Abstract
Background
The Barcelona Clinic Liver Cancer (BCLC) classification of hepatocellular carcinoma (HCC) has proved useful in the management of HCC patients. However, BCLC-recommended first-line treatment is not always applicable in clinical practice.
Objective
We performed a multicentre retrospective analysis of reasons for deviation from first-line treatment in 2008–2012.
Methods
One to three-year survival data were analysed using Kaplan-Meier method.
Results
A total of 407 consecutive HCC patients (66.6 ± 3 years, 83% male) with cirrhosis were included. Tumours were detected during surveillance in 53% of patients, grouped as Child-Pugh A (67%), B (25%) and C (8%); and BCLC A (including stage 0, 44%), B (26%), C (15%) and D (15%). In 31% of patients, first-line treatment was not feasible (51% in early stages) due to: technical reasons (74%); patient non-conformity (20%); medical decision (3%); and disease progression (3%). One to three-year survival of patients not receiving the recommended first-line treatment was similar to that of patients treated according to BCLC recommendations (log-rank,
Conclusion
In real-life practice one-third of HCC patients could not receive first-line BCLC treatment. In our cohort of patients, similar short and medium-term survival was observed. Long-term prospective studies are required to determine the best alternative treatment option when BCLC first-line treatment is not feasible.
Introduction
Hepatocellular carcinoma (HCC) currently represents a major complication in cirrhotic patients worldwide. 1 In the last two decades survival has improved due to increased surveillance and early HCC detection which allows potentially curative treatments. 2 In addition, the management of HCC has been protocolised and scientific societies have elaborated guidelines defining surveillance, diagnosis and therapeutic strategies.3,4 These guidelines recommend the Barcelona Clinic Liver Cancer (BCLC) staging system since it reliably predicts prognosis and outlines the best evidence-based treatment available. This staging system has been validated in different studies.5–7 It includes prognostic variables mainly related to tumour status, liver function and health performance status. All of these variables permit treatment allocation following a treatment regime. However, first-line treatment options are not always applied in real-life clinical practice for various reasons, including patient characteristics, tumour-dependent factors or available medical resources. In fact, current clinical practice guidelines specify that such recommendations have to be adapted when dealing with local suboptimal scenarios. Non-application rates range from 10–75% depending on the type of treatment recommended. 8 Scarce data is available regarding the overall proportions of patients that do not receive the BCLC-recommended treatment,9,10 and the resulting impact on survival has not been specifically addressed. The aims of the present study were to evaluate reasons for divergence from BCLC first-line treatment, to assess survival in HCC patients not receiving such treatment, and to determine whether stage migration treatment is useful in terms of survival.
Patients and methods
The study included consecutive HCC patients attended by a gastroenterologist and diagnosed at one of the three participating institutions: a non-referral centre (Complejo Hospitalario Universitario de Pontevedra), a referral centre (Hospital Universitario de Canarias) and a referral centre with a liver transplant program (Hospital Universitario Marqués de Valdecilla), between January 2008–September 2012 and followed until April 2014. The study was performed in accordance with the ethical guidelines of the 1975 Declaration of Helsinki, and approved by the Clinical Research Ethics Committees of the three hospitals.
We excluded non-cirrhotic patients and clinical trial participants. Liver cirrhosis was diagnosed on the basis of clinical signs, biopsy and/or ultrasound characteristics.
At diagnosis, patients were individually assessed by a multidisciplinary team in each centre and, whenever feasible, assigned to receive one of the following first-line treatment options in accordance with HCC therapeutic guidelines:4,11–13 resection, liver transplant, radiofrequency ablation or percutaneous phenolisation (BCLC A), transarterial chemoembolisation (TACE) (BCLC B) or sorafenib treatment (BCLC C). In selected cases with large HCC or TACE contraindications, radioembolisation was performed in accordance with patient or physician decision. Patients included on the waiting list for liver transplant were treated with locoregional procedures as bridging therapy.
All the guideline-recommended treatments were available in the three participating centres, although liver transplant candidates had to be referred to another centre for treatment (except for Hospital Universitario Marqués de Valdecilla). HCC was treated by hepatic lobectomy or segmentectomy. Liver transplantation was assigned following the Milan criteria 14 when the patient met the conditions of abstinence from alcohol, age under 70 years and absence of severe comorbidity. Percutaneous local ablation under real-time sonographic guidance included radiofrequency therapy (StarBurst XL probe, RITA medical systems Inc., Mountain View, California, USA) and ethanol injection (1–10 ml, 99.5%), according to location, number (≤3 nodules) or size of the tumour (≤5 cm). Additional sessions were performed until disease control, complications or absence of response were observed. TACE was performed after identifying the tumour-feeding artery, either with lipiodol (Laboratoire Guerbet, Aulnay-Sous-Bois, France) and gelatin sponge particles (Gelfoam, Upjohn, Kalamazoo, Michigan, USA) or with drug-eluting beads (300–500 microns, DC Beads, Biocompatibles UK Ltd, UK) using doxorubicin in both cases. Retreatment was considered every 3–6 months when tumour activity was observed except in cases with symptomatic progression or a history of serious adverse events, extrahepatic spread or contraindication. Patients were grouped for analysis according to application or non-application of BCLC-recommended first-line treatment regardless of whether that involved a curative option or not. Reasons for not applying the BCLC first-line treatment were recorded as: patient non-conformity (the patient refused the proposed treatment option and demanded an alternative treatment), disease progression while on the liver transplant waiting list, medical decision and technical issues including comorbidity (morbid obesity, cardiopulmonary disease, cerebrovascular disease, second active tumour, severe thrombopenia), tumour location (subcapsular, proximity to relevant organs), radiological non-visibility, poor portal blood flow, presence of refractory ascites and large nodule size or multicentricity.
After treatment, all patients underwent regular follow-up as recommended in the guidelines until death from any cause,4,11–13 with either computed tomography or magnetic resonance imaging. When recurrence occurred the patient was re-assessed in terms of BCLC staging, and treated accordingly. In cases that were lost to follow-up, the medical record was used to check for date of death.
Statistical analysis
Follow-up length and survival is expressed as median (range) from time of diagnosis. One, two and three-year survival rates were determined using Kaplan–Meier survival curves, and compared using the log-rank test. Quantitative variables are expressed as median and range, and categorical variables as absolute values and proportions. Differences between subgroups were evaluated using chi-squared test or Fisher’s exact test for categorical variables, and by Student’s
For the analysis of survival of patients grouped according to BCLC first-line treatment application or not, those classified as D (Child-Pugh C not amenable to liver transplant and performance status > 2,
All statistical analyses were performed using SPSS package version 20 (SPSS Inc., Chicago, Illinois, USA).
Results
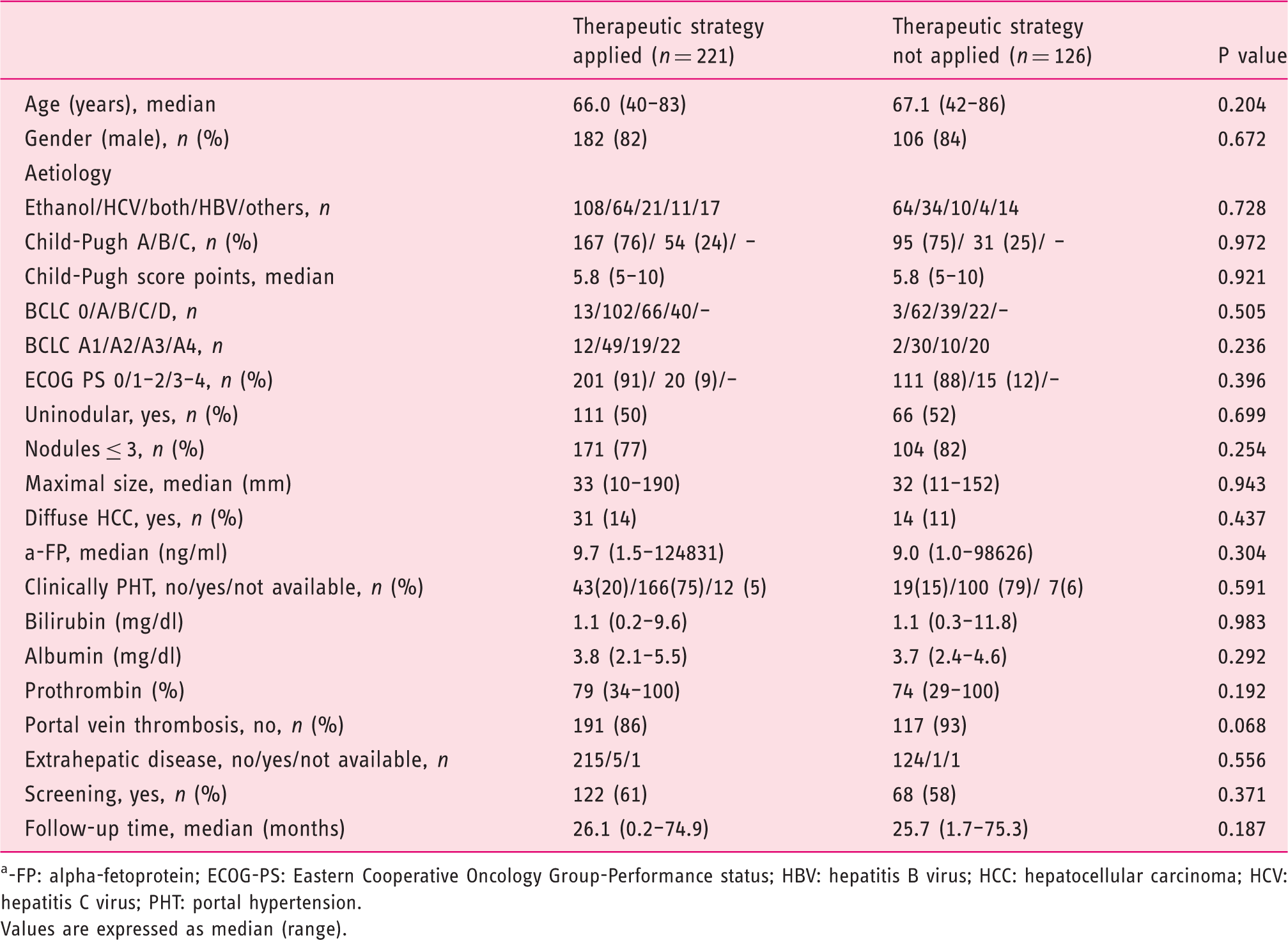
Patient characteristics
Characteristics of patients according to participating centres
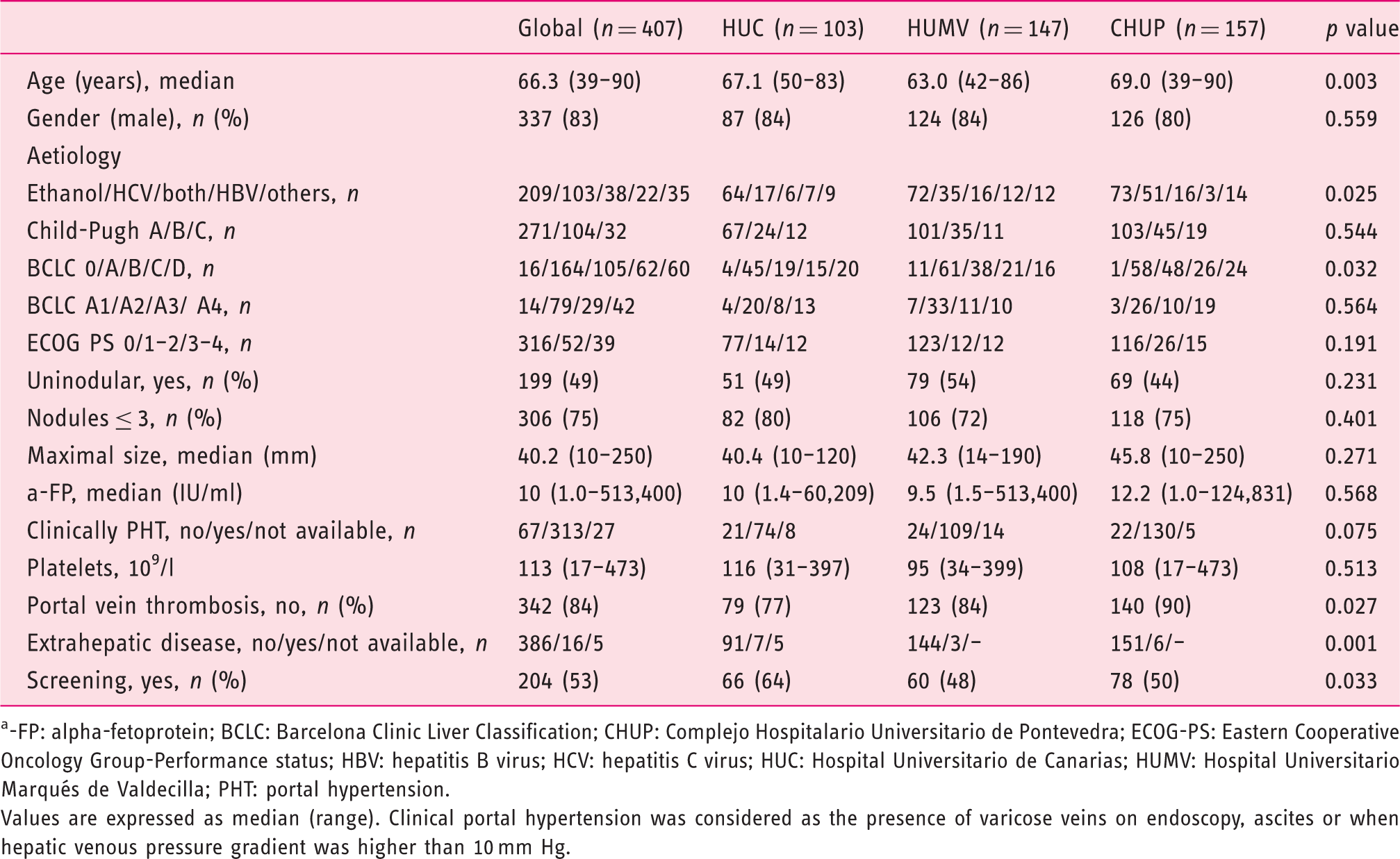
-FP: alpha-fetoprotein; BCLC: Barcelona Clinic Liver Classification; CHUP: Complejo Hospitalario Universitario de Pontevedra; ECOG-PS: Eastern Cooperative Oncology Group-Performance status; HBV: hepatitis B virus; HCV: hepatitis C virus; HUC: Hospital Universitario de Canarias; HUMV: Hospital Universitario Marqués de Valdecilla; PHT: portal hypertension.
Values are expressed as median (range). Clinical portal hypertension was considered as the presence of varicose veins on endoscopy, ascites or when hepatic venous pressure gradient was higher than 10 mm Hg.
Application of BCLC treatment strategy
In 30.9% of the study population the BCLC first-line treatment was not feasible. Reasons for divergence are shown in Figure 1. Supplementary Material, Figure 1, shows the clinical characteristics of patients not receiving BCLC first-line treatment according to tumour stage. Most divergence occurred in patients with early stage disease (51.5% stage A, 31% B, 17.5% C). Fifty-two patients (42.3%) were changed from a curative treatment to a non-curative treatment, predominantly TACE. Patients treated in accordance with the BCLC strategy received a higher proportion of curative treatments than those who did not (51.8% vs 21.0%; Reasons for non-application of Barcelona Clinic Liver Cancer (BCLC) therapeutic strategy, type of technical difficulty and final treatment applied. (a) Causes of divergence from the BCLC classification and (b) type of technical difficulty. (c) Final treatment applied for each BCLC stage. One patient on the liver transplant (LT) waiting list died before receiving any treatment.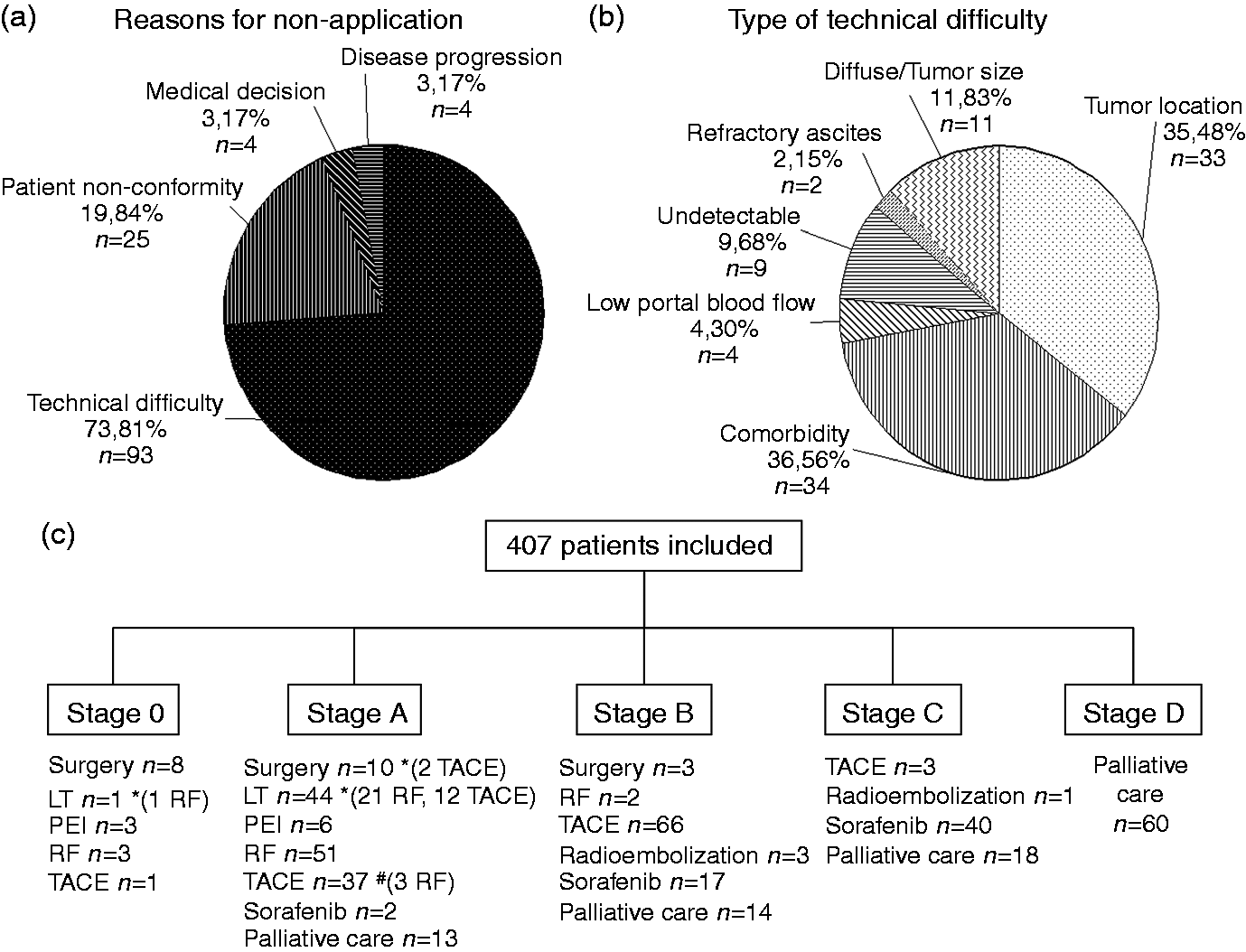
Initial treatment finally assigned for patients with HCC was as follows: surgical resection 4.9%, liver transplant 11.1%, percutaneous treatment 16.0%, TACE 26.4%, sorafenib 14.5%, palliative management 25.8% and other (radioembolisation) 1.3%. One patient died while awaiting liver transplantation. Figure 1(c) shows treatments according to BCLC stage. Thirteen patients with early-stage disease did not receive specific treatment (palliative care when symptomatic); five refused any treatment from the beginning and the other eight patients due to technical difficulties and final non-conformity.
Impact on survival
At the end of follow-up, median survival of the 407 patients was 21.8 months (range, 0.2–75.3). Cumulative one, two and three-year survival rates of the whole cohort were 65.9%, 47.7%, and 37.8%, after a median follow-up of 21.8 months (95% CI: 18.3–26.3). Overall survival according to BCLC stage and treatment received are shown in Figure 2(a) and 2(b), respectively.
Cumulative survival of patients with hepatocellular carcinoma. (a) Survival according to Barcelona Clinic Liver Cancer (BCLC) classification and (b) according to type of treatment received.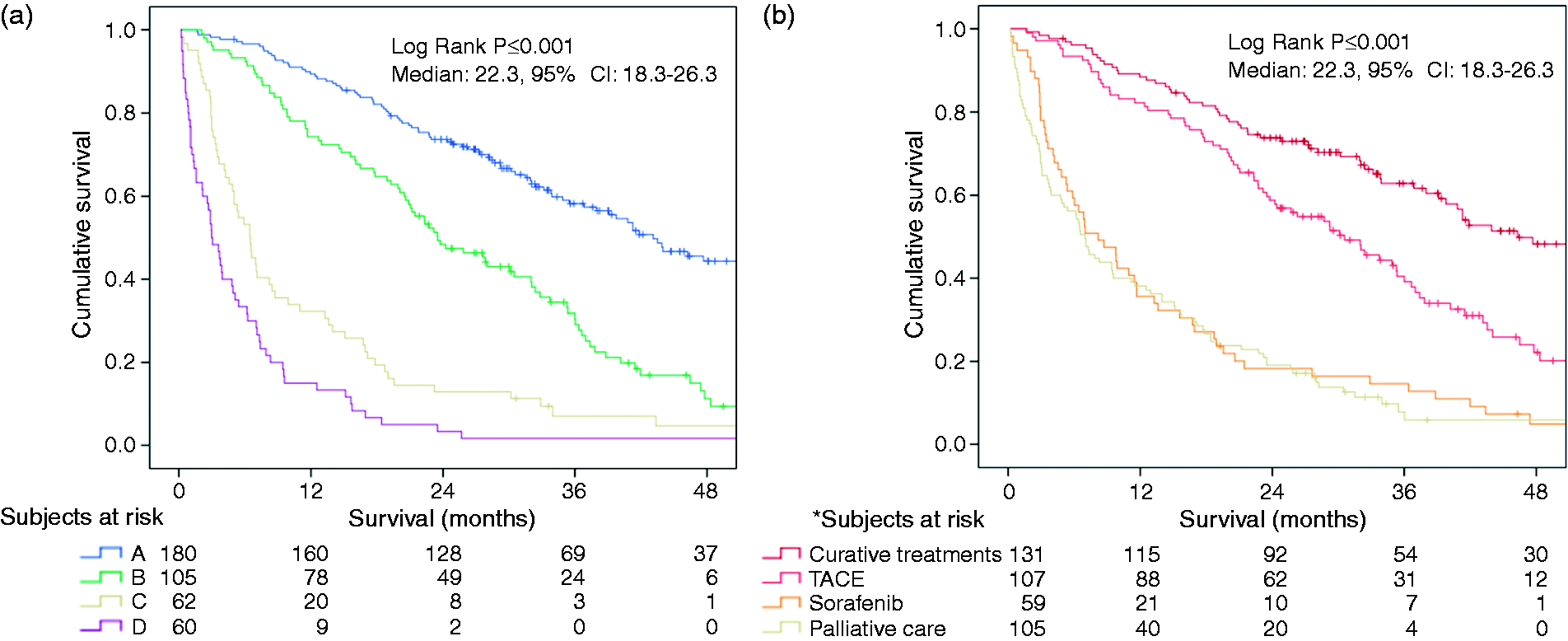
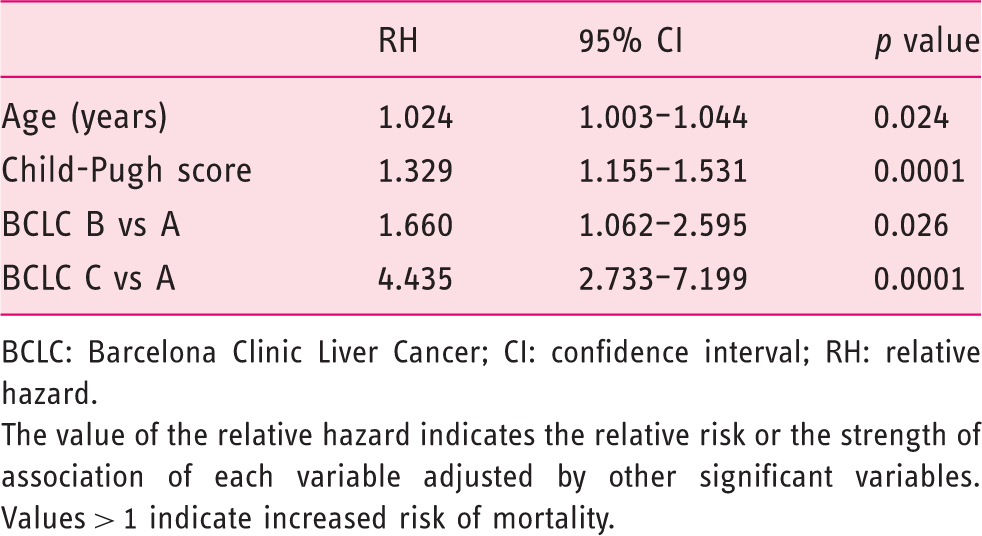
On comparing survival according to application of BCLC first-line treatment or not, similar results were found between groups (log-rank Survival of patients grouped according to application or non-application of Barcelona Clinic Liver Cancer (BCLC) therapeutic strategy. (a) Global survival curve of patients according to application or non-application of BCLC. Survival in stage BCLC A (b), B (c) and C (b). Clinical features of patients grouped according to application or non-application of Barcelona Clinic Liver Cancer (BCLC) therapeutic strategy -FP: alpha-fetoprotein; ECOG-PS: Eastern Cooperative Oncology Group-Performance status; HBV: hepatitis B virus; HCC: hepatocellular carcinoma; HCV: hepatitis C virus; PHT: portal hypertension. Values are expressed as median (range). Factors independently associated with survival BCLC: Barcelona Clinic Liver Cancer; CI: confidence interval; RH: relative hazard. The value of the relative hazard indicates the relative risk or the strength of association of each variable adjusted by other significant variables. Values > 1 indicate increased risk of mortality.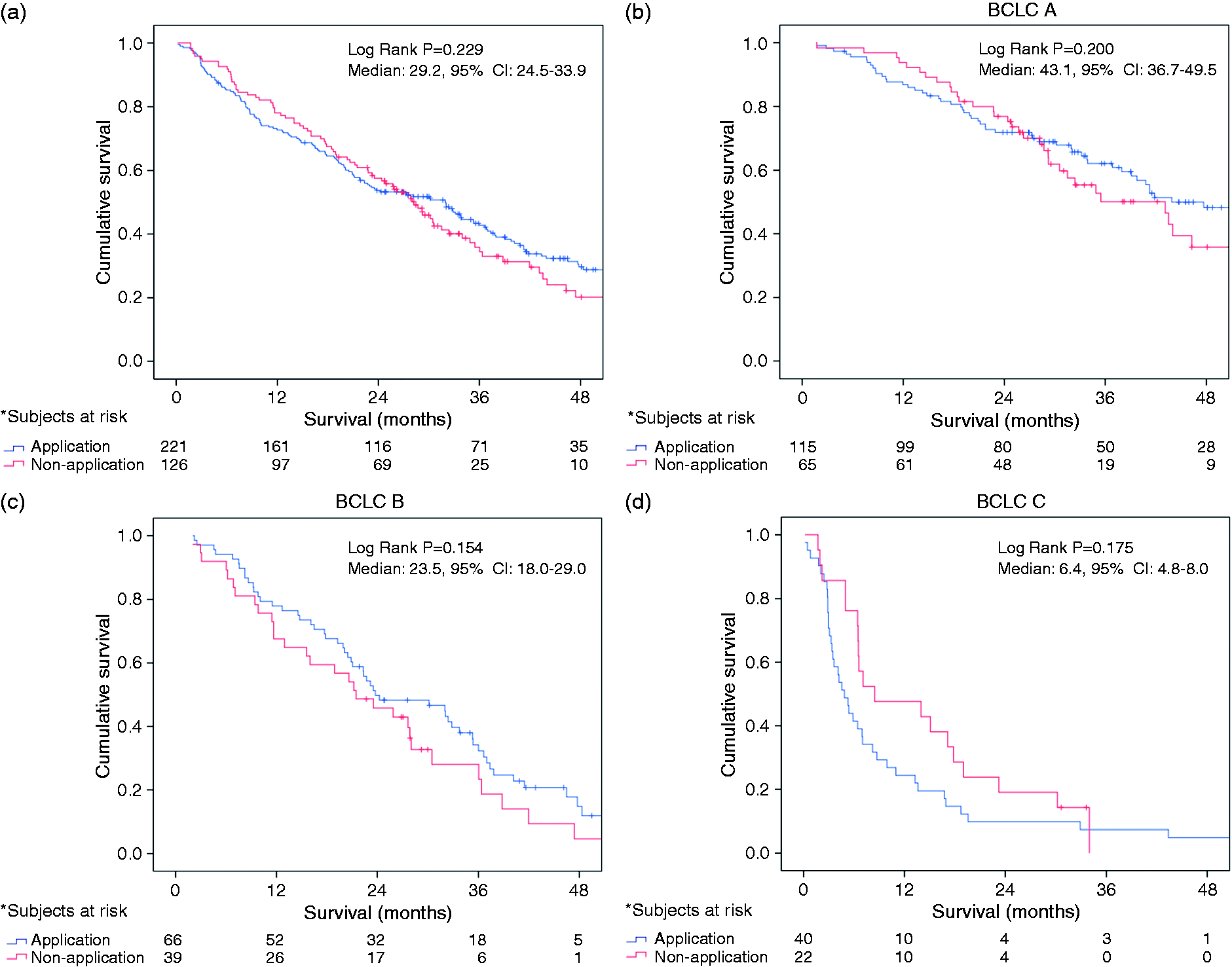
Patients with stage BCLC A treated with TACE showed similar survival compared with those that received radiofrequency ablation, ethanol injection, liver resection or transplant after a median follow-up of 32.0 months (range, 1.2–72.9) (log-rank Transarterial chemoembolisation (TACE) and survival in patients with Barcelona Clinic Liver Cancer (BCLC) stage A. (a) Survival of patients with BCLC stage A hepatocellular carcinoma treated with TACE or curative treatment (radiofrequency ablation, percutaneous ethanol injection, surgery resection and liver transplant); and (b) excluding liver transplant patients. CI: confidence interval.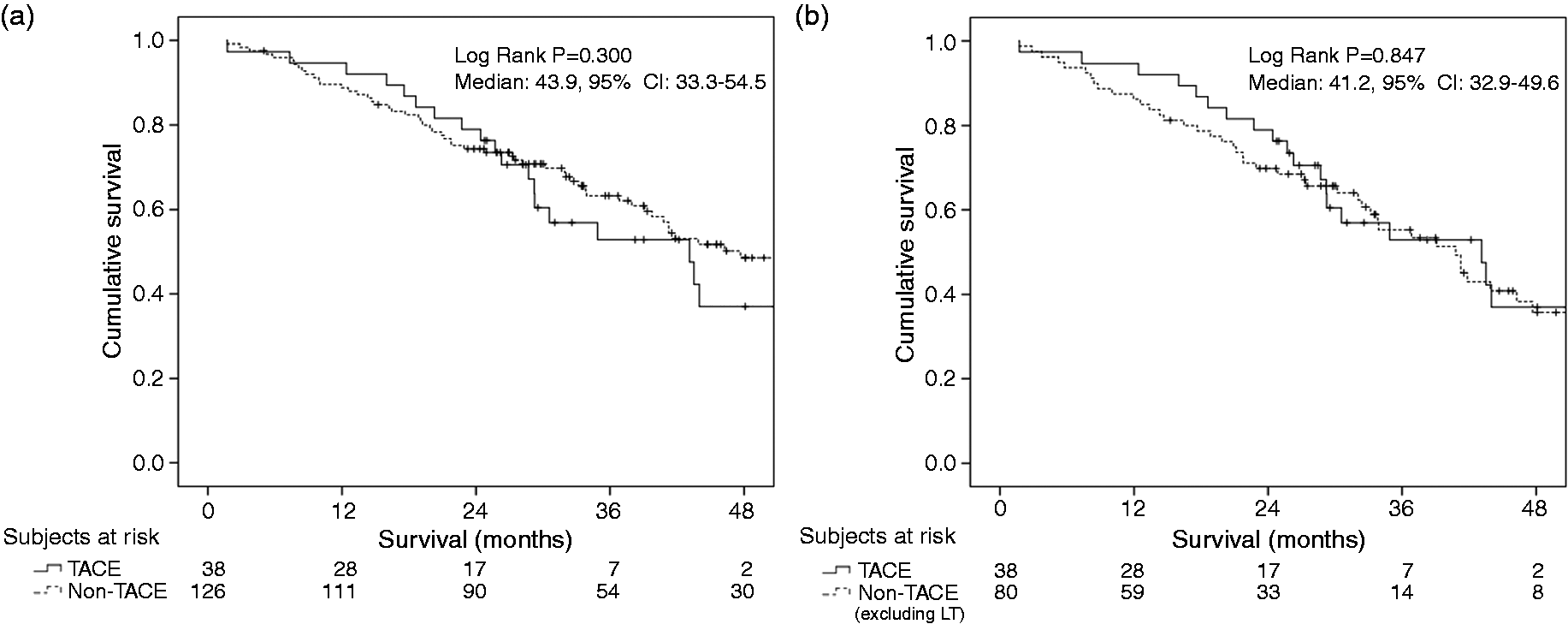
Discussion
The current study showed that in real-life clinical practice up to one third of patients with HCC did not undergo the first-choice treatment recommended by clinical guidelines as a consequence of technical difficulties due to tumour location or patient comorbidity. Our analysis provides further evidence that the BCLC staging system and type of treatment received has good predictive capacity for survival. However, short and medium-term survival rates in our cohort of patients did not differ between patients receiving or not receiving the first-line treatment option.
Most cases of divergence from BCLC first-line treatment involved locoregional treatments. The proportion of HCC patients potentially treatable by locoregional approaches is increasing as screening measures have been applied and therefore the proportion of non-application could dramatically increase. Percutaneous ethanol injection and radiofrequency ablation are effective in controlling tumour progression and improving prognosis.15,16 Nevertheless, their use is subject to technical limitations. First, unfavourable tumour locations such as those adjacent to the gastrointestinal tract, diaphragm or gallbladder carry a high risk of complications and this may discourage use of a percutaneous approach.8,17–19 The rate of first-line treatment non-application due to high-risk HCC location ranges from 10 to 25%, which is not irrelevant taking into account that most of these tumours are still at curative stages of development. In addition, recurrence is present in two-thirds of treated cases at five years, so this number could be even higher as new evaluations and treatments are considered during follow-up. In certain cases, when locoregional treatment is firmly considered, combinations of ablative treatments are used to minimise adverse effects resulting in improved outcomes. 20 In addition to the risk of complications, radiofrequency ablation is often not feasible in HCC with nearby vessels that hinder or impede tumour exposure to treatment. 21 On the other hand, highly experienced centres may not impose restrictions based on difficult tumour locations. In this regard no significant differences have been reported in complications or in local tumour progression rate between the percutaneous approach in HCC of difficult locations or not.22,23 Nevertheless, special care was taken to avoid difficult location-related complications. In addition, a higher number of procedures was performed in difficult-to-treat nodules and sessions of longer duration were required. Despite the existence of these resource-consuming techniques, our results suggest that the use of second-line treatment options, in extremely difficult cases ineligible for first-line treatment, may provide satisfactory results.
In addition to technical difficulties, comorbidity may also account for some non-application. HCC is more likely to develop in obese patients 24 and severe obesity itself may preclude a percutaneous approach or therapies requiring anaesthesia. Diabetes, which is usually also present in metabolic syndrome and associated to non-alcoholic fatty liver disease, is a risk factor for HCC, and vasculopathy may also interfere with treatment options. 25 Finally, the increased prevalence of HCC among the elderly may further contribute to first-line non-application as comorbidity increases.1,26
Other factors potentially involved in non-application, although with less impact in our setting, are patient non-acceptance, physician bias regarding the perceived ineffectiveness of certain treatments in particular cases, as well as health resources and restrictive local regulations. 27 Undoubtedly, optimal management of HCC should be aimed at improving access to first-line BCLC recommended treatment. Whenever this approach is unfeasible, our results shed light in terms of outcome.
Notably, most deviations from first-line treatment recommendations occurred in patients with early BCLC stage, which is not surprising given the wide choice of therapeutic options for this stage. In this regard, our results also indicate that TACE was associated with satisfactory survival rates in patients not eligible for curative treatments, corroborating recently reported data.19,28–31 Although both lipiodol and bead techniques were performed, our results indicate that TACE may be of value in these cases. Indeed, improved survival using TACE has been attributed to better early and accurate staging and bead chemoembolisation.32–35 However, potential obstacles to TACE application are non-visibility, size of the nodule, and poor portal blood flow. In any case, individualised evaluation of HCC treatment is mandatory after a first-line option has been ruled out.
Finally, the initially striking finding that 30% of HCC patients did not receive BCLC recommended first-line treatment should be viewed in the context of worldwide data, or at least those from Western countries, showing underutilisation of recommended HCC treatments.36–39 Several studies have evaluated the use of curative and palliative treatments in different types of HCC populations, finding that only one third of patients actually received treatment. More recently, a European study, specifically designed to assess adherence to current guidelines, reported improper assignment to different therapeutic choices in 40% of patients, particularly those with early stage HCC. 9 The observed results, once again, indicate the need for multidisciplinary expert evaluation 40 and a flexible treatment choice for HCC patients, which may often diverge from guideline recommendations. 41 In this regard, by assessing the causes of divergence from BCLC recommendations, our study is useful to establish strategies to increase appropriate HCC treatment.
Our findings must be interpreted within the limitations of the study, mainly the retrospective nature of the analysis. Despite similar cumulative survival rates, median follow-up for the entire cohort was approximately two years and therefore survival rates over longer periods (e.g. beyond five years) are not available. However, application of first-line therapies, especially transplantation, may have a favourable impact on long-term survival (compared with non-transplanted patients with higher non-HCC cirrhotic-related mortality). However, this large study population very well represents real-life clinical practice with a full spectrum of patients presenting HCC at various evolutionary stages. In addition, our findings are based on a multicentre experience. The three participating centres followed the European Association for the Study of the Liver HCC guidelines and clinical staging of HCC was well defined according to the BCLC strategy. Besides, all patients were enrolled within a short period, just after the latest guidelines update and treated homogenously according to the current state of the art. Secondly, alternative treatment options are less frequently applied as stages advance, so the statistical power of our results for patients with BCLC B and C is limited due to the small sample size.
In conclusion, one third of patients with HCC did not receive the first-line treatment recommended in current guidelines. The main reasons for deviation were technical difficulties due to tumour location and patient comorbidity. Short and medium-term survival in these patients was not affected in our cohort of patients, probably due to the successful use of alternative approaches, especially TACE in selected patients not eligible for curative treatments. Prospective studies with long term follow-up are required to determine the best second-line or alternative treatment option when BCLC first-line treatment is not feasible.
Footnotes
Funding
M. Hernández-Guerra is the recipient of a research grant from Instituto de Salud Carlos III (FIS1914/10) and Programa de Intensificación de Actividad Investigadora (INT10/173).
Conflict of interest
The authors declare they have no conflict of interest.
Acknowledgments
The authors wish to thank the members of the multidisciplinary teams involved in HCC management in each centre and the ‘Fundación para la Investigación Biomédica Rafael y Clavijo’ for editorial support and Alejandro Jimenez for statistical analysis support.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
