Abstract
Background
Detection of early oesophageal cancer in surrounding normal tissue can be challenging, but detection is essential to determine the subsequent treatment. Dysplastic tissue can be detected by using electrical impedance spectroscopy (EIS).
Objective
The aim of the present study was to evaluate the feasibility and value of EIS in the diagnosis of oesophageal neoplasia.
Methods
This prospective ex-vivo study included 23 patients with early oesophageal cancer (17 with Barrett’s cancer and six with early squamous cell cancer). Immediately after endoscopic resection, the electrical properties of the resected specimens were investigated using a pencil probe (5 mm in diameter, frequency range from 100 Hz to 1 MHz). Punch biopsies were taken from the measured site in order to compare the results of EIS with histology.
Results
EIS was able to detect dysplastic oesophageal mucosa with a high rate of accuracy (82% in Barrett’s oesophagus and 100% in squamous oesophagus) A total of 54 different sites in 26 tumours were evaluated.
Conclusions
EIS was able to differentiate reliably between non-neoplastic and neoplastic oesophageal mucosa. Using EIS, it might be possible to use it for targeted biopsies and to avoid unnecessary biopsies during cancer surveillance in future.
Introduction
Oesophageal cancer has an increasing incidence in the world. 1 Oesophageal Barrett’s neoplasia is at present in the Western world more frequent than squamous cell carcinoma of the oesophagus. 1 The epithelial changes pass through premalignant stages, known as Barrett’s metaplasia, low-grade dysplasia (LGD), and finally high-grade dysplasia (HGD)/adenocarcinoma. 1 In recent years, there has been a shift in therapy from radical oesophagectomy to local endoscopic treatment for early stages of oesophageal cancer. It is therefore desirable to detect neoplastic lesions at an early stage, in order to allow curative endoscopic treatment.
However, HGD and early oesophageal cancer are often hard to detect, as only subtle mucosal abnormalities are visible on white-light endoscopy. Most guidelines have so far recommended carrying out targeted biopsies of suspicious lesions and four-quadrant biopsies in Barrett's epithelium. These untargeted biopsies have a low diagnostic yield and are cost-intensive and time-consuming. It would therefore be desirable to develop a technique that could be applied easily as a virtual biopsy, with immediate diagnosis and a high accuracy for detecting neoplastic lesions without the need for advanced skills on the part of the endoscopist.
Biological tissues have complex electrical impedance, depending on their biological structure.2,3 When electrical potential is applied to tissue (e.g. oesophageal tissue), the current flows through the intracellular spaces (at high frequencies) and extracellular spaces (at low frequencies). 4 The oesophageal mucosa is usually covered with layers of squamous cells. This type of epithelium is compact, with tight intercellular spaces, resulting in a relatively high electrical resistance. In precancerous conditions and inflammation, the integrity of the mucosa is altered and the extracellular spaces are dilated. This alteration can be assessed using an intraluminal impedance technique, by measuring a drop in the resistance. 5
During the progressive development of cancer, the biological cell structure changes due to chromatin modification and an expanding cell volume, which results in a reduction in the extracellular space. With the reduction in extracellular space, the resistance for low-frequency current, mostly flowing through the extracellular space, increases, resulting in a greater impedance magnitude in the lower-frequency range. 6 These changes in electrical properties can be used to differentiate between healthy oesophageal tissue and neoplastic lesions.
The aim of the present study was to investigate the feasibility and value of bioimpedance spectroscopy in human oesophageal cancer in comparison with non-neoplastic oesophageal mucosa.
Patients and methods
This study was designed as a prospective outcome study and fulfilled the requirements for the local ethics committee in the state of Hesse (Germany). It was registered in the German Clinical Trials Register (no. DRKS00003358).
Seventeen consecutive patients with early Barrett’s carcinoma and six patients with early squamous cell carcinoma in whom endoscopic resection was planned were included in the study. The patients’ mean age was 64 years (range 46–85 years, interquartile range 56–70); there were 18 men and five women.
Endoscopic resection was performed using a ligation device without submucosal injection, as described previously. 7 Immediately after removal of the target lesion, the resected specimens were pinned onto cork and different sites on them were investigated for their electrical impedance properties using a pencil probe (5 mm in diameter) incorporating four gold electrodes with a diameter of 0.5 mm. Measurements were performed using a frequency range from 100 Hz to 1 MHz, with an applied input potential of 20 mV. The electrodes were connected to an impedance analyzer (Solartron 1260 and Solartron 1294; Solartron Analytical, AMETEK Advanced Measurement Technology, Farnborough, UK). Calibration was performed by placing the probe in phosphate-buffered saline with known conductivity.
A total of 54 different sites were evaluated in 47 endoscopic resection specimens. Immediately after the measurement, a punch biopsy (Biopsy Punch; Stiefel, Research Triangle Park, NC, USA) with a diameter of 5 mm was taken precisely from the measured site in order to compare the results of the impedance measurement with the histological results.
As the electrical impedance magnitude increases when greater pressure is applied to the measuring probe,
8
the pressure was kept constantly around 4.5 kPa during all measurements. This was achieved by pressing the probe downwards until the scale underneath showed a value close to 9 g (Figure 1). Finally, the punch biopsy and resected specimens were sent to pathology for further histological evaluation. The pathologist was blinded to the results of the impedance measurements.
The experimental set up, with the impedance probe and a scale for pressure measurement underneath.
Histological and statistical analysis
The measurements were divided into two groups on the basis of the patients’ diagnoses. The first group consisted of lesions deriving from Barrett’s mucosa, and the second of lesions originating in the squamous mucosa of the oesophagus. The Barrett’s group was also divided into two subgroups depending on the histological results: Barrett’s metaplasia/LGD or HGD/T1 carcinoma (m1–sm3). The squamous mucosa group was also divided into two subgroups, with normal squamous tissue/LGD or HGD/T1 carcinoma (m1–sm3).
The arithmetic mean for the impedance magnitude was then calculated and correlated with the subgroups. The arithmetic mean was defined as the characteristic impedance magnitude for neoplastic or non-neoplastic lesions in the two major groups.
Finally, the characteristic impedance magnitude was compared with the histological findings for each measurement, and the sensitivity, specificity, and accuracy were calculated. On a receiver operating characteristic (ROC) curve, the cut-off value was chosen carefully in order to obtain a preferably high sensitivity for the new method. Statistical calculations were performed with PASW 18 (SPSS, IBM Corporation, New York, USA).
Results
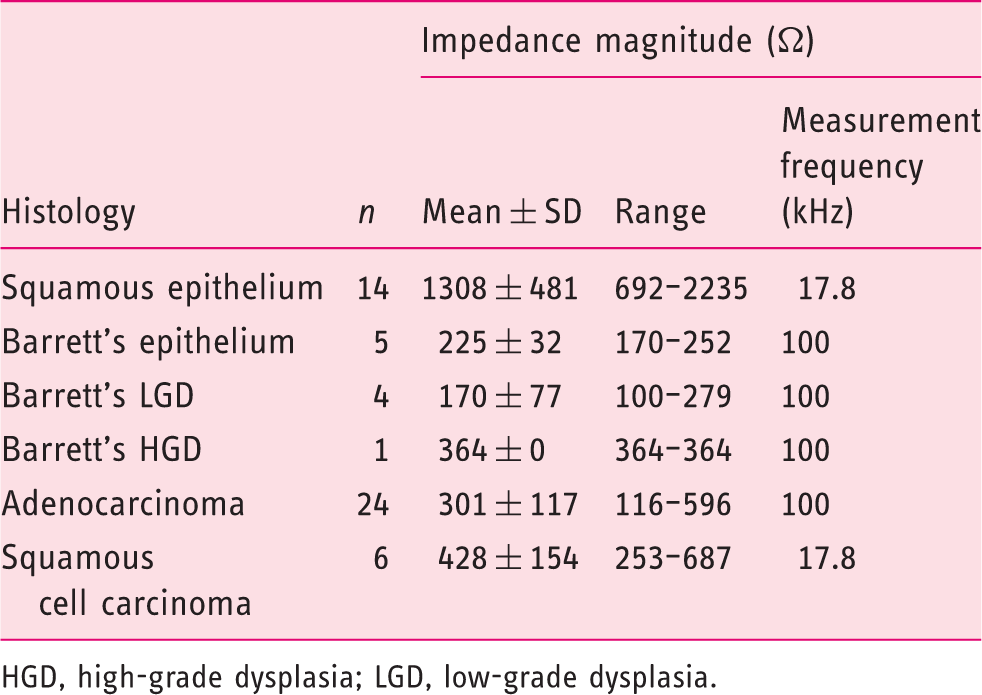
Impedance from 100 Hz to 1 MHz was measured from 54 samples in 23 patients. Impedance magnitudes |Z| are shown as an arithmetic mean over the whole frequency range for Barrett’s epithelium/LGD and HGD/adenocarcinoma in Figure 2. The arithmetic mean over all frequencies for normal oesophageal squamous tissue and squamous cell carcinoma is shown in Figure 3. The histological results are shown in Table 1.
Impedance spectroscopy over 17 frequencies, comparing the histological results of Barrett’s epithelium with high-grade dysplasia/adenocarcinoma. Impedance spectroscopy over 17 frequencies, comparing the histological results of squamous epithelium and different stages of squamous cell cancer. Detailed histological results and mean impedance magnitude HGD, high-grade dysplasia; LGD, low-grade dysplasia.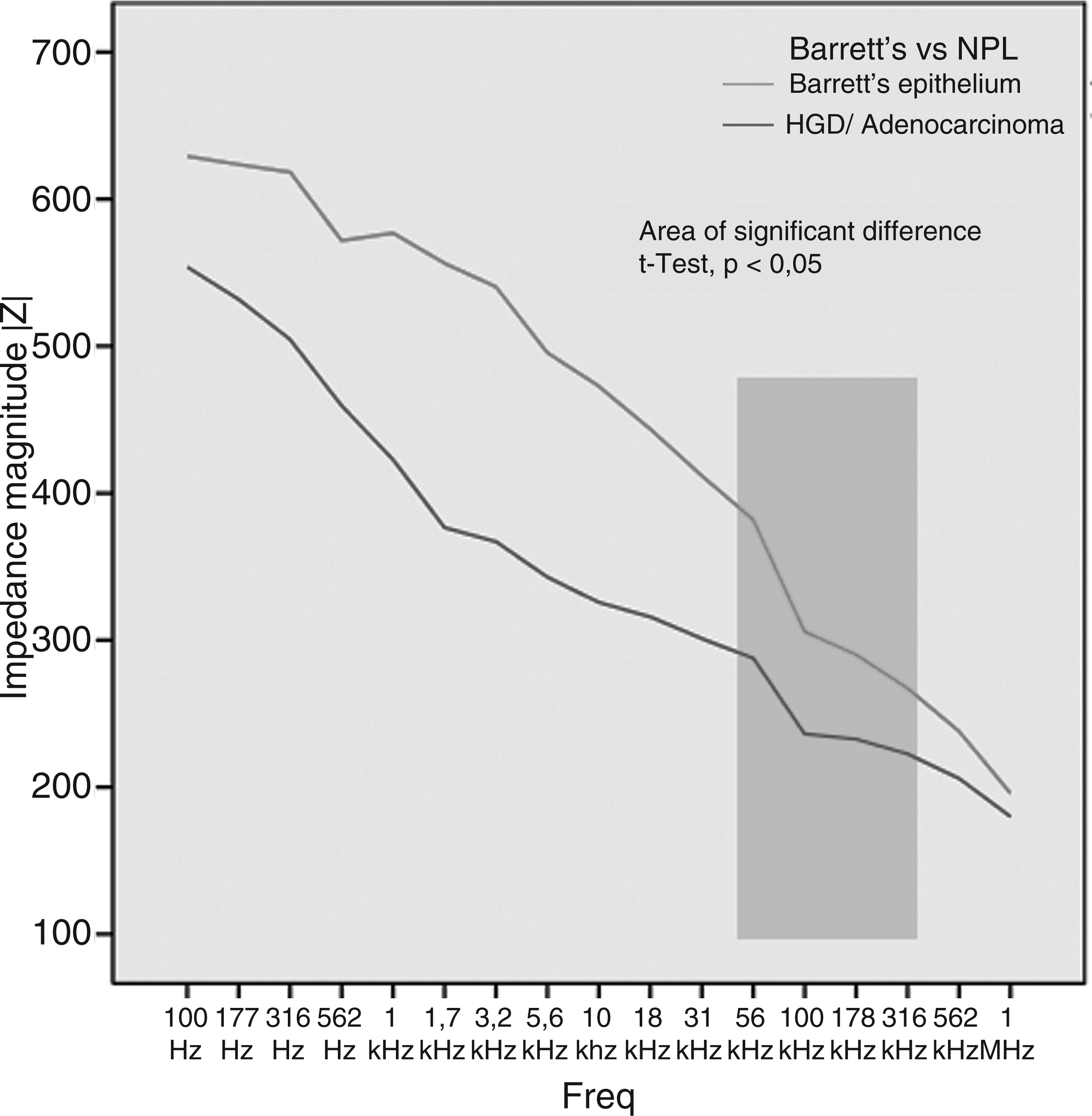
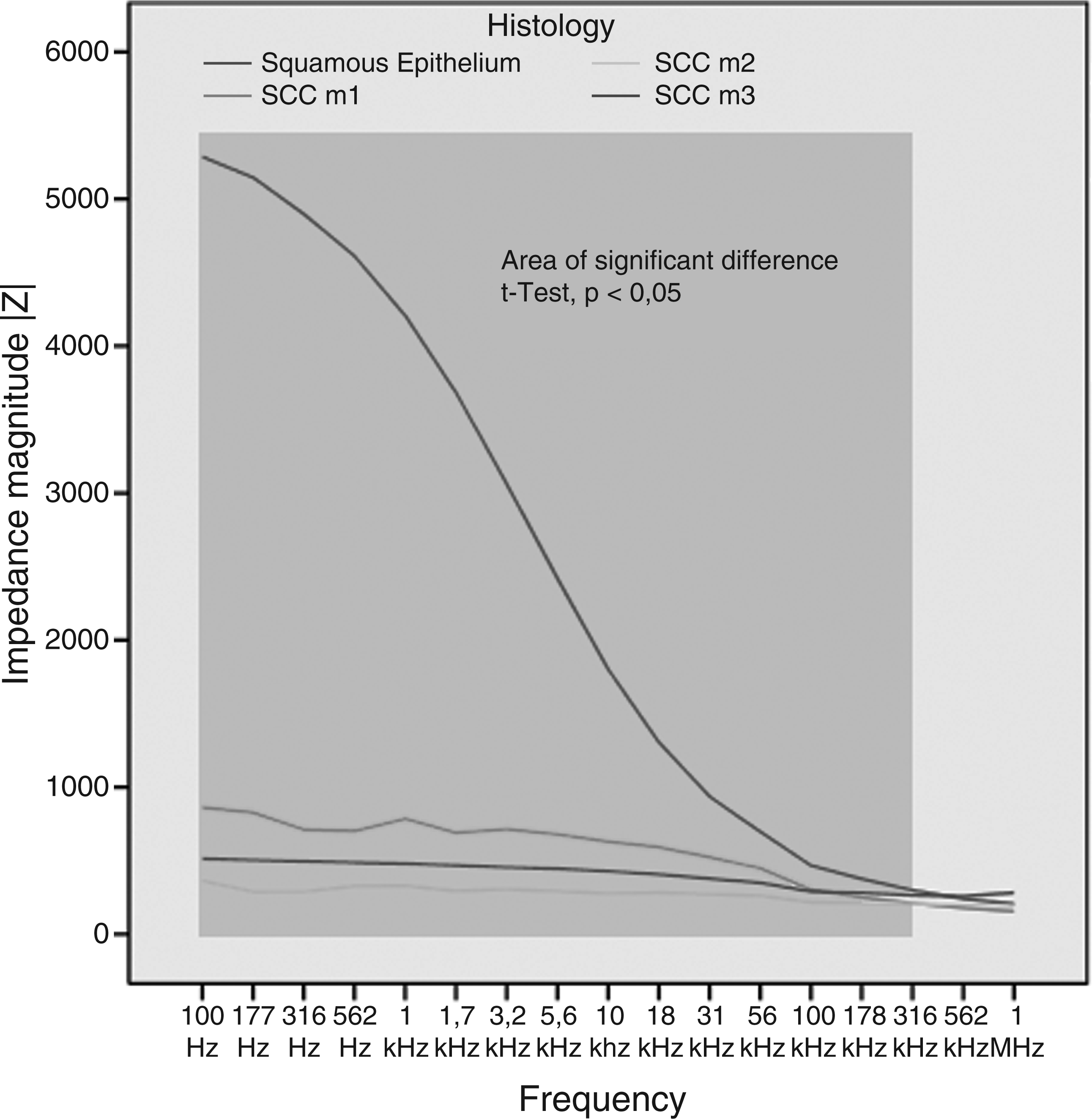
In Barrett’s epithelium, the measured impedance magnitude showed a significant difference only in a small frequency interval (from 56 kHz to 316 kHz) (t-test, p < 0.05) for differentiation between non-neoplastic Barrett’s mucosa and HGD/adenocarcinoma. In squamous epithelium, the impedance magnitude values showed a highly significant difference (t-test, p ≤ 0.004) almost over the whole frequency range measured (from 100 Hz to 316 kHz) for differentiation between normal squamous epithelium and squamous cell cancer.
With regard to the significant frequencies alone, ROC analysis showed the best predictive potentials at 100 kHz for HGD/adenocarcinoma and 17.8 kHz for squamous cell carcinoma, respectively. A cut-off value with an impedance magnitude of >174.84 Ω suggested a high likelihood of a diagnosis of adenocarcinoma, and a magnitude of <689.20 Ω was always malignant in the presence of squamous tissue.
Electrical bioimpedance was able to differentiate between neoplastic and non-neoplastic Barrett’s oesophagus and squamous epithelium with an accuracy of 82% in Barrett’s oesophagus and 100% in squamous oesophagus. The results are shown in Table 2. The sensitivity and specificity of the novel method were 97% (95% CI 82–100%) and 78% (95% CI 56–92%), respectively, for detection of malignant lesions in the oesophagus. The positive predictive value was 86% (95% CI 69–95%) and the negative predictive value was 95% (95% CI 72–100%). The positive likelihood ratio was calculated with 4.45 and the negative likelihood ratio 0.04.
Discussion
The detection of dysplasia in early oesophageal cancer is essential for determining the subsequent treatment. Early stages of oesophageal cancer can be treated with endoscopic resection, whereas advanced neoplasia may lead to oesophagectomy. There are as yet no established methods or devices enabling the endoscopist to reliably identify subtle neoplastic lesions during everyday routine work, although some recently described methods may be promising.
As shown in the present series, bioimpedance spectroscopy can reliably discriminate between normal oesophageal epithelium and squamous and adenocarcinoma in human tissue ex vivo. Although there are different approaches in the field of new imaging-techniques in the oesophagus, none of them have so far become established in clinical routine work. There is a need for a simple-to-use, easily applied probe-based device that is able to distinguish between normal, inflammatory, metaplastic, and neoplastic tissue without the need for the endoscopist to have advanced skills. A device of this type would save time and money and would significantly improve the detection of neoplastic lesions in Barrett’s and squamous oesophagus.
It is known that biological tissue with inflammation or dysplasia changes its electrical properties, which can be monitored using EIS. Recent studies have suggested that the impedance technique is capable of detecting precancerous lesions in a wide range of different tissues. Impedance technology has been shown to differentiate reliably between benign and malignant tissue in the prostate 9 and to be able to identify malignant melanoma, 10 cervical cancer, 11 and malignant tissue in the upper gastrointestinal tract.12–14
In addition to several smaller pilot studies, there have been two larger studies investigating the impedance technique in malignant lesions. In a Europe-wide multicentre study, 10 183 patients underwent probe-based impedance spectroscopy for detection of malignant melanomas. The authors reported a sensitivity of 95% and specificity of 49% for the novel method. In a study including 176 women, Abdul et al. 11 reported a sensitivity of 74% and a specificity of 53% for detection of cervical intraepithelial neoplasia. The first transendoscopic impedance measurement in gastric tissue was recently described by Keshtkar et al. 15 Although significant differences between gastric lesions were confirmed, there was considerable overlapping in the impedance curves.
Interestingly, the studies mentioned above reported relatively lower accuracy rates in comparison with the present results. This may be due to the fact that in the other studies, the pressure applied during the measurements may not have been adequately controlled. This is of course essential to ensure comparability between trials.8,16 Further developments are needed, including the design of a probe that can be advanced through the endoscope. The new technique may then open up a wide range of possible uses in the upper and lower gastrointestinal tract, including the bile ducts.
In the present study, LGD and non-neoplastic Barrett’s epithelium were found to have significantly lower transepithelial resistance than HGD and adenocarcinoma. In healthy squamous epithelium, the impedance magnitude is significantly higher than in early stages of squamous cell cancer. The new technique may therefore be useful particularly in cancer-screening programmes.
On the basis of these results, an impedance magnitude greater than 174.84 Ω would suggest that a biopsy should be taken in Barrett’s epithelium. This easy decision criterion would be suitable for all endoscopists, with no need for special training. Transferring these results to squamous epithelium, a drop in the impedance to <689.20 Ω would indicate squamous cell carcinoma with a very high degree of accuracy.
Although the results of the present study are very promising, it has some limitations. The specificity observed was only moderate. In addition, the specimens were investigated immediately after endoscopic resection, but not in vivo. In our experience, time has an influence on the accuracy of EIS. Moreover this, the use of diathermic current during resection may lead to potential tissue damage that might result in alterations in the impedance values. The data are therefore transferable to the clinical setting only to a limited extent. Also endoscopic resection was only performed in diagnosed oesophageal cancer. It remains to be clarified whether molecular field effects from neoplastic tissue to non-neoplastic tissue have an influence on the impedance measurements and whether the device therefore needs to be evaluated in patients without neoplasia. Further on, it has to be investigated whether diagnostic tools like Lugol staining and acetic acid have an influence on the new device. It is conceivable that prior to EIS the use of phosphate-buffered saline with known conductivity is necessary.
In conclusion, EIS was able to reliably differentiate ex vivo between non-neoplastic and neoplastic oesophageal mucosa in patients with known neoplasia. Therefore EIS holds the potential for further development to target biopsies during surveillance endoscopy. To the best of our knowledge, this is the first time that early squamous carcinoma has been evaluated using EIS. Further trials are required in order to clarify the diagnostic impact of this new technique and to evaluate whether fewer biopsies may be required as a result of it during the diagnostic work-up in patients with oesophageal cancer. Bioimpedance spectroscopy may thus have the potential to serve as a method of virtual biopsy in the future.
Trial registration
Results of impedance spectroscopy in comparison with histological findings
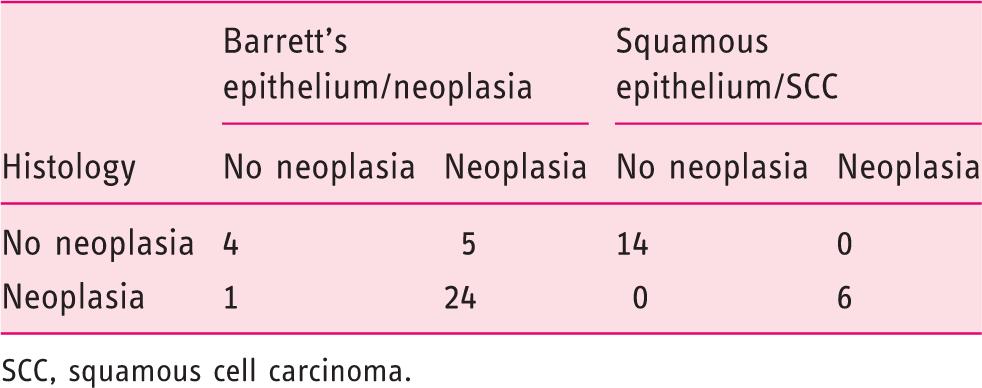
SCC, squamous cell carcinoma.
Footnotes
Acknowledgement
HSK-Wiesbaden receives trial equipment and clinical support from Fujinon’s research funds.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors declare that there is no conflict of interest.
