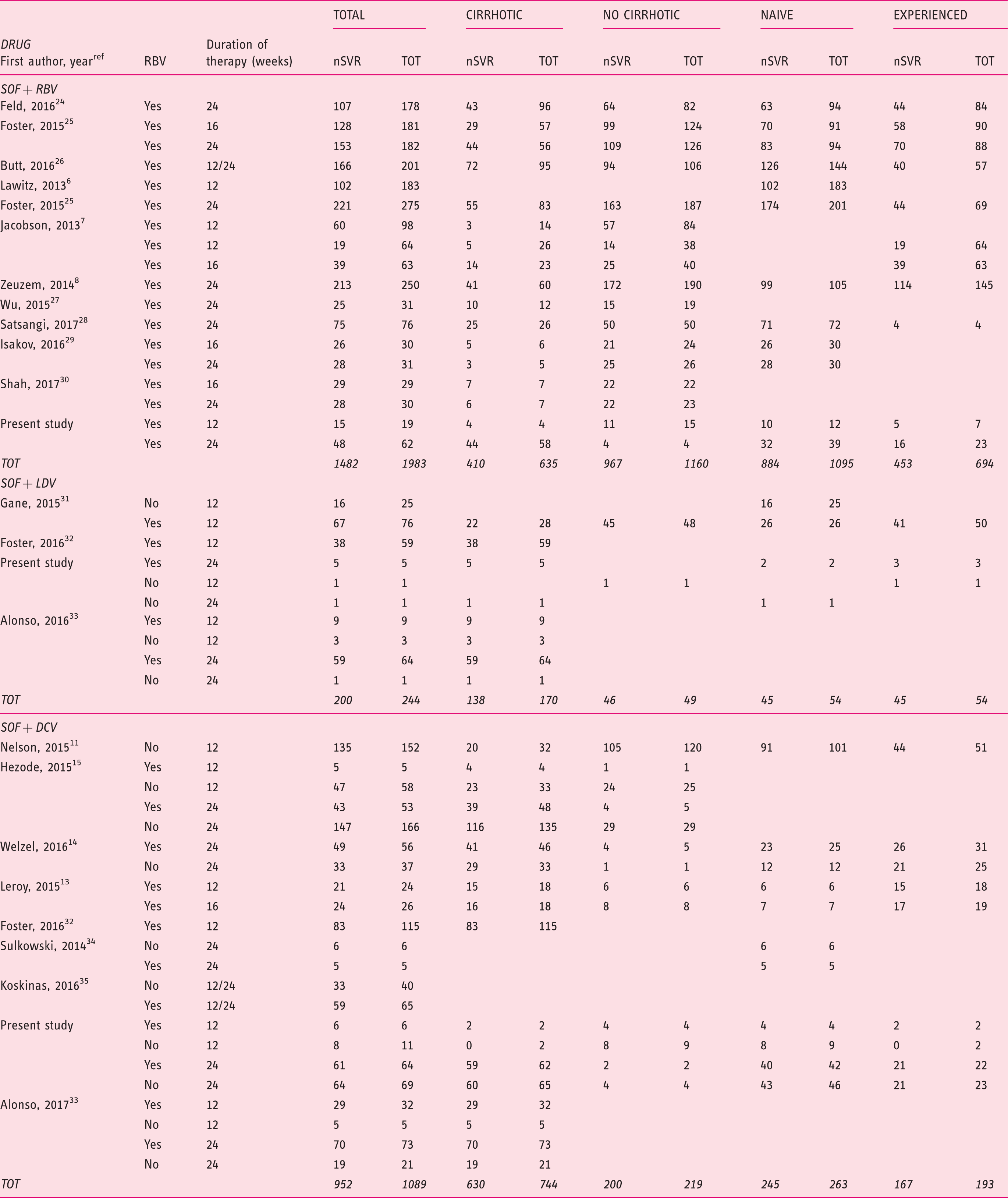
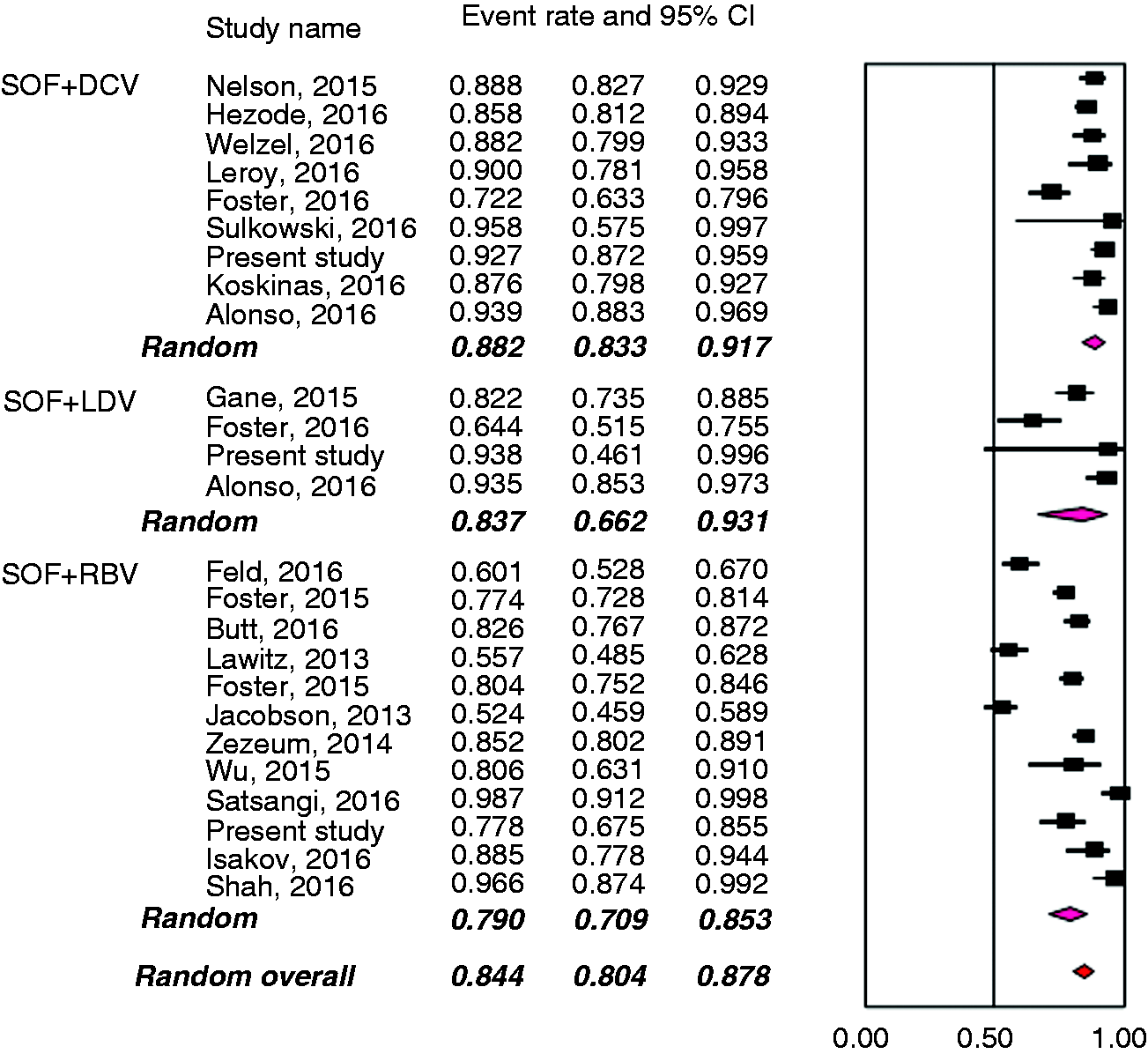
Weighted estimates of SVR12 rates of 3311 HCV genotype 3-infected patients, broken down by treatment regime.
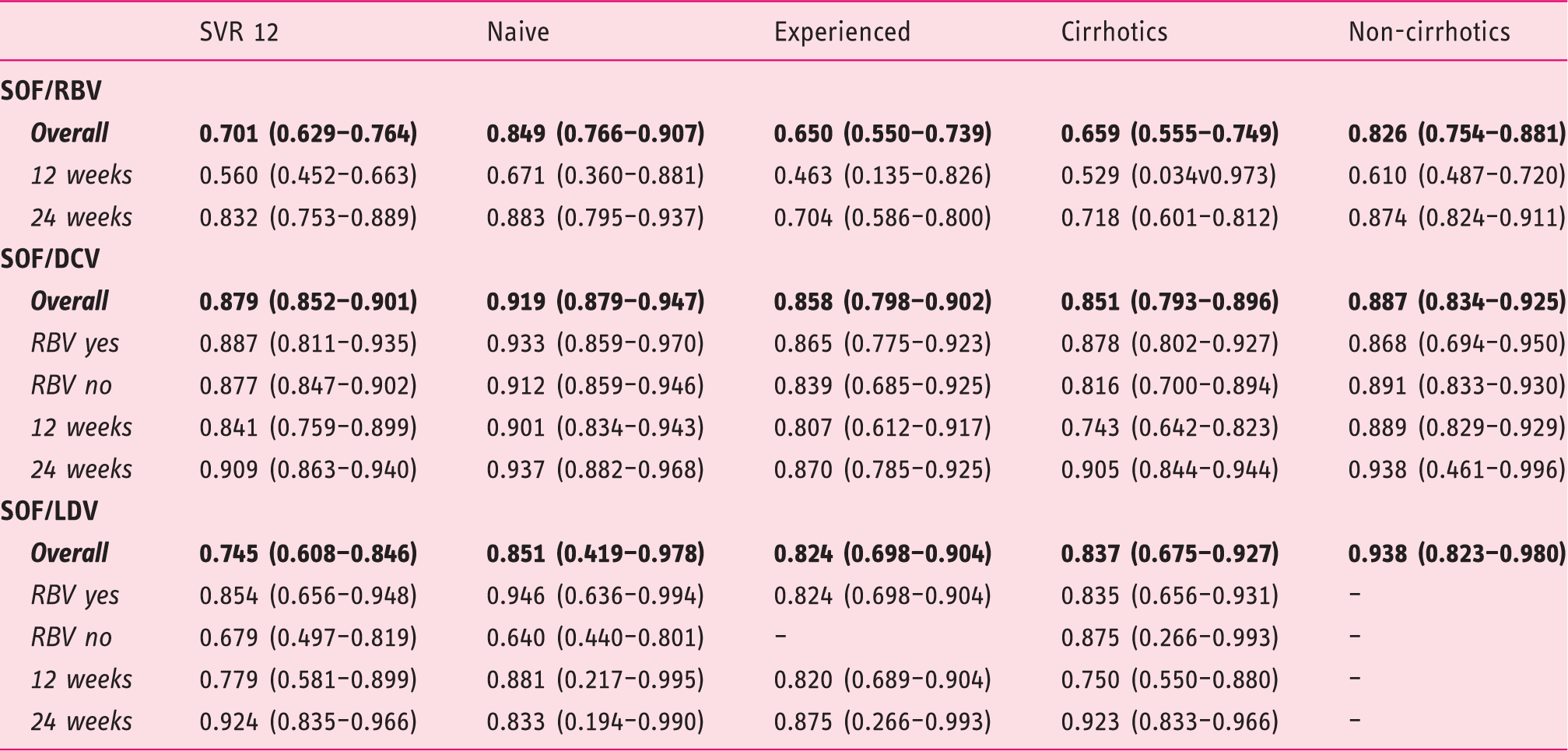
SVR: sustained virological response; HCV: hepatitis C virus; SOF: sofosbuvir; RBV: ribavirin; DCV: daclatasvir; LDV: ledipasvir.
Eleven studies, including the present investigation,6–8,24–30 evaluated the regimen of sofosbuvir plus RBV, administered to a total of 1983 patients: The SRV12 rates were higher when treatment lasted for 24 weeks, in naïve patients compared to experienced patients, and in non-cirrhotics compared with those with cirrhosis.
Information on the outcome following treatment with sofosbuvir/daclatasvir with or without RBV was retrieved from eight studies11,13–15,32–35 and the present investigation; a total of 1089 patients received this therapeutic regimen. The inclusion of RBV in the schedule did not affect the therapeutic outcome in the different subgroups of patients, whether naïve or treatment experienced, whether cirrhotics or non-cirrhotics. Treatment duration did not influence the outcome in the subgroups of naïve patients and in those without cirrhosis, whereas prolongation of treatment to 24 weeks produced higher SVR12 rates in treatment-experienced patients and in those with liver cirrhosis.
The combination of sofosbuvir/ledispavir has been evaluated to date in only 244 patients enrolled in three studies31–33 and in the present investigation. With this therapeutic regimen, higher SVR rates were registered when RBV was included in the therapeutic schedule and when therapy lasted for 24 weeks. When patients treated with this combination therapy were subdivided by difference in the administered regimen, the value of adding RBV was documented only in naïve patients, and the benefit of 24 weeks’ treatment duration persisted only in those with cirrhosis.
Discussion
The optimal regimen of the new DAAs for patients with HCV genotype 3, and especially for those who have progressed to liver cirrhosis, is still an unsolved issue. The regimen containing sofosbuvir in combination with RBV is currently recommended;17,19 however, this schedule may ensure an optimal outcome for non-cirrhotic patients treated for 24 weeks, but remains far from ideal in cirrhotic patients, whether naïve or treatment experienced. Real-life studies or compassionate use programs have reported better outcome when a combination of two DAAs was administered to HCV genotype 3-infected patients.14,15 In these programs, SVR rates as high as 86% were reported in cirrhotic patients, whether naïve or treatment experienced, using a 24-week regimen.14,15 These promising results, however, emerged from studies with a limited number of genotype 3 patients, so that the information cannot be considered conclusive.
Our study enrolled a large cohort (n = 233) of patients infected by the HCV genotype 3, the majority of them presenting with liver cirrhosis. A merit of this investigation is the simultaneous evaluation of the outcome following therapy with all available DAA schedules: Indeed, only 34.3% (80/233) of our patients were treated only with an NS5B inhibitor (sofosbuvir) in combination with RBV, whereas for the remaining two-thirds of patients, two NS5A inhibitors (daclatasvir or ledipasvir) were administered in combination with sofosbuvir, with or without RBV. We found an overall SVR12 rate of 88.0%, without difference in non-cirrhotic vs cirrhotic patients (87.0% vs 88.0%), and in treatment-naïve vs experienced patients (90.0% vs 84.0%). The SVR rates differed in relation to BMI (favoring BMI < 30) and to the schedule of treatment used (favoring sofosbuvir in combination with daclatasvir or ledispavir).The most relevant finding of our study was the demonstration of a similar high efficacy of schedules containing an NS5A inhibitor (daclatasvir or ledipasvir) in association with sofosbuvir, and independently from the use of RBV in cirrhotic vs non-cirrhotic patients (92.2% vs 94.4% for daclatasvir, and 100% vs 100% for ledipasvir).Of relevance, the prior two schedules independently predicted an SVR in the entire cohort of patients, as well as in the cirrhotic cohort. This finding is in contrast with data coming from registration trials using the same schedule of drugs but showing significant different rates of SVR in non-cirrhotic (96%) and cirrhotic patients; in the latter subset of patients the reported rates varied from 63% to 83% for a treatment duration of 12 weeks, to 89% for 16 weeks of treatment.11,13 No information from registration trials is available concerning a prolonged treatment of 24 weeks. On the other hand our results seem to be consistent with and even better than those observed in real-life studies. In particular, the European and French Expanded Access Program showed an improvement of the rate of SVR, ranging between 81% and 88% when the treatment with sofosbuvir + daclatasvir was extended to 24 weeks, with no difference between RBV-free and RBV-containing regimens.14,15 Overall, our data, in agreement with prior observations, sustain the possibility that the high rate of relapse previously observed in cirrhosis HCV-3 patients might be related to the use of sub-optimal schedules of therapy (sofosbuvir and RBV) and indicate as an optimal schedule the use of sofosbuvir in combination with either of the two NS5A inhibitors.
It is important to note that in our survey the stage of cirrhosis did not represent a factor influencing the rate of SVR, even if a numerically lower rate of success was obtained in Child C class (63.0%). Although the number of Child C patients in our study was low, our results are similar to the figure from larger studies focused on decompensated cirrhosis.14,15,32
HCV infection is known to be associated with hepatic steatosis. In the case of genotype 3 infection, this is clearly a cytopathic effect of the virus because it is present in approximately two-thirds of infected individuals and can resolve completely in patients who achieve an SVR to therapy. 36 In the era of IFN-based therapies, liver steatosis was a known negative predictor of SVR, while its role in DAA-based therapies is not established. Unfortunately steatosis was not a variable included in the original database; therefore, although it would have been interesting data we cannot speculate on this matter and further studies are needed.
With the aim to reinforce the identification of the best IFN-free DAA regimens to be administered to patients with genotype 3 infection, we conducted a systematic review of the literature and combined data from 20 studies (including the present one) on a total of 3311 HCV-genotype 3 patients. In our meta-analysis we did not include the schedule with sofosbuvir in association to the third-generation NS5A inhibitor velpatasvir, as only a single trial has been made available so far. 12 The results per regimen demonstrated that the combination that achieved the highest SVR12 rate for the overall population of HCV 3-infected patients was the combination of sofosbuvir/daclatasvir, which achieved SVR12 rates ∼90% in naïve non-cirrhotic patients with a short treatment duration of only 12 weeks, and without the need for RBV co-administration. However, this regimen needs to be administered for 24 weeks in patients with cirrhosis and in those who failed a previous course of antiviral therapy. The large number of patients treated with this therapeutic schedule (n = 1989) lends support to our conclusions.
In our meta-analysis we also considered the therapeutic outcome after treatment of 1983 genotype 3-infected patients with the still recommended schedule of sofosbuvir with RBV. The registered SVR12 rates in these patients were inferior to those seen when two DAAs (sofosbuvir and daclatasvir) were administered. Unacceptably low rates were registered when the sofosbuvir/RBV regimen was given for 12 weeks, whereas the rates improved by extending treatment duration to 24 weeks; however, this could remain a therapeutic option only for the subset of genotype 3-infected patients naïve to therapy and who have progressed to a non-cirrhotic stage. Based on this evidence, we would suggest this schedule should no longer be considered for these patients, considering the availability of more effective regimens.
Finally, we also had the opportunity to review the data on the efficacy of the regimen contemplating the use sofosbuvir in combination with ledispavir. The registered SVR12 rates following this schedule were superior to the results achieved with the administration of sofosbuvir/RBV but numerically inferior to those with the combination of sofosbuvir and daclatasvir. From the available information it seems that the sofosbuvir/ledispavir combination would be effective whenever RBV was included in the schedule and treatment prolonged to 24 weeks. However, we acknowledge the small number of patients evaluated in our investigation (n = 244) and the large range of CIs for several sub-analyses on different subgroups of genotype 3 patients treated with this last combination of two DAAs. Previous limitations did not allow us to draw a firm conclusion on the efficacy of this therapeutic regimen. Further evaluation of this regimen in HCV genotype 3 patients is warranted.
In conclusion, the results of this investigation further reinforce the concept that HCV genotype 3-infected patients, and in particular those who have progressed to cirrhosis, should no longer be considered difficult-to-treat individuals, provided that an optimal therapeutic schedule is applied. According to the presented data, the optimal therapeutic regimen for HCV genotype 3-infected patients without cirrhosis appears to be the combination of sofosbuvir with daclatasvir, administered for 12 weeks, without the use of RBV. In difficult-to-treat patients the results coming from meta-analysis suggest extending therapy to 24 weeks with or without ribavirin.
Footnotes
Acknowledgments
Co-authors of the ITAL-C consortium: Maria Guarino (Gastroenterology Unit, Department of Clinical Medicine and Surgery, University “Federico II” of Naples); Michele Barone (Section of Gastroenterology, Department of Emergency and Organ Transplantation, AOU Policlinico, University of Bari);Vito Carretta (Liver Unit, Department of Internal Medicine, Hospital of Venosa); Immacolata Carraturo (Division of Infectious Diseases “Fazzi” Hospital, Lecce); Giuseppe Cuccorese (Division of Internal Medicine, “Monsignor Dimiccoli” Hospital, Barletta).
Ethics approval
The study protocol was approved July 27, 2016 by the Ethics Committee of the Casa Sollievo della Sofferenza Hospital, in San Giovanni Rotondo, Italy. Each patient included in the study provided a written informed consent. All the study’s procedures were conducted according with the provisions of the Declaration of Helsinki and Good Clinical Practice Guidelines.
Informed consent
Written consent was obtained from the patient for publication.
Conflict of interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
