Abstract
Background
Endoglin is an endothelial cell membrane receptor essential for angiogenesis and highly expressed on the vasculature of many tumor types, including hepatocellular carcinoma (HCC). TRC105 is a chimeric IgG1 anti-CD105 monoclonal antibody that inhibits angiogenesis and tumor growth by endothelial cell growth inhibition, ADCC and apoptosis, and complements VEGF inhibitors.
Objective
The aim of this phase II study was to evaluate the efficacy of anti-endoglin therapy with TRC105 in patients with advanced HCC, post-sorafenib.
Methods
Patients with HCC and compensated liver function (Childs-Pugh A/B7), ECOG 0/1, were enrolled to a single-arm, phase II study of TRC105 15 mg/kg IV every two weeks. Patients must have progressed on or been intolerant of prior sorafenib. A Simon optimal two-stage design was employed with a 50% four-month PFS target for progression to the second stage. Correlative biomarkers evaluated included DCE-MRI as well as plasma levels of angiogenic biomarkers and soluble CD105.
Results
A total accrual of 27 patients was planned. However, because of lack of efficacy and in accordance with the Simon two-stage design, 11 patients were enrolled. There were no grade 3/4 treatment-related toxicities. Most frequent toxicities were headache (G2; N = 3) and epistaxis (G1; N = 4). One patient had a confirmed partial response by standard RECIST criteria and biologic response on DCE-MRI but the four-month PFS was insufficient to proceed to the second stage of the study.
Introduction
Until recently the role of systemic therapy in the management of hepatocellular carcinoma (HCC) was minimal. This changed with the publication of the landmark Sorafenib Hepatocellular Carcinoma Assessment Randomized Protocol (SHARP) study in 2008, which demonstrated that sorafenib improved overall survival in patients with HCC and preserved liver function compared to placebo, and resulted in sorafenib becoming the standard-of-care option for disease that occurs in the setting of preserved liver function and that is not amenable to surgery, ablation or chemoembolization. 1 While the median survival advantage in the SHARP study was approximately three months, it provided the rationale to focus on the development of anti-angiogenic therapies in HCC. Since then, however, the results of several phase III studies that attempted to build on the initial promise of sorafenib have been disappointing, and sorafenib remains the only approved treatment in HCC.
Endoglin (CD105) is an endothelial cell membrane receptor that is highly expressed on tumor vasculature, including that of HCC.2,3 Endoglin is essential for angiogenesis and its expression is upregulated by hypoxia and vascular endothelial growth factor (VEGF) inhibition.4,5 TRC105 is a chimeric immunoglobulin (Ig)G1 anti-CD105 monoclonal antibody that inhibits angiogenesis (through the competitive inhibition of the activating endoglin ligand bone morphogenetic protein (BMP)) in addition to mediating antibody-dependent cellular cytotoxicity (ADCC).6,7 We conducted an open-label, single-arm phase II study in a post-sorafenib patient population to assess the efficacy of TRC105 in HCC.
Patients and methods
Patients
Eligible patients were at least 18 years old and had histopathological confirmation of HCC by the Laboratory of Pathology of the National Cancer Institute (NCI) prior to entering this study. Other eligibility criteria included: Eastern Cooperative Oncology Group (ECOG) performance status score of 0–2; adequate bone marrow, liver, and renal function; disease not amenable to potentially curative liver transplantation, resection or ablative techniques. In addition, disease must have progressed on or been untreatable by transhepatic arterial chemoembolization (TACE); patients must also have progressed on or been intolerant of prior sorafenib. If liver cirrhosis was present, patients must have had a Child-Pugh A or B (seven points) classification. In addition, patients with cirrhosis were required to have had esophagogastric endoscopy within the previous six months prior to study entry for the assessment of varices. Use of prior systemic therapy or major surgery for cancer was not permitted within four weeks before study treatment. Concomitant treatment of underlying cancer was prohibited. All patients provided written informed consent. This study was approved by the NCI Institutional Review Board, ClinicalTrials.gov identifier: NCT01375569.
Study design
Patients who satisfied the eligibility criteria were enrolled in a single-arm, phase II study of TRC105 at the recommended phase II dose of 15 mg/kg intravenously (IV) every two weeks. Patients were premedicated with dexamethasone, acetaminophen, H2-blockade and an antihistamine prior to initial dosing and dexamethasone was then discontinued in the absence of infusion reactions. Staging was performed by either contrast-enhanced computed tomography (CT) or magnetic resonance scan (MRI) scan every eight weeks. Objective response and progression was evaluated in this study using the international criteria proposed by the Response Evaluation Criteria in Solid Tumors (RECIST) Committee. A Simon optimal two-stage design was employed, aiming to rule out 25% four-month time to progression (TTP) and targeting a 50% four-month TTP. Modified-RECIST criteria proposed by the European Association for Study of Liver (EASL) were also used to evaluate response as a secondary endpoint of this study. 8
Safety
All adverse events and serious adverse events (SAEs) occurring within 30 days of the last dose were reported according to the National Cancer Institute Common Terminology Criteria for Adverse Events v4.0.
Pharmacodynamic studies
Correlative biomarkers of TRC105’s effect were evaluated with radiologic techniques as well as assays performed on peripheral blood or serum. All tests were performed at multiple time points including baseline and during the first and second cycles of treatment. Dynamic contrast enhanced (DCE)-MRI was employed to detect effects on tumor vasculature. In our study imaging with DCE-MRI was performed at three time points (at baseline, during cycle one on day 2 or 3 and during cycle 2 day 1 ± 2 days) and two parameters Ktrans (the transfer constant from blood plasma to extracellular extravascular space (EES) and Kep (reflux rate) were measured at each time point. MRI was performed on a 3 T MR system (Philips Achieva, Best, the Netherlands) with a dedicated receive-only phased array coil.
Plasma biomarker tests were performed on VEGF, placental-derived growth factor (PlGF), basic fibroblast growth factor (bFGF), and vascular endothelial growth factor receptor 1 (sVEGFR1) using assay plates from Meso-Scale Discovery (MSD, Gaithersburg, MD) according to the product manual. The concentrations of the cytokines were determined with recombinant standards and expressed as pg/ml. Blood samples were collected in ethylenediaminetetraacetic acid (ETDA)-containing Vacutainers at pretreatment (baseline), day 15 of the first, day 1 of the second cycles, and the follow-up time. After centrifugation, plasma samples were immediately frozen and stored at −80℃.
The soluble endoglin immunoassay was developed and performed on an MSD assay platform using capture and detection antibodies acquired from R&D Systems (Minneapolis, MN). Recombinant CD105 (R&D Systems) was used to determine the specific concentrations of soluble CD105 in plasma samples. The addition of CD105 antibody TRC105 in vitro completely inhibited the detection of soluble CD105. Briefly, 5 µl of the anti-CD105 capture antibody (R&D Systems, Minneapolis, MN) at 36 µg/ml was coated in each well of MSD 96 standard binding plate overnight at 4℃. The plates were blocked with 3% bovine serum albumin (BSA) for one hour. For detection, 25 µl/well of 10× diluted plasma samples or serially diluted recombinant CD105 standards were added and incubated for two hours, followed by 3× washes with wash buffer (phosphate-buffered saline (PBS) + 0.05% Tween 20). Next, 25 µl/well of 800 ng/ml anti-CD105 detection antibody (R&D Systems) was added and incubate at room temperature (RT) for two hours. After wash, 25 µl/well of 1 ug/ml MSD SULFO-TAG streptavidin (1% BSA, 1 × PBS) was added and further incubated for one hour. For detection, 150 µl/well of 1 × MSD Read Buffer was added and the assay plate was analyzed with an MSD Sector Imager.
Statistical methods
The primary purpose of this study was to evaluate the TTP of TRC105 as a second-line treatment in patients with refractory HCC. TTP was the time from consent until first documented progression of disease. Similar studies conducted in the post-sorafenib setting had resulted in 1.5 and two-month median TTP. The primary endpoint of this study was chosen as the proportion of patients who were without progression of their disease after four months on-study. The sample size and interim stopping rule was determined using a Simon optimal two-stage design. Given historical data, a median two-month time to progression—translating into a four-month progression-free rate of 25%—was considered not promising (p0 = 0.25), while a 50% four-month TTP rate (that is, a median time to progression of four months) was considered promising (p1 = 0.50). With alpha = 0.10 and beta = 0.10, 10 patients were to be accrued to the study in the first stage with continued enrollment to the second stage of 17 patients (27 total patients) only if three or more of the first 10 patients were progression free at four months.
Paired data from angiogenic biomarkers obtained at C1D15 and C2D1 were compared to pretreatment results using a Wilcoxon signed rank test. All p values are two tailed and presented without adjustment for multiple comparisons. Plasma biomarker analysis was performed with GraphPad.
Results
Patient characteristics (Table 1)
Patient characteristics
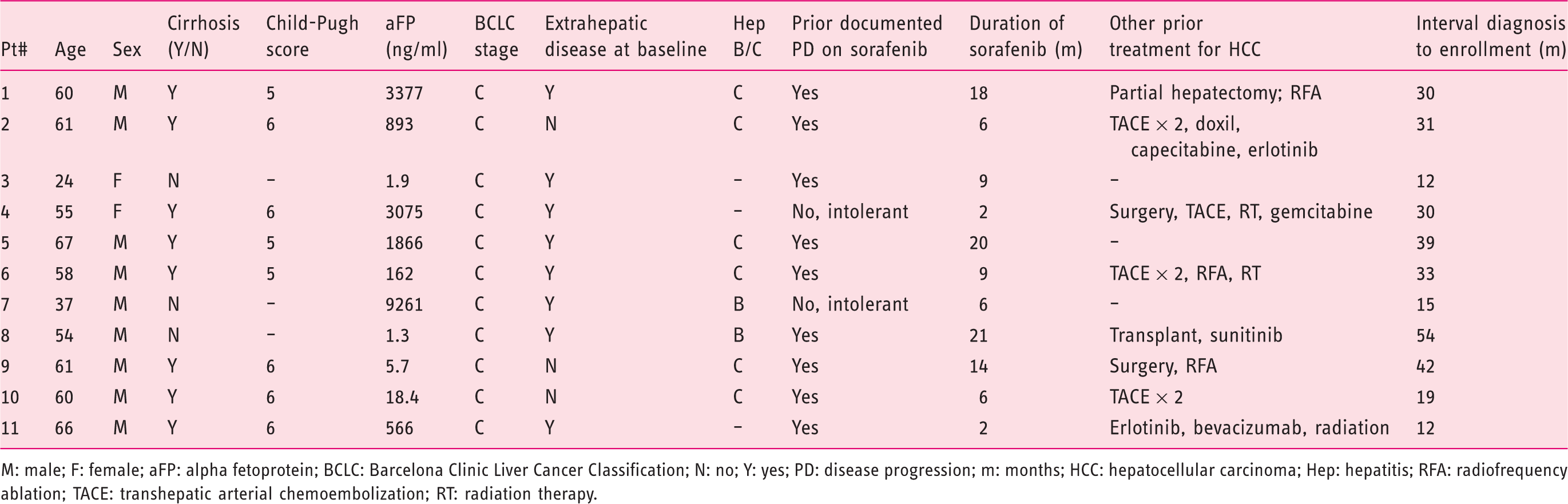
M: male; F: female; aFP: alpha fetoprotein; BCLC: Barcelona Clinic Liver Cancer Classification; N: no; Y: yes; PD: disease progression; m: months; HCC: hepatocellular carcinoma; Hep: hepatitis; RFA: radiofrequency ablation; TACE: transhepatic arterial chemoembolization; RT: radiation therapy.
Safety
Overall treatment was well tolerated. The most frequent toxicities were headache (grade 2; N = 3) and epistaxis (grade 1; N = 4). Two patients developed grade 1 or 2 infusion reactions that did not recur when the infusion time was extended. There were no grade 3 or 4 treatment-related toxicities except one patient with ischemic heart disease who developed a non-Q wave myocardial infarction (grade 3) during an episode of hypertension during the first infusion and was replaced. No other patient discontinued treatment because of toxicity and no dose reductions were required.
Efficacy
In patients with HCC who have failed previous sorafenib, the median time to progression is approximately two months.9,10 Our choice of TTP as study endpoint was based on the recommendation of an expert panel convened by the American Association for the Study of Liver Diseases.
11
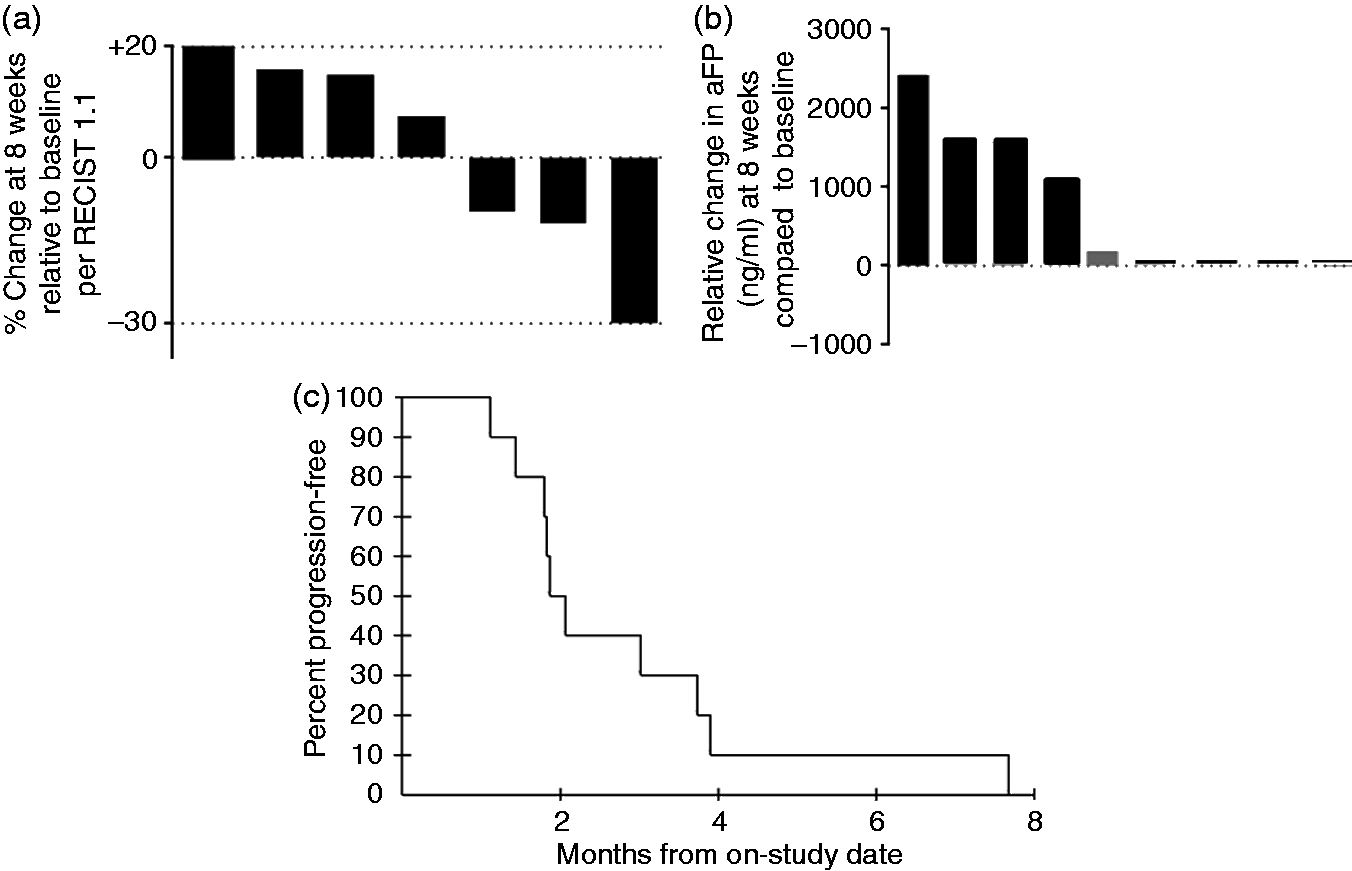
The median time to tumor progression in this study was 12 weeks (range 4–32) (Figure 1(c)). The median overall survival was 6.6 months. One patient demonstrated a confirmed partial response (PR) by RECIST (Figures 2 and 1(a)). This 54-year-old man received an orthotopic liver transplant for HCC and underlying hepatitis B, and subsequently developed pulmonary HCC metastases. He was treated with sorafenib for 21 months followed by sunitinib with a best response of stable disease. He achieved a PR with TRC105 at week 8, subsequently confirmed, and remained in study for 32 weeks before developing clinical progression with bronchial tumor obstruction. No other patient was progression free at 16 weeks by RECIST criteria and when the EASL-modified-RECIST criteria were applied no additional responding patients were identified. As per the Simon two-stage statistical design, this degree of activity was insufficient efficacy to proceed to the second stage of the study.
Waterfall plots for all evaluable patients showing (a) percentage change in tumor dimension at eight weeks (per RECIST 1.1) relative to baseline and (b) relative change in alpha-fetoprotein (ng/ml) at eight weeks compared to baseline; (c) Kaplan-Meier curve for study population. RECIST: Response Evaluation Criteria in Solid Tumors. (a–d) MRI scan at baseline (a, c) and 16 (b, d) weeks for patient 8 showing aggregate 34% reduction in two paracardiac target lesions. MRI: magnetic resonance imaging.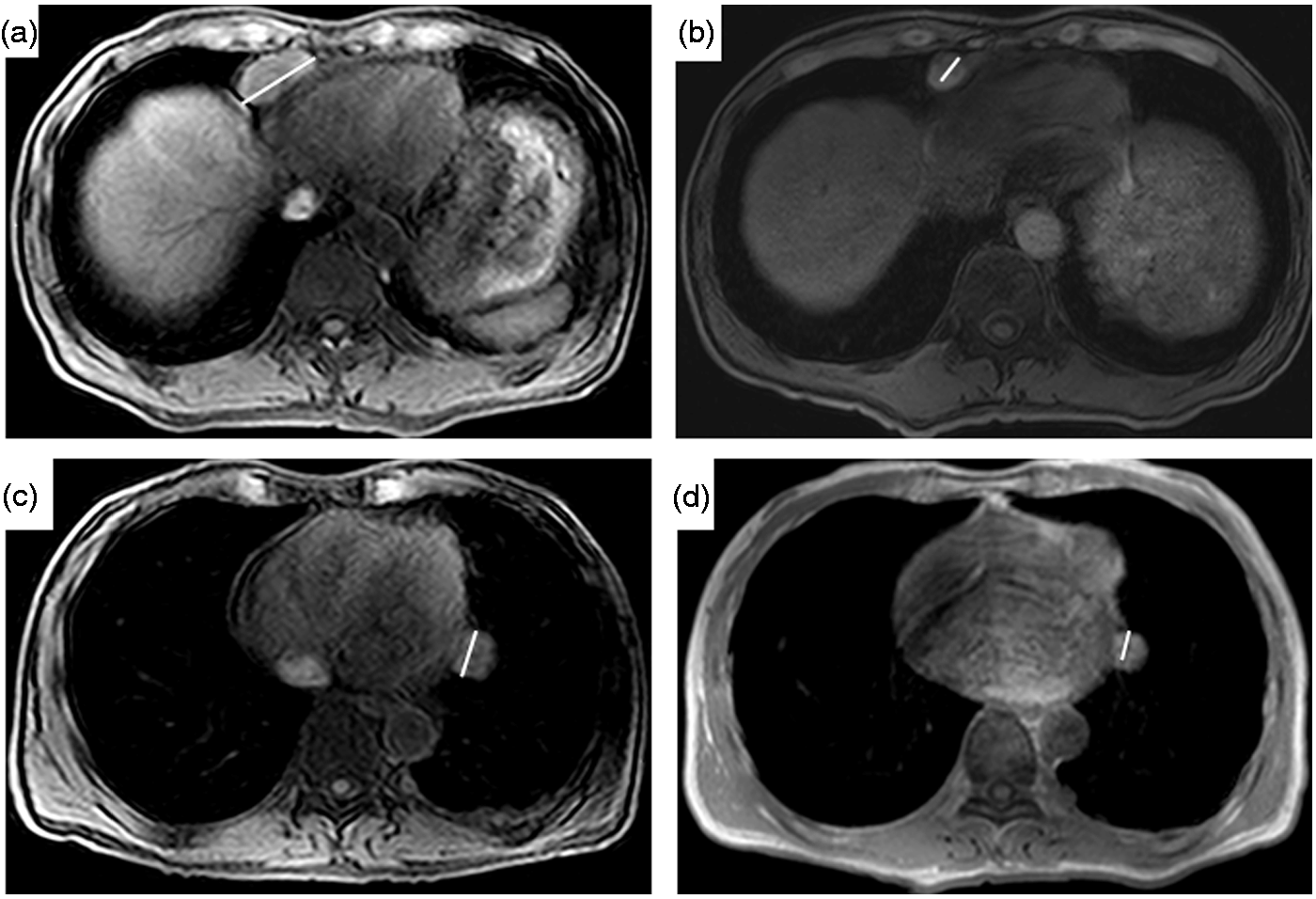
Pharmacodynamics
Levels of CD105 in addition to angiogenic biomarkers for the patient population taken at baseline, Cycle 1 Day 15 (C1D15) and Cycle 2 Day 1 (C2D1)
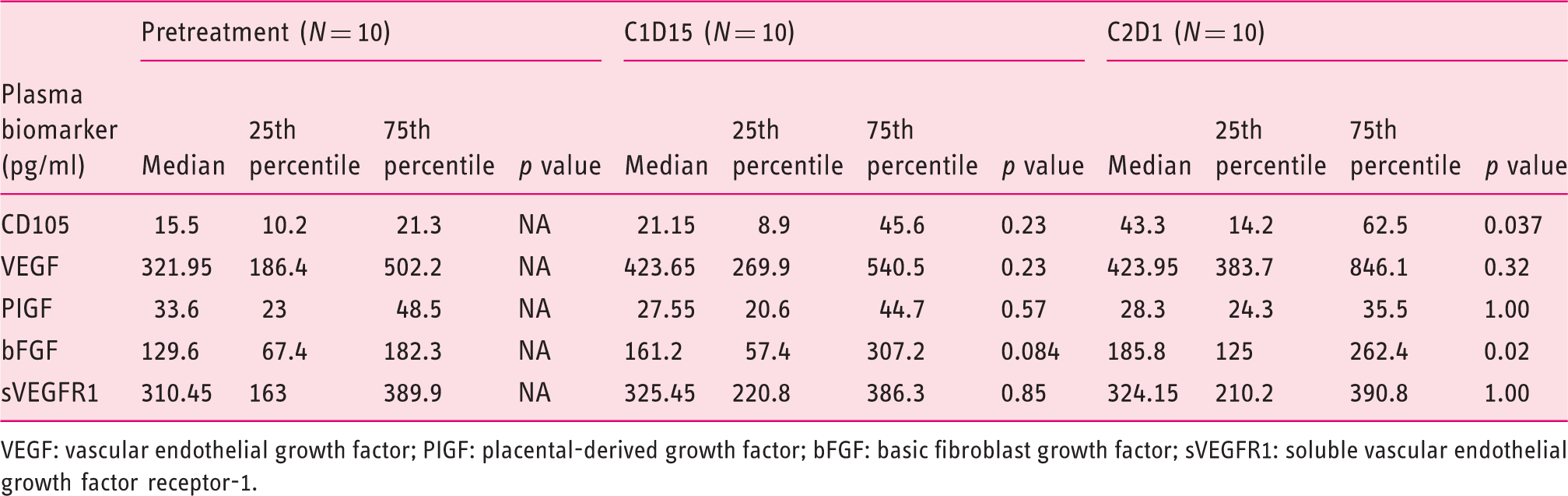
VEGF: vascular endothelial growth factor; PIGF: placental-derived growth factor; bFGF: basic fibroblast growth factor; sVEGFR1: soluble vascular endothelial growth factor receptor-1.
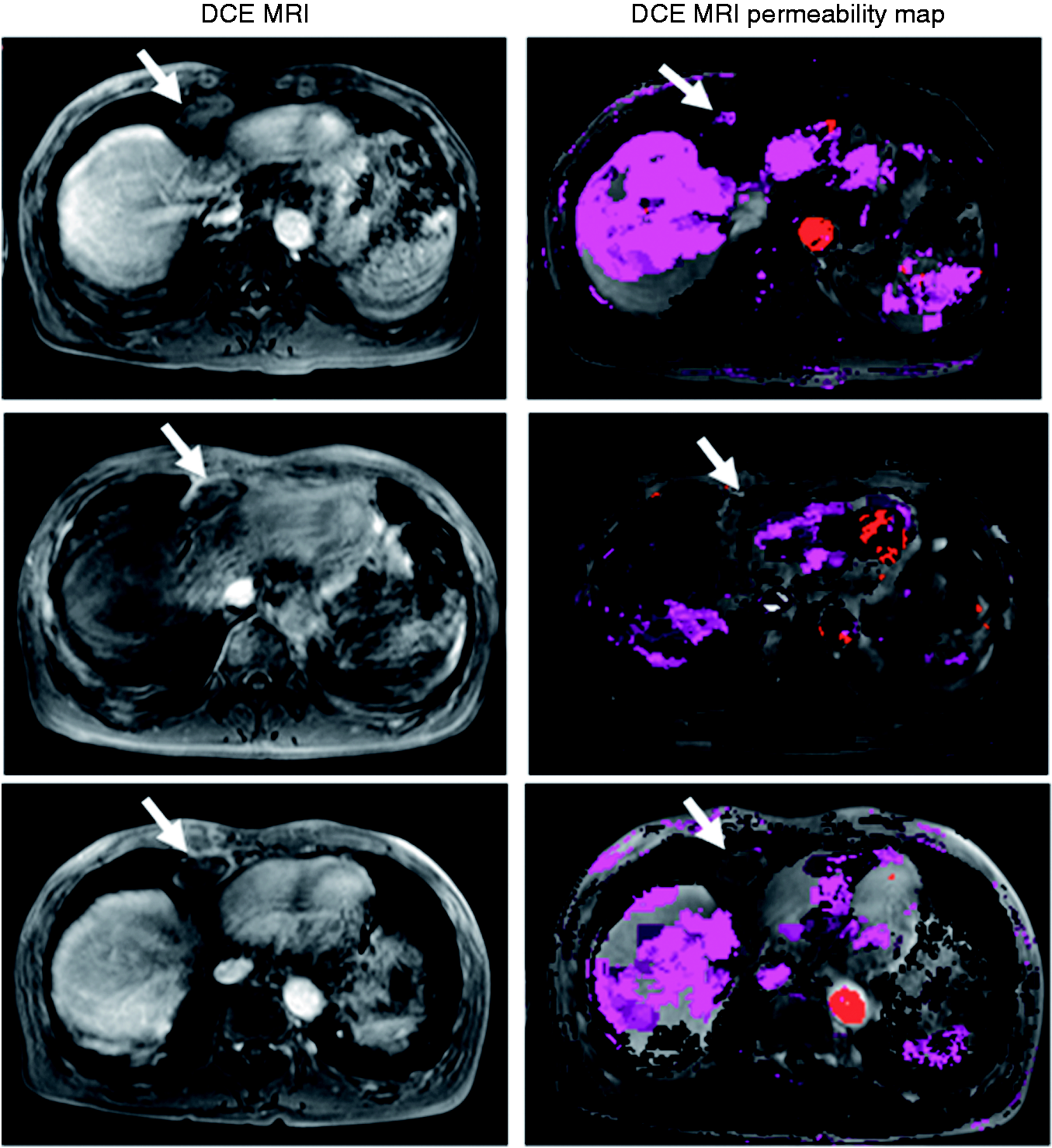
DCE-MRI of patient 8 showing marked reduction in Kep and Ktrans consistent with reduced tumor lesion perfusion. DCE-MRI: dynamic contrast enhanced-magnetic resonance imaging; Kep: reflex rate; Ktrans: transfer constant from blood plasma to extracellular extravascular space.
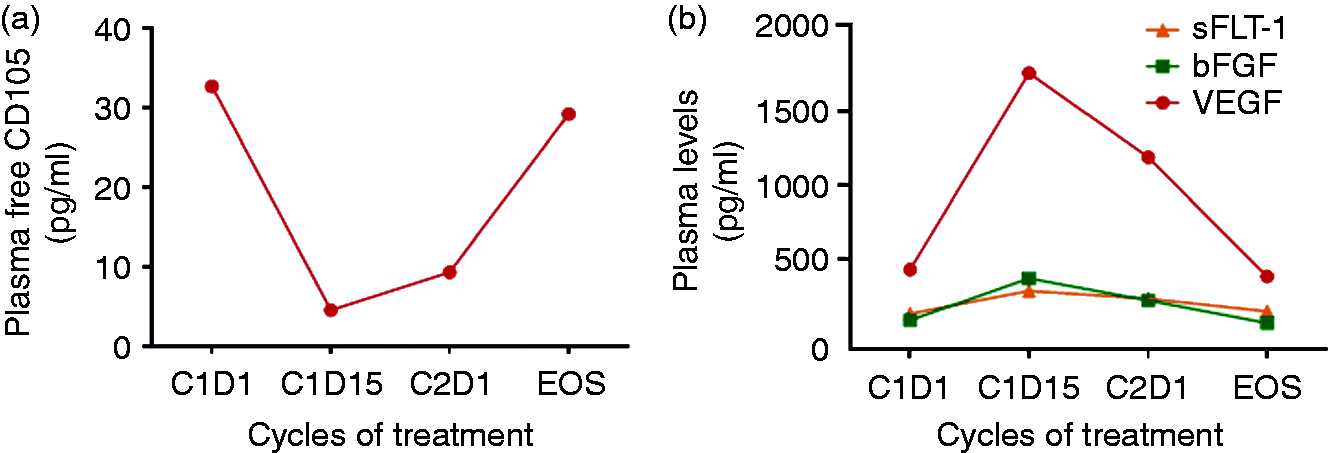
Levels of a) CD105 and b) angiogenic factors in plasma of patient 8 taken at Cycle 1 days 1 and 15, cycle 2 D1 and at the time of progression (f/u). f/u: follow-up.
DCE-MRI has been used extensively with novel anti-angiogenic agents in phase I and II trials to monitor their effect on tumor vasculature, reflecting both tumor perfusion and permeability.12,13 In this study we evaluated the perfusion of the tumors with the analysis of DCE-MRI. Four out of 10 patients (40%) underwent DCE-MRI scans at baseline and at the two follow-up time points. Ktrans and Kep were decreased in three patients, which persisted on the second follow-up DCE-MRI scan. One patient had increase of both Ktrans and Kep and this patient had progression of disease on the second follow-up DCE-MRI (see Table 1). The patient who demonstrated a confirmed PR by RECIST had the greatest decrease of both Ktrans and Kep (Figure 3).
Discussion
Endoglin (CD105) is an attractive target in solid tumor oncology given that it is essential for endothelial cell proliferation and angiogenesis. 5 Mice lacking endoglin die in utero from the absence of angiogenesis. 3 While not specific to malignancy per se, the presence of neo-vasculature in the non-pregnant adult is generally limited to neoplasia, retinopathy and recovery from injury or surgery. CD105 is densely expressed on the proliferating endothelial cells of many tumor types and has been correlated with a poor prognosis. 5 In HCC, CD105 was found to be expressed in 100% of surgically resected specimens (N = 113). 2 In that analysis expression was found to be highly specific for tumor areas in that neither the normal nor adjacent para-carcinomatous tissue stained positively for CD105 by immunohistochemistry. HCC has been considered responsive to angiogenic treatment given the ineffectiveness of conventional cytotoxic chemotherapy and the fact that the sole proven treatment for advanced disease—sorafenib—has anti-angiogenesis as its putative main mode of action. 14 This assumption has been challenged in recent times, however, following the failure of other agents (e.g. ramicurumab and brivanib) whose mode of action is either wholly or partially related to anti-angiogenesis.15–18
We did not see robust single-agent activity in this phase II study of anti-endoglin therapy in a post-sorafenib HCC population needed to proceed to the second-stage per the Simon two-stage design. Only one of the patients met the prespecified endpoint of being progression free at four months. Interestingly, this patient was also the only patient both to achieve a confirmed PR by RECIST criteria and who exhibited DCE-MRI findings consistent with a strong anti-angiogenic effect. This particular patient was status post-liver transplantation and was taking everolimus as an immunosuppressant, a medication shown to have anti-angiogenic as well as other properties and that may have contributed to his response. 19 It was also noteworthy that this patient was the only participant who had a decrease in soluble-free CD105 levels, implying continuous serum levels of TRC105, which inhibited detection of soluble CD105 in the immunoassay. For the population as a whole, median free CD105 levels were increased after completion of the first cycle compared to baseline. This finding is consistent with that reported by Liu et al. in a larger study evaluating different doses of TRC105 ranging from 0.3 to 15 mg/kg every two weeks as well as some patients receiving 10 and 15 mg/kg weekly. In that study soluble endoglin levels increased when TRC105 was administrated at doses ≥3 mg/kg every two weeks. 6 These investigators—recently designated as a Molecular Reference Laboratory for the Alliance cooperative group (formerly CALGB)—went on to replicate this finding in vitro using human umbilical vein endothelial cells (HUVEC) as a model system. They concluded that the increase in soluble CD105 levels following TRC105 treatment may be due to several factors, including prolonged stabilization of soluble endoglin due to TRC105 binding or increased shedding of soluble endoglin induced by TRC105 binding at the cell membrane. Hawinkels et al. have shown that endoglin shedding was mediated by matrix metalloproteinase (MMP)-14 and resulted in vitro in reduced spontaneous and VEGF-induced endothelial sprouting. 20
The clinical and correlative data in our phase II study suggest that TRC105 therapy will not be effective as a single agent in the present schedule in HCC. Only one patient achieved an objective response and was progression free at four months. The patient population was small and heterogeneous, including one case of fibrolammellar variant 21 in addition to the patient referred to above who was post-transplant. However, the differential effects of TRC 105 on specific angiogenic biomarkers compared to anti-VEGF agents suggest there is scope and rationale for combined approaches. VEGF pathway blockade—either by specific inhibition with antibody or as a result of multikinase inhibition—causes intratumoral hypoxia, which in turn leads to upregulation of HIF-1a and the resultant transcription of many pro-angiogenic genes. 22 It was noteworthy that our responding patient experienced an elevation in VEGF levels. There appears to be a close interplay between CD105 and VEGF levels. 6 Endoglin expression is upregulated in response to hypoxia induced by anti-angiogenic, or—more specifically—anti-VEGF agents. 6 In the above-mentioned dose escalation study of TRC105 Liu et al. reported biomarker data in 32 patients in which VEGF family members (VEGF-D, PlGF and soluble VEGF-R1) were found to decrease (or trend down) in a majority of patients. 6
It has been reported that following bevacizumab levels of PlGF and VEGF-D increase—the latter of which has been shown to be predictive of benefit—while the converse has been seen with TRC105.23,24 This all reflects differing points of impact of bevacizumab and TRC105 on the angiogenic process and suggests potentially complementary activity between anti-endoglin treatments and other anti-VEGF strategies. In vitro studies have shown that, as single agents, both TRC105 and bevacizumab efficiently blocked HUVEC tube formation, and the combination of both agents achieved even greater levels of inhibition. 25 Treatment in our study was well tolerated, suggesting that there is scope for combination approaches. Combination with bevacizumab has been recently reported to be well tolerated in patients with advanced solid tumors. 26 We are currently evaluating TRC105 in combination with sorafenib in an untreated HCC population. 27 One slight concern with combining anti-angiogenic agents is the bleeding risk, especially in a prone, generally thrombocytopenic HCC population. 28 In our study, we mandated an upper endoscopy to exclude high-risk esophageal or gastric varices, an important consideration in this patient group.
In summary, despite intriguing partial response by RECIST and a correlative response to TRC105, we did not see sufficient efficacy results in this single-arm, phase II study of anti-endoglin therapy in a post-sorafenib HCC population to warrant continued single-agent development. Preclinical and correlative biomarker data suggest that the role of anti-endoglin therapy will be in combination with another anti-angiogenic strategy.
Footnotes
Funding
This work was supported by the Intramural Research Program of the National Institutes of Health (NIH), NCI, Center for Cancer Research.
Conflict of interest
None declared.
Acknowledgments
The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the United States government.
