Abstract
Background
The Milan criteria are recommended to select hepatocellular carcinoma (HCC) patients for liver transplantation (LT). The utility of other selection criteria, such as the alpha-fetoprotein-adjusted-to-HCC-size (AFP-UTS) criteria, is still unclear.
Objective
We investigated, in HCC patients who underwent LT, the survival and the recurrence after LT according to AFP-UTS and Milan criteria, the impact of early recurrence and the correlation between radiological and pathological staging.
Methods
Adult HCC patients undergoing deceased donor LT at the Medical University of Vienna between 1997 and 2014 were retrospectively analysed.
Results
Among 166 patients included, the number of patients who fulfilled Milan or AFP-UTS criteria was the same (139 [84%] each), although not all of them were the same individuals; 127 patients (77%) fulfilled both Milan and AFP-UTS criteria. Median overall survival of patients within AFP-UTS was 126.9 months vs. 34.2 months outside AFP-UTS (5-year survival rate 71% vs. 43%;
Conclusions
The overall survival of patients within AFP-UTS criteria was favourable with a 5-year survival rate above 70%. Early recurrence is associated with worse survival after LT. The AFP-UTS criteria may be more suitable to exclude patients at high risk of (early) recurrence than Milan criteria.
Introduction
Hepatocellular carcinoma (HCC) is the seventh most common type of malignant tumour, with a steadily growing incidence rate, and is the fourth most frequent cause of cancer-related death. 1 HCC mostly develops in cirrhotic patients and is the most frequent cause of death in this group.2–4 For selected patients, liver transplantation (LT) is the most suitable treatment option as it cures both underlying liver cirrhosis and HCC. 5
As patients need to be on immunosuppression after LT, HCC recurrence usually results in poor prognosis. 6 In particular, early recurrence, defined as recurrence diagnosed within the first 12 months after LT, was a strong predictor for dismal survival after LT. 7 Unfavourable survival rates in the early years of LT for HCC8–11 necessitated the definition and application of stricter criteria for patient selection in order to diminish the recurrence rate by minimizing the risk of vascular invasion and micrometastases at the time of LT. Nowadays, the ‘Milan’ or ‘Mazzaferro’ criteria (1 tumour ≤5 cm or 3 tumours ≤3 cm) 12 are applied by most liver transplant centres in Europe. 13 Consequently, outcome has decisively improved and survival rates similar to those of patients transplanted for non-malignant indications (over 70% at 5 years) have been reported. 12 , 14 As the Milan criteria are very strict and thus exclude a large number of patients from LT, there is an ongoing discussion about broadening the selection criteria for LT in HCC or adapting them by including surrogate parameters for tumour biology.15–19
Yao and colleagues proposed the so-called ‘UCSF criteria’ (1 nodule ≤6.5 cm, or ≤3 nodules ≤4.5 cm and total tumour diameter ≤8 cm) and reported a 5-year survival rate of 75% for patients fulfilling them, but subsequent studies showed lower survival rates. 18 , 20 In 2009, Mazzaferro and his team published the so-called ‘Up To Seven criteria’, which allow LT for patients whose sum of the number of HCC nodules plus the size of the largest nodule does not exceed 7. 15 They also proposed the ‘Metroticket’ – a prognostic model based on tumour number and size according to explant pathology that estimates the probability of 3-year and 5-year survival for a specific patient. 15 Other groups proposed selection criteria that incorporate alpha-1-fetoprotein (AFP), a surrogate marker for tumour biology and strong predictor for recurrence and survival after LT, for example in combination with Milan criteria 21 or total tumour volume. 22 A recent study compared various selection criteria, including criteria that incorporate AFP, by retrospective analysis and found that the outcome would have been very similar to the Milan criteria regardless of which criteria were used. 23
In 2017, the ‘Metroticket’ was updated to allow evidence-based pre-transplant evaluation of post-transplant outcome by means of radiological assessment of tumour number and size as well as AFP. 24 Based on the ‘Metroticket 2.0’ calculation model, the ‘AFP-adjusted-to-HCC-size’ (AFP-UTS) criteria (‘Up To Seven’ for AFP <200 ng/ml, ‘Up To Five’ for AFP 200–400 ng/ml and ‘Up To Four’ for AFP 400–1000 ng/ml) were proposed for patient selection. 24
In this study, we investigated recurrence and survival after LT for HCC according to AFP-UTS and Milan criteria, and evaluated the correlation between radiological staging before LT and pathological staging on explant histology. We also assessed risk factors for early recurrence and the outcome of patients with early recurrence.
Materials and methods
Patients
We included all adult patients who underwent deceased donor liver transplantation for HCC at the Medical University of Vienna between 1997 and 2014. Patients with missing follow-up data or pre-transplant imaging were excluded. Patients with incidental HCC on explant histology were also excluded. In general, patients who fulfilled the Milan criteria 12 were candidates for LT. However, patients exceeding the Milan criteria, but who had undergone successful downstaging and/or who fulfilled expanded criteria (Up-To-Seven 15 or UCSF criteria 18 ) could also be considered for LT after multidisciplinary board discussion. Absolute contraindications included ongoing alcohol abuse, ongoing extrahepatic malignancies, extrahepatic metastases, severe cardiac and/or pulmonary diseases, severe pulmonary hypertension (mPAP >45 mm Hg), and ongoing severe infection. Relative contraindications included advanced age, persistent non-adherence and morbid obesity. 6 , 25
Bridging treatments were done whenever feasible regarding technical/anatomical aspects and liver function. In general, local ablation (percutaneous ethanol instillation (PEI) or radio frequency ablation (RFA)) was used in patients with a singular HCC tumour and transarterial chemoembolization (TACE) was performed in patients with multiple tumour nodules.
Assessment of AFP-UTS criteria
AFP-adjusted-to-HCC-size (UTS-AFP) criteria were assessed as previously published in the ‘Metroticket 2.0’ study by Mazzaferro and colleagues. 24 In brief, a patient was defined as being within AFP-UTS if the following criteria were fulfilled: ‘Up To Seven’ (meaning that the sum of the number of HCC nodules plus the size of the largest nodule in cm must not exceed 7) for AFP <200 ng/ml, ‘Up To Five’ for AFP 200–400 ng/ml and ‘Up To Four’ for AFP 400–1000 ng/ml. Patients with an AFP above 1000 ng/ml were defined as AFP-UTS out, regardless of their tumour characteristics.
Only nodules with a diameter of at least 1cm that fulfilled the European Association for the Study of the Liver (EASL) criteria 5 were considered as HCC nodules. After bridging therapy, in a partly necrotic tumour nodule the complete maximum diameter was measured, including any necrotic areas. Completely necrotic nodules were not counted as HCC nodules in the assessment of number of tumour nodules and size of the largest nodule.
Data collection
All data were collected retrospectively from the centre’s LT database.
HCC was diagnosed by pre-transplant biopsy or by radiological imaging, (contrast enhanced magnetic resonance imaging (MRI) and/or multiphase computed tomography (CT)). All blood parameters recorded for this study were taken at admission for LT in the ISO-certified laboratory of the Medical University of Vienna. AFP was usually determined at an earlier time point. The latest AFP value before LT was used for this study. In most patients, this AFP value was measured around the date of last radiological assessment before LT. Pathological analysis was performed by a senior liver pathologist and tumour grading was staged according to Edmondson and Steiner. 26 The Barcelona Clinic Liver Cancer (BCLC) system was used for tumour staging. 4 Child–Pugh score was recorded to describe liver function. 6
All LT procedures were performed at the Medical University of Vienna with grafts from brain dead deceased donors by specialized transplant surgeons. After successful LT, routine follow-up consisted of clinical examination and radiological surveillance with CT or MRI every 6 months. 6 , 25
Statistical analyses
Baseline tumour characteristics and clinical data are presented using descriptive statistics. Time to recurrence (TTR) was defined as the time from the date of LT until the date of recurrence of HCC, diagnosed either by imaging or by biopsy. Patients without documented recurrence were censored at the day of the last radiological follow-up examination or at the date of death if no radiological follow-up had been conducted before death. Early recurrence was defined as recurrence diagnosed within the first 12 months after LT.
7
Overall survival (OS) was defined as the time from the date of liver transplantation until the date of death. Patients who were still alive at 31 May 2019 (end of follow-up) were censored at the time of last contact. Survival plots were calculated by the Kaplan–Meier method and compared by log rank test (univariate analysis). Afterwards, variables that reached a
A
Ethical considerations
This retrospective analysis was approved by the local human research ethics committee of the Medical University of Vienna (approved on 13 November 2015; reference number 1759/2015). The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee. As this study is a retrospective analysis of anonymized patient data, written informed consent was not obtained.
The abstract to this manuscript has previously been presented as poster with the abstract number 873 at the scientific conference ‘The Liver Meeting 2019’ of the American Association for the Study of Liver Diseases (AASLD) in Boston, Massachusetts, USA. 27
Results
Patient characteristics
Of the 224 patients transplanted for HCC at our centre, 53 patients were excluded due to insufficient radiological documentation prior to LT and five patients because of missing follow-up data (Supplemental Figure 1).
Table 1 displays the baseline patient characteristics of the 166 eligible patients. All patients received deceased donor LT. There were 145 male patients (87.3%) and 21 (12.7%) female patients. In explant histology, the tumour nodules of 103 patients (62.0%) were within Milan criteria and 31 patients (18.7%) exhibited microvascular invasion. Of the 63 patients who exceeded Milan criteria in explant histology due to nodule number or size, 13 patients (20.6%) were Milan out because of number of nodules (more than 3 nodules, size of the largest nodule ≤3 cm), 43 patients (68.3%) because of tumour size (singular HCC >5 cm or ≤3 nodules, size of the largest nodule >3 cm) and 7 patients (11.1%) because of both (more than 3 nodules, size of the largest nodule >3 cm). Ten patients (6.0%) had a serum AFP level of 200 ng/ml or higher, and median serum AFP level was 9.56 ng/ml. Notably, the number of patients within radiologically assessed AFP-UTS and Milan criteria was the same (139 [83.7%] each), although not all of them were the same individuals; only 127 patients (76.5%) fulfilled both Milan and AFP-UTS criteria. Fifteen patients (9.0%) fulfilled neither AFP-UTS nor Milan criteria. Bridging treatment was done in 121 patients (72.9%); in 45 patients (27.1%) PEI was used, 37 patients (22.3%) received TACE and 16 patients (9.6%) were treated with RFA.
Patient characteristics.
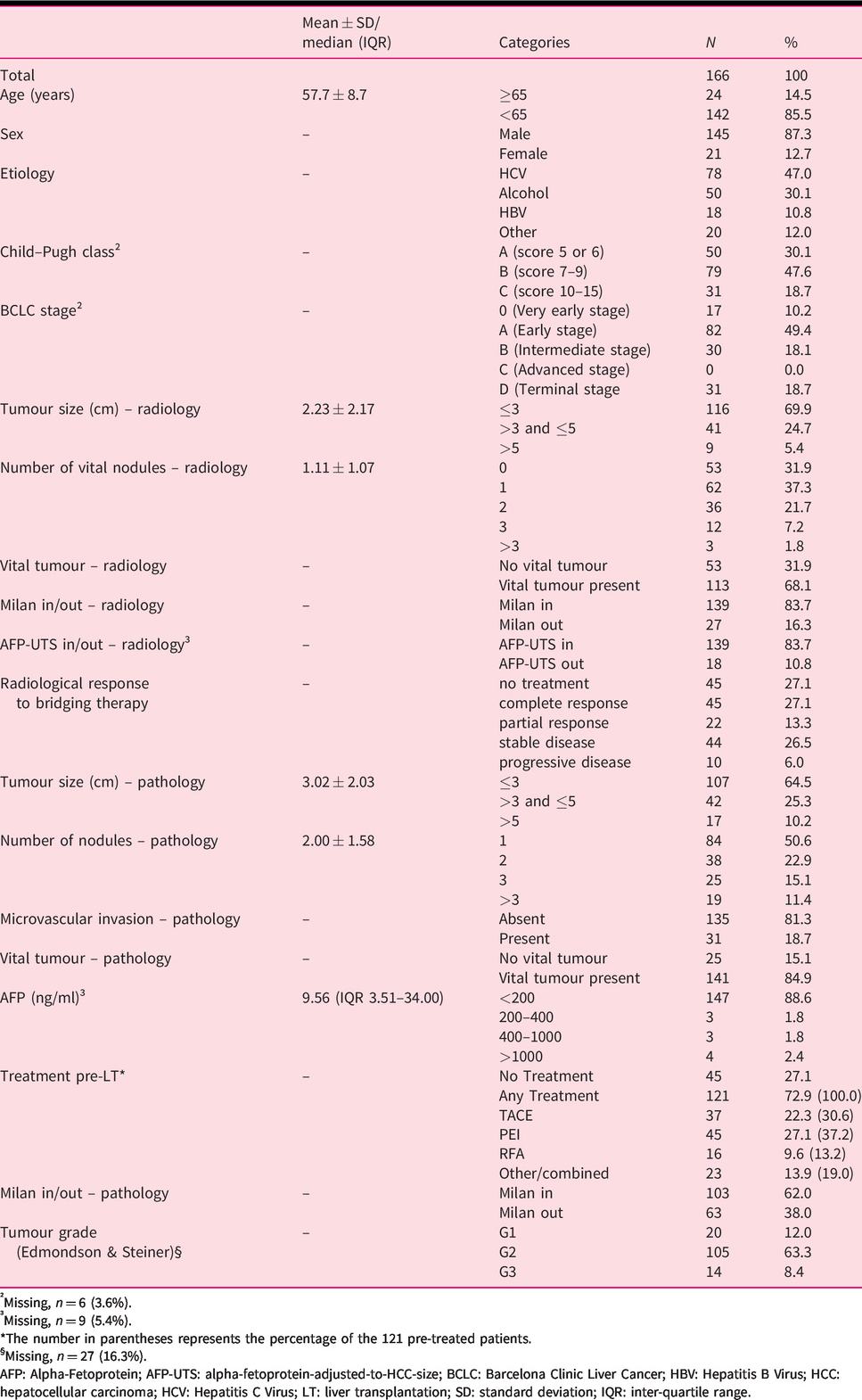
†Missing,
‡Missing,
*The number in parentheses represents the percentage of the 121 pre-treated patients.
§Missing,
AFP: Alpha-Fetoprotein; AFP-UTS: alpha-fetoprotein-adjusted-to-HCC-size; BCLC: Barcelona Clinic Liver Cancer; HBV: Hepatitis B Virus; HCC: hepatocellular carcinoma; HCV: Hepatitis C Virus; LT: liver transplantation; SD: standard deviation; IQR: inter-quartile range.
Radiological assessment was done with CT in 140 patients (84.3%) and with MRI in 26 patients (15.7%). Median time from radiological assessment to LT was 39 days (95% confidence interval (CI) 29.3–48.7 days). Median time from diagnosis of HCC to LT was 6.7 months (95% CI 5.9–7.5 months). In 36 patients who were diagnosed in a referral hospital, the exact date of diagnosis could not be determined.
Time from listing to LT was available in 112 patients (67.5%) who were transplanted in 2002 or later. In these patients, median time on the waiting list was 7.2 months (95% CI 6.0–8.4 months).
Correlation of AFP with tumour characteristics
We also assessed the correlation of serum AFP levels with tumour characteristics and found that high AFP (≥200 ng/ml) was significantly associated with not fulfilling the Milan criteria (70.0% vs. 36.7%;
Recurrence and survival of the whole cohort
Median duration of follow-up was 111.0 months. Of 166 patients included, 32 (19.3%) had recurrence of HCC after LT, and, in these patients, median TTR was 21.2 months (range 2.6–182.8 months).
Patients outside AFP-UTS had a significantly higher 1-/3-/5-/10-year recurrence rate (30.4%/54.9%/63.9%/63.9%) than patients within AFP-UTS (4.5%/14.5%/17.8%/24.7%;
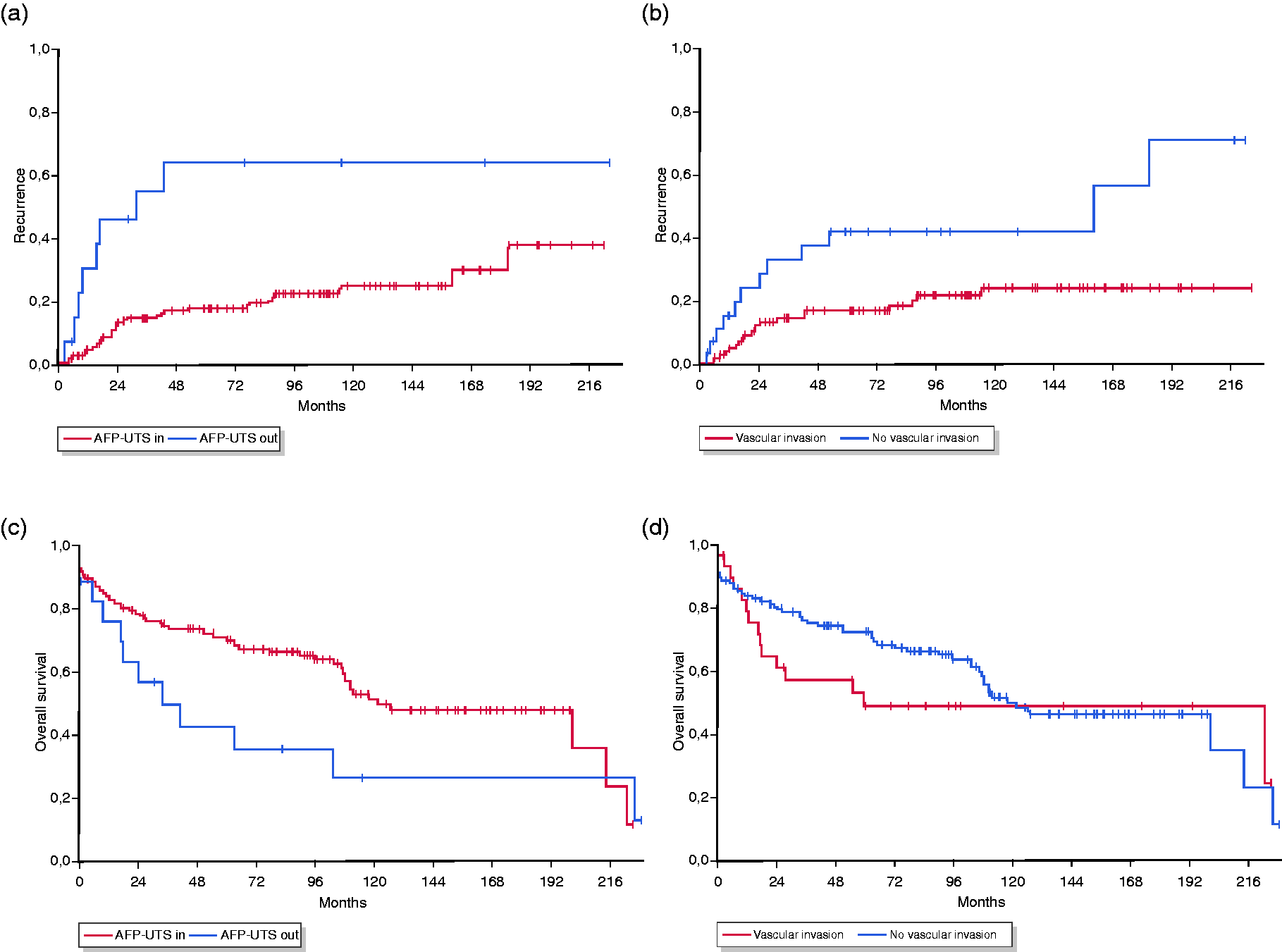
Kaplan–Meier plots.
Univariate analysis of time to recurrence.
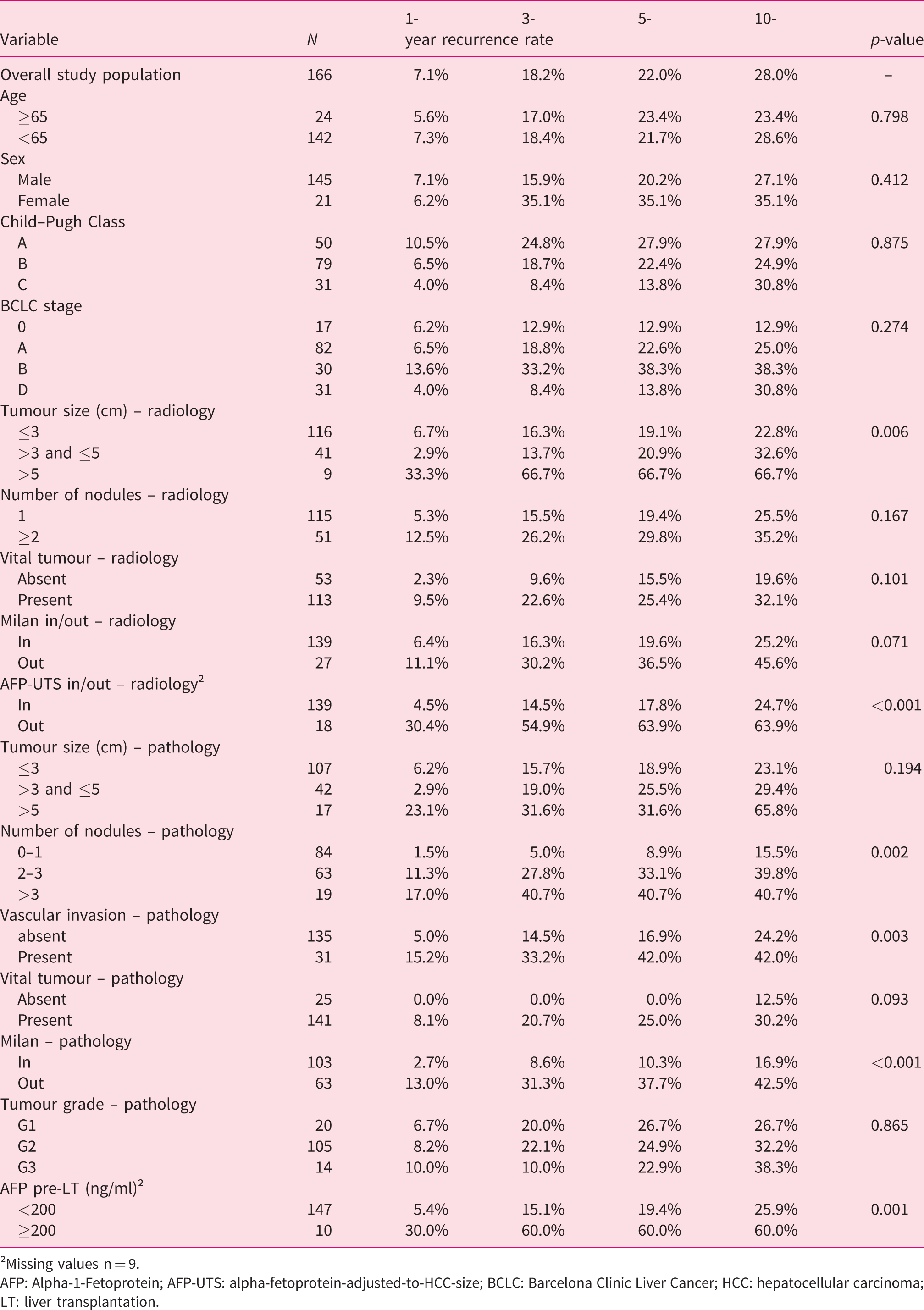
†Missing values n = 9.
AFP: Alpha-1-Fetoprotein; AFP-UTS: alpha-fetoprotein-adjusted-to-HCC-size; BCLC: Barcelona Clinic Liver Cancer; HCC: hepatocellular carcinoma; LT: liver transplantation.
Multivariate analysis of time to recurrence.
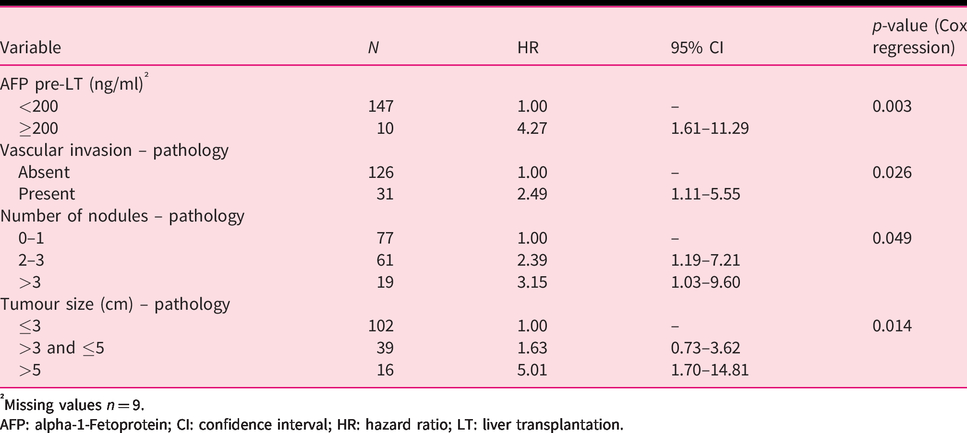
†Missing values
AFP: alpha-1-Fetoprotein; CI: confidence interval; HR: hazard ratio; LT: liver transplantation.
When patients were stratified into four groups according to Milan and AFP-UTS criteria in pre-LT radiological assessment, Milan in/AFP-UTS out patients had a higher 5-year recurrence rate than Milan out/AFP-UTS in patients: 5-year recurrence rate for Milan in/AFP-UTS in (
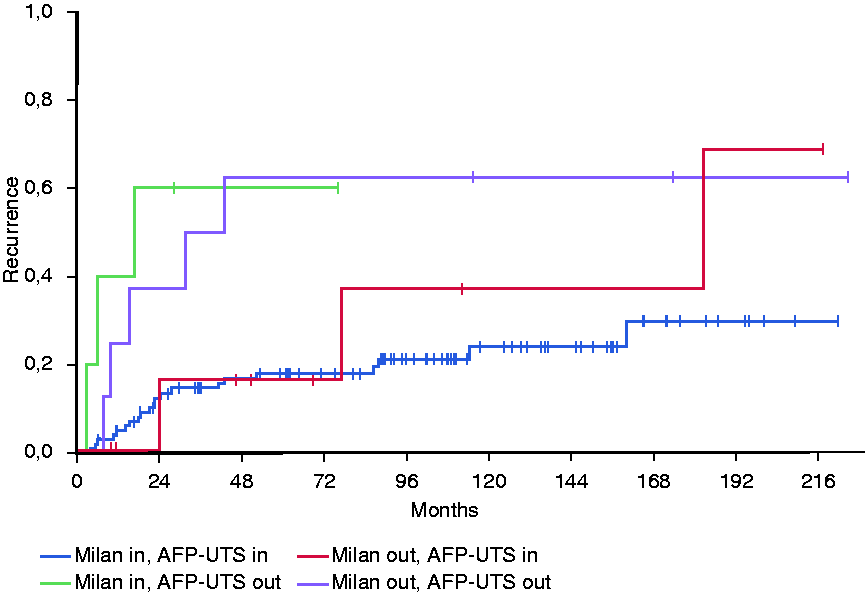
Kaplan–Meier Plot of time to recurrence (TTR) according to AFP-UTS and Milan criteria. Patients are split into four groups: both in, Milan out/AFP-UTS in, Milan in/AFP-UTS out, both out (
In total, 72 (43.4%) patients died during follow-up. Median OS of the total study population was 118.2 months (95% CI 66.2–170.3 months) with a 5-year survival rate of 68.1%. Patients outside AFP-UTS had a lower 1-/3-/5-/10-year survival rate (76.2%/50.0%/42.9%/26.8%) than patients within AFP-UTS (83.7%/74.9%/70.5%/51.7%;
When patients were stratified into four groups according to Milan and AFP-UTS criteria in pre-LT radiological assessment, there was no significant difference in OS between the four groups: median OS was 126.9 (95% CI 83.1–170.8) months for Milan in/AFP-UTS in, 27.9 (95% CI 0.0–141.3) months for Milan out/AFP-UTS in, 24.5 (95% CI 10.1–39.0) months for Milan in/AFP-UTS out, and OS 41.5 (95% CI 0.0–87.3) months for Milan out/AFP-UTS out (
Correlation between radiological and pathological assessments
A relevant number of patients were beyond the Milan criteria in explant histology, in both radiological Milan in (33.8%) and AFP-UTS in (33.8%) subgroups. There was no significant difference in the percentage of patients with vascular invasion in explant pathology between radiological Milan in and out patients or between AFP-UTS in and out patients, respectively. Notably, radiological AFP-UTS out was associated with a higher early recurrence rate but Milan out was not (Table 4).
Correlation of radiological Milan and AFP-UTS criteria with tumour characteristics and early recurrence.
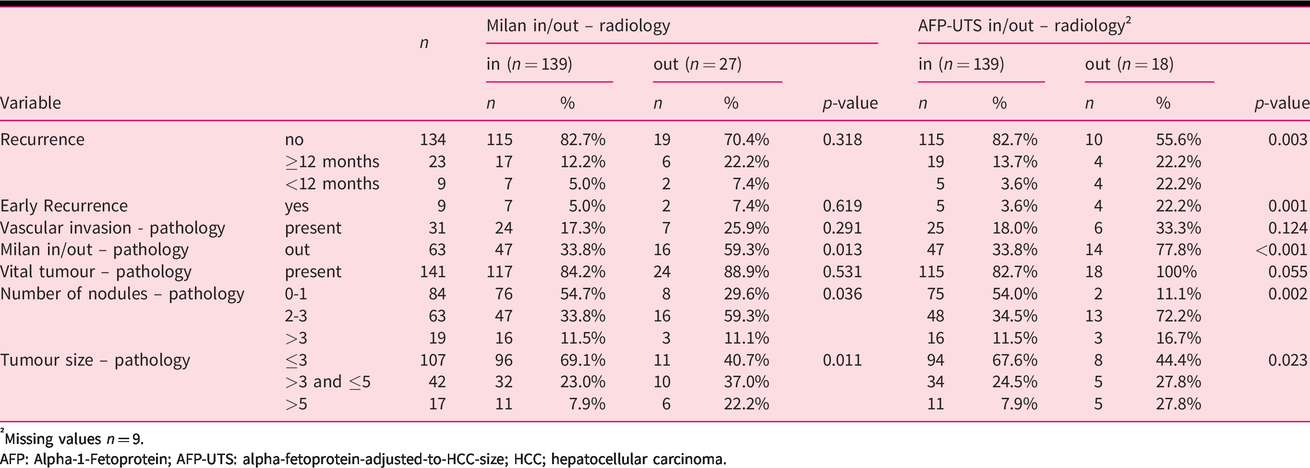
†Missing values
AFP: Alpha-1-Fetoprotein; AFP-UTS: alpha-fetoprotein-adjusted-to-HCC-size; HCC; hepatocellular carcinoma.
Recurrence and survival of patients within Milan according to radiological assessment
Supplemental Tables 3 and 4 show the OS rates and recurrence rates of patients who fulfilled the Milan criteria according to radiological imaging. The 5-year recurrence rate was significantly higher in patients with microvascular invasion (42.8% vs. 14.3%;
Early recurrence is associated with poor survival
Early recurrence (defined as recurrence within the first 12 months after LT
7
) was significantly associated with dismal survival (median OS 122.1 months, 95% CI 66.1–178.1 vs. 17.2 months, 95% CI 5.8–28.6;
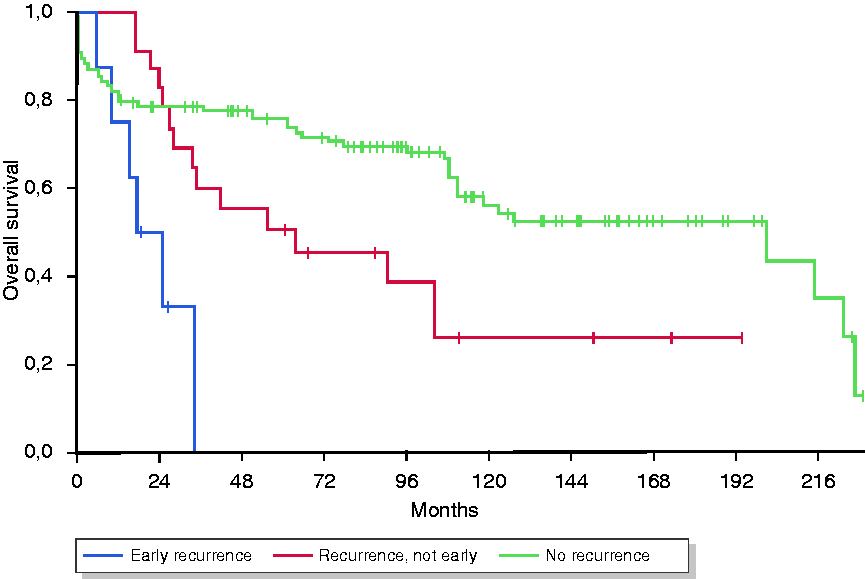
Kaplan–Meier Plot of overall survival (OS) according to early recurrence: no recurrence vs. recurrence after 12 months vs. early recurrence (
Early recurrence was significantly associated with AFP-UTS in pre-LT assessment but not with radiological Milan criteria. Other variables associated with early recurrence included AFP pre-LT as well as vascular invasion, number of nodules, and Milan status – all according to explant histology (Supplemental Table 5).
Discussion
In our cohort of 166 HCC patients who underwent LT, the OS of patients within AFP-UTS criteria was excellent, with a median OS of 126.9 months (vs. 34.2 months for those outside AFP-UTS) and a 5-year survival rate of 70.5% (vs. 42.9%). This is in line with the results of the ‘Metroticket 2.0’ study 24 and slightly exceeds the recommended minimum of 70%, which is the survival rate to be expected from a cohort of patients who received LT for a non-malignant indication. 19
However, expanding the criteria for LT from Milan to AFP-UTS would not have increased the number of patients eligible for LT, as the number of patients who fulfilled the Milan or AFP-UTS criteria according to radiological imaging was the same (139 each), although not all of them were the same individuals (only 127 patients fulfilled both Milan and AFP-UTS criteria).
In our cohort, the recurrence rate was significantly lower in patients fulfilling AFP-UTS criteria (5-year recurrence rate 17.8% for AFP-UTS in vs. 63.9% for AFP-UTS out,
Vascular invasion is a known risk factor for high recurrence and poor survival in HCC patients undergoing LT.
4
,
6
Consequently, the criteria for pre-LT should ideally minimize the risk of microvascular invasion in explant histology. However, in our cohort, 18% of the patients who were AFP-UTS in and 17% of the patients within Milan according to pre-LT imaging exhibited vascular invasion in explant histology. Thus, neither Milan nor AFP-UTS criteria are able to completely rule out known risk factors for post-LT recurrence such as vascular invasion. In the subgroup of patients within Milan on pre-LT imaging (
Not surprisingly, those with vascular invasion in explant histology showed a significantly higher recurrence rate (5-year recurrence rate 14% vs. 43%;
The major limitations of this study are the small number of patients in some subgroups, particularly in the AFP-UTS out group, and its retrospective character with all its potential confounders.
In conclusion, the outcome of patients fulfilling AFP-UTS criteria was favourable in our cohort with a 5-year survival rate above 70%. Early recurrence and vascular invasion according to explant histology were associated with worse outcome after LT. Even though AFP-UTS criteria incorporate AFP, a marker for disease biology, they were not able to rule out vascular invasion with a higher certainty compared with the Milan criteria. However, AFP-UTS criteria showed a better association with recurrence, particularly early recurrence, than the radiologically assessed Milan criteria.
Supplemental Material
sj-pdf-1-ueg-10.1177_2050640620948665 - Supplemental material for Alpha-fetoprotein-adjusted-to-HCC-size criteria are associated with favourable survival after liver transplantation for hepatocellular carcinoma
Supplemental material, sj-pdf-1-ueg-10.1177_2050640620948665 for Alpha-fetoprotein-adjusted-to-HCC-size criteria are associated with favourable survival after liver transplantation for hepatocellular carcinoma by Tobias Meischl, Susanne Rasoul-Rockenschaub, Georg Györi, Bernhard Scheiner, Michael Trauner, Thomas Soliman, Gabriela Berlakovich and Matthias Pinter in United European Gastroenterology Journal
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TM, SR-R, GG, BS and TS declare no potential conflicts of interest.
MT has served as a speaker and/or consultant and/or advisory board member for Albireo, Bristol-Myers Squibb, Dr Falk Pharma, Gilead, Intercept, MSD, Novartis, Phenex Pharmaceuticals and Regulus, and has received research funding from Albireo, Dr Falk Pharma, Gilead, Intercept, MSD and Takeda. MT is listed as a co-inventor on patents on the medical use of nor-ursodeoxycholic acid.
GB has received grant support from Sandoz, Chiesi, MSD, Shire; speaking honoraria from Neovii, Chiesi, Astellas; consulting/advisory board fee from Astellas, Neovii, Chiesi, Sanofi; and travel support from Astellas, Chiesi, Neovii, Biotest.
MP has served as a speaker and/or consultant and/or advisory board member for Bayer, Bristol-Myers Squibb, Eisai, Ipsen, Lilly, MSD, and Roche.
Ethics approval
This retrospective analysis was approved by the local human research ethics committee of the Medical University of Vienna (approved on 13 November 2015; reference number 1759/2015). The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
As this study is a retrospective analysis of anonymized patient data, written informed consent was not obtained.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
