Abstract
Background
The rates of perforation and bleeding are important quality measures of colonoscopy performance.
Objective
The objective of this article is to assess the frequency of colonoscopy-related bleeding and perforation in Swedish counties and to relate these findings to patient characteristics.
Method
Data on 593,308 colonoscopies performed on adults from 2001 to 2013 were retrieved from Swedish inpatient and outpatient registers. Covariates were assessed in a multivariate Poisson regression model. The correlation between perforation and bleeding was calculated with Pearson’s bivariate correlation formula.
Results
The relative frequency of bleeding and perforation vary across counties (bleeding: 0.02%–0.27%; perforation: 0.02%–0.27%). There were significant positive correlations between the relative frequency of bleeding and perforation at the county level, both including (r = 0.792, p < 0.001) and excluding polypectomies r = 0.814 (p < 0.001). The relative risks of these conditions in the counties ranged from 0.12, p < 0.001, to 1.53, p = 0.05 (bleeding) and from 0.17, p = 0.002, to 2.42, p < 0.001 (perforation).
Conclusions
There are substantial differences in colonoscopy performance in Sweden. These differences do not seem to be explained by patient characteristics.
Key summary
Summarise the established knowledge on this subject
Colonoscopy is a relatively safe procedure; however, there are risks of adverse events (AEs). Perforation and bleeding are serious AEs that may have lethal outcomes. There is scant knowledge about the impact of patient-related factors that may minimise colonoscopy-related perforation and bleeding.
What are the significant and/or new findings of this study?
There is large variation in the relative frequencies of both perforation and bleeding across Swedish counties and individual colonoscopy units. There is a strong positive correlation between the frequency of perforation and the frequency of bleeding at the county level, as well as at the colonoscopy unit level. Differences between counties do not seem to be explained by patient characteristics. It seems that the bleeding and perforation rates reflect the colonoscopy performance rather than differences in therapeutic colonoscopy rates or the complexity of therapeutic procedures across Swedish counties.
Introduction
Colonoscopy is an important diagnostic and therapeutic procedure, as well as a primary and secondary screening tool for colorectal cancer (CRC). It is a relatively safe procedure; however, there are risks of adverse events (AEs). Moderate and severe colonoscopy-related AEs include the perforation of the bowel, major bleeding, splenic injuries and death. In a recent meta-analysis and literature review by Reumkens et al., the perforation rate was estimated to be 0.5/1000 colonoscopies, with a 95% confidence interval (CI) of 0.4–0.7, and the bleeding rate was estimated to be 2.6/1000 colonoscopies, with a 95% CI of 1.7–3.7. 1 These figures include colonoscopies performed for diagnostic, screening and therapeutic purposes. Lower frequencies of perforation and bleeding were observed in colonoscopies performed for screening purposes. Specifically, for perforations, the frequency was 0.3/1000 (95% CI: 0.2–0.5), and for bleedings, the frequency was 2.4/1000 (95% CI: 0.9–4.6) in such cases. Death caused by colonoscopy is an uncommon AE. In a recent study, the death rate as a result of bleeding or perforation was estimated to be 0.013% in a Swedish cohort study of 593,315 colonoscopies from 2001 to 2013. 2 This is consistent with another study by deRoux and Sgarlato, which showed the colonoscopy death rate in a small population in New York City to be 0.014% between 2005 and 2009. 3 In Sweden, there are few studies concerning colonoscopy-related AEs. 2,4,5
In this retrospective register study of colonoscopies performed from 2001 to 2013, we have assessed the perforation and bleeding rates in Swedish counties and colonoscopy units. Our hypothesis was that differences in these rates could be explained by patient-related characteristics.
Materials and methods
The setting for this cohort study was the entire Swedish population (9.5 million inhabitants at the end of the study). This study was approved by the local ethical committee at Karolinska Institutet (Dnr: 2015/690-31/2) and conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in prior approval by the institution’s human research committee.
Geographical classification
In April 2017, Sweden had a land area of 407,340 km2 and 10,032,357 inhabitants.
6
Sweden is divided into 21 counties, and the northern parts of the country are sparsely populated (see Figure 1). Each county organises healthcare, including administration, hospitals and primary care, as well as specialist, inpatient and outpatient care. However, private entrepreneurs are also providers, and the extent to which this occurs differs by region. The number of colonoscopy units varies by county (from one to 12 in our register). The majority of endoscopies are financed by public taxes and thus independent of private and public providers. As with other areas of healthcare, colonoscopy units differ by county in terms of organisation, procedural routines, who performs the procedure and how the units are equipped. In the following presentation, counties are anonymised and designated by randomly selected numbers.
Swedish counties and numbers of inhabitants per square kilometre, 2016.
6
The majority of areas in the country are sparsely populated.
Patients and registers
The data were collected retrospectively from the patient registries (National Hospital Discharge Register and National Outpatient Register) for residents of Sweden who were ≥18 years old, as previously described in detail. 2 Using the codes for colonoscopy (UJF32 or UJF35), individuals who underwent one or more colonoscopies from 2001 to 2013 were included in the study. Patients in whom a polypectomy in the colon or rectum (e.g. rectoscopy) was performed during a procedure other than a colonoscopy, as well as patients for whom there was only a code for polypectomy but no code for endoscopy, were not included. All the data in the dataset were anonymised. With the present legislation, procedures performed by nurses are not recorded in the registers, and therefore, data about nurse endoscopies are not available. Moreover, because of their various reimbursement mechanisms, some private caregivers do not report their colonoscopies. In the two counties with the most inhabitants, as many as 50% of colonoscopies are not registered for the mentioned reasons. In addition, the endoscopist’s specialty is not available in the registers. Diagnoses were searched using the International Classification of Diseases (ICD 7, ICD 9 and ICD 10).
AEs
The AEs considered were bleeding and perforation within 30 days of the procedure. The National Hospital Discharge Register and the National Outpatient Register were searched using the codes for those diagnoses (T812 for perforation and T810 for bleeding). If an individual underwent more than one colonoscopy on the same day, only one procedure was considered in the risk calculations.
Covariates
The covariates considered in the analyses were age, sex, comorbidity, polypectomy, general anaesthesia, the geographical location of the hospital and time period. Data were included as to whether a polypectomy was performed, but there were no data regarding the number and size of the polyps for each procedure. The comorbidity diagnoses used in the analyses were diabetes mellitus, diverticular disease of the colon, Crohn disease, ulcerative colitis and ischaemic heart disease. The general anaesthesia covariate included information regarding whether the procedure was performed under general anaesthesia, but there were no data about what type of medical preparation was employed. According to Swedish clinical practice, propofol is used during anaesthesia in the majority of cases. Two time periods were compared: 2001–2006 and 2007–2013.
Statistical analysis
The Pearson bivariate correlation formula was used in the correlation analyses. A Poisson regression with robust standard errors was used in both the univariate and multivariate analyses. A Poisson regression is often used for count data, but it can also be used in analysing relative risks in cases of binary outcomes . 7 All the covariates mentioned above were included in the multivariate analysis. The covariates were either categorised or dichotomised (yes or no). Participants were categorised into seven groups based on age, with the youngest age group being set as a reference. All the covariates in the multivariate analysis were selected a priori, and therefore, a p value of less than 0.05 was considered significant and sufficient for multiple comparisons. The dataset was large enough to allow for several comparisons. When analysing the correlations between and relative frequencies of bleeding and perforation at the colonoscopy unit level, we excluded sites that performed fewer than 500 colonoscopies per year, on average, from 2001 to 2013. Such low numbers of colonoscopies would be difficult to interpret and could be a result of reporting bias. In the correlation analysis, at the county level, all the registered colonoscopies are included. In addition, they are all included in the multivariate regression analysis, along with the frequencies and relative frequencies of bleeding and perforation.
All analyses were performed using Stata 13.1 (StataCorp, College Station, TX, USA).
Results
Frequencies and relative frequencies of bleeding and perforations in Swedish counties during 2001–2013.
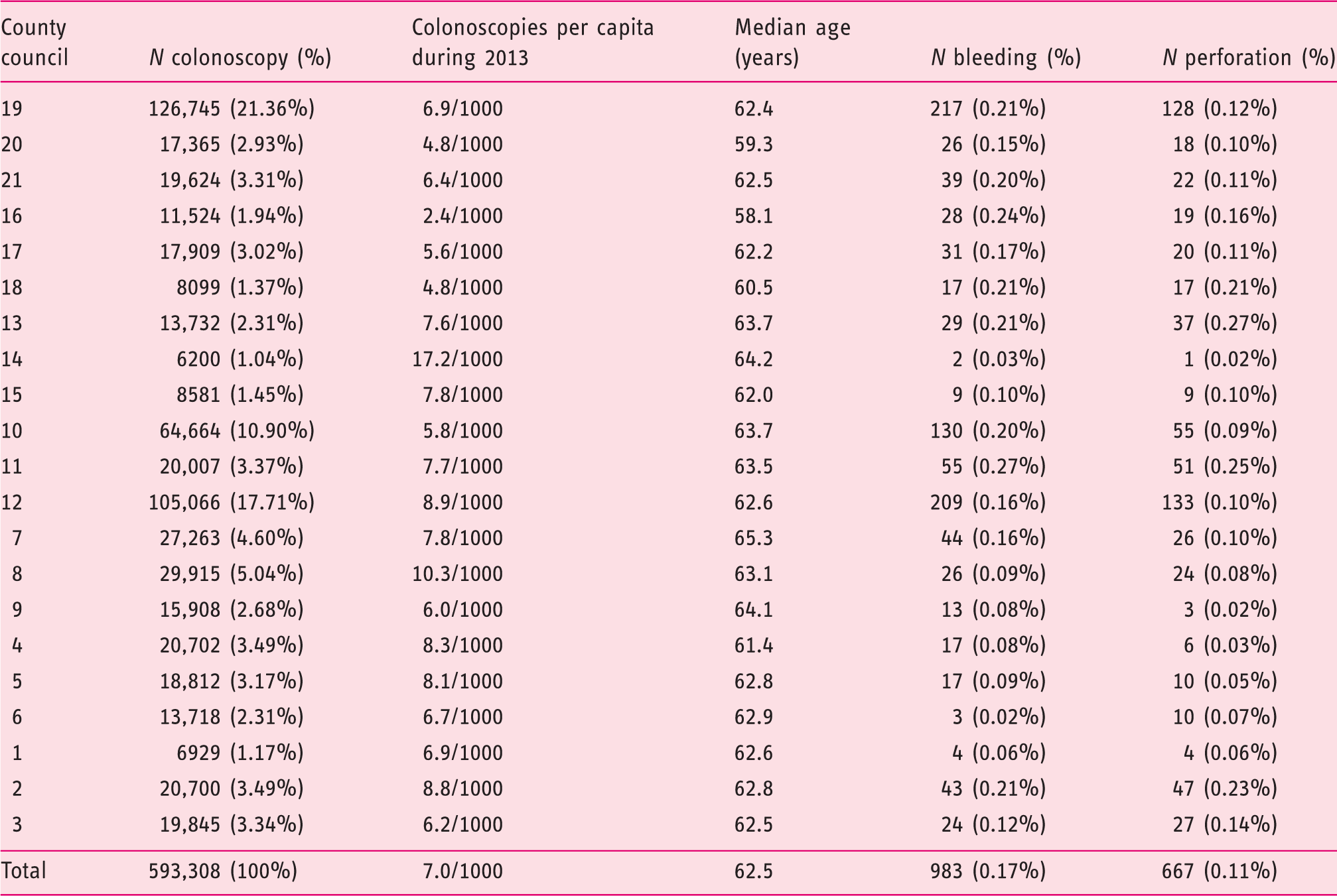
Bleeding and perforations per total number of colonoscopies in respective counties 2001–2013. Regions sorted from lowest to highest relative to frequency of bleeding.
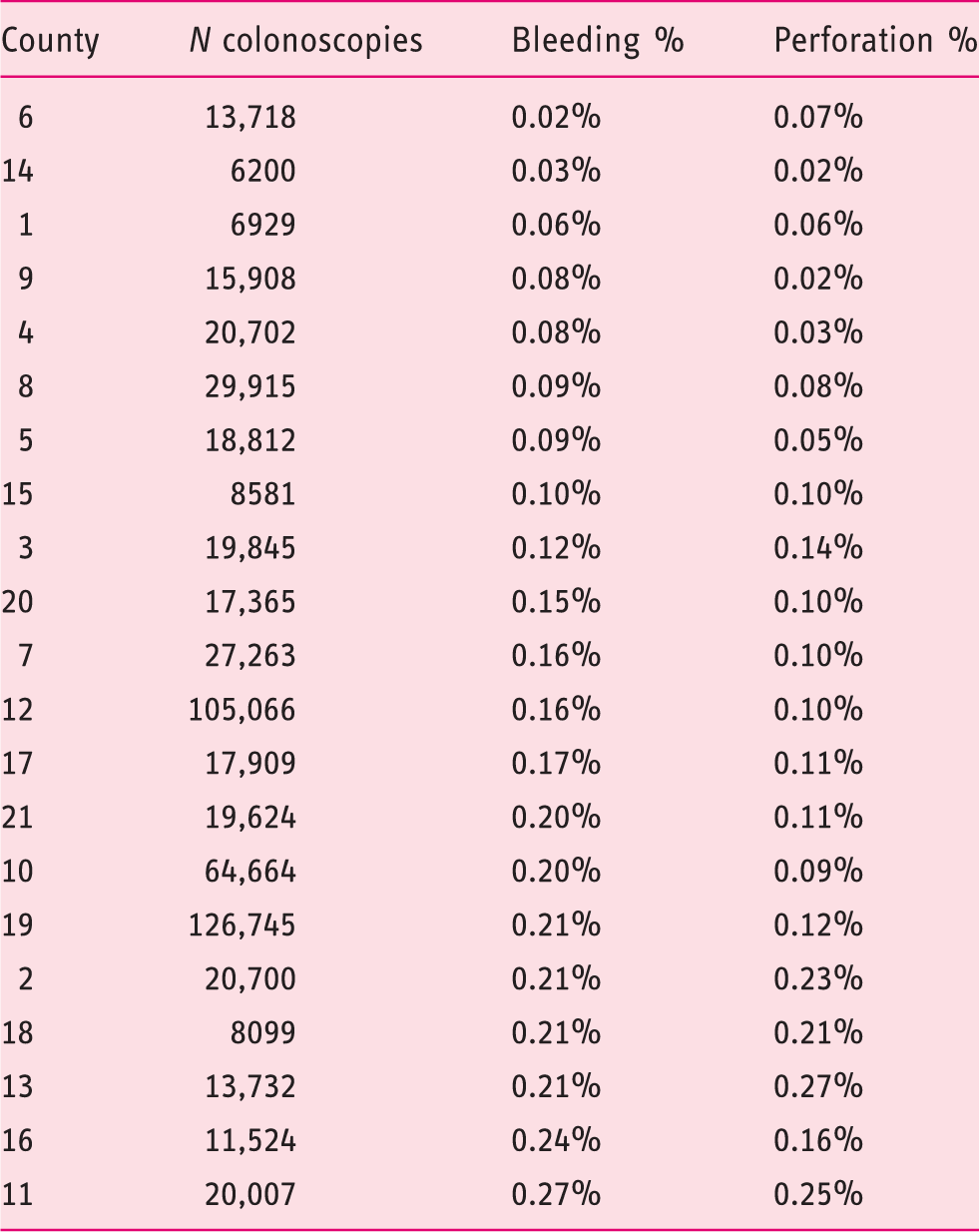
The Pearson correlation coefficient between the relative frequency of bleeding and the relative frequency of perforation at the county council level is 0.792 (p < 0.001). Figure 2 illustrates this correlation. There is also a correlation between the relative frequency of bleeding and the relative frequency of perforation at the colonoscopy unit level (r = 0.61, p < 0.001). There is no significant correlation between how many procedures were performed at either the county level or the colonoscopy unit level and the relative frequencies of the above-mentioned AEs.
Relative frequency of perforation vs relative frequency of bleeding for each county (dots).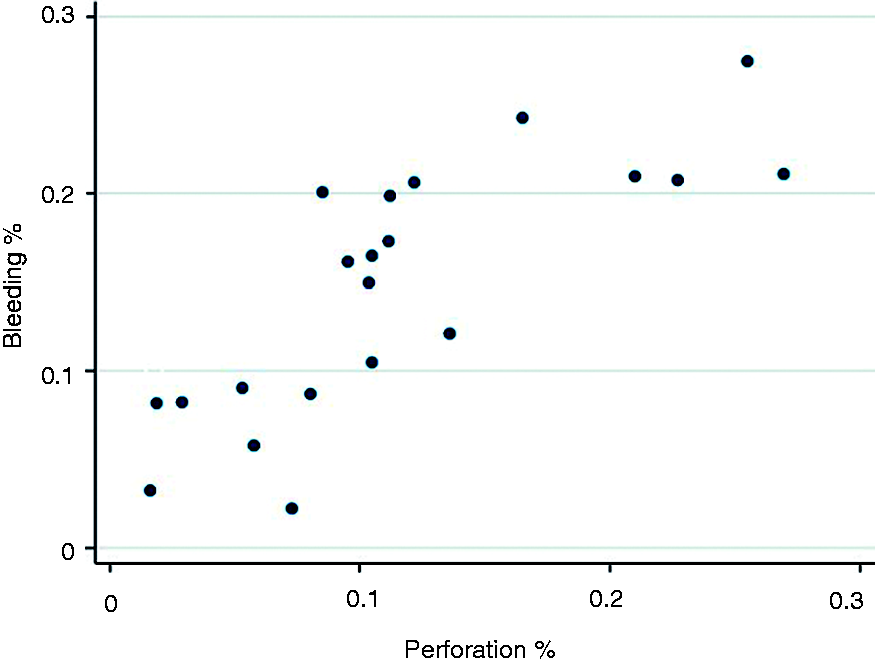
Multivariate analyse of bleeding and perforation in Sweden 2001–2013.
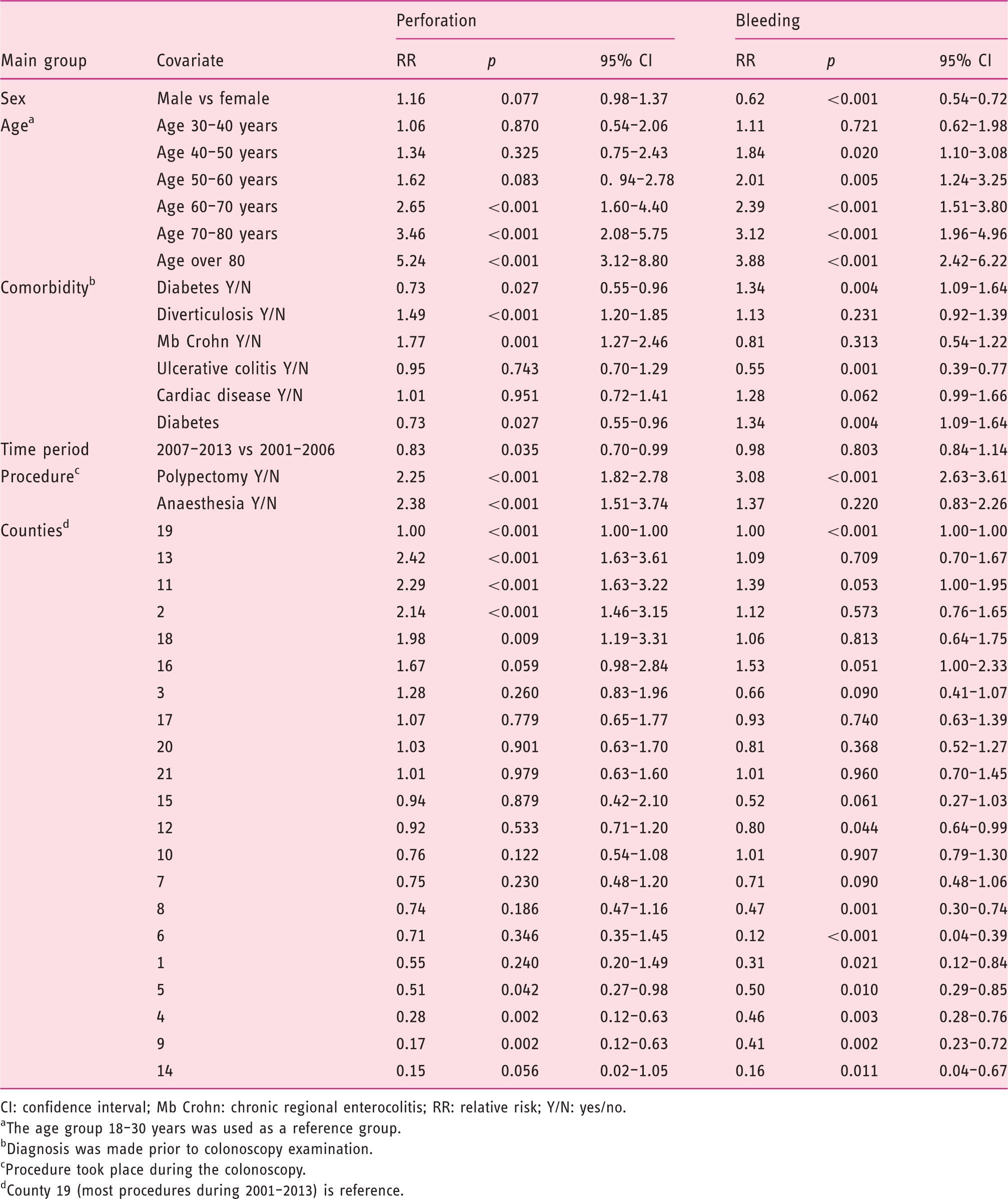
CI: confidence interval; Mb Crohn: chronic regional enterocolitis; RR: relative risk; Y/N: yes/no.
The age group 18–30 years was used as a reference group.
Diagnosis was made prior to colonoscopy examination.
Procedure took place during the colonoscopy.
County 19 (most procedures during 2001–2013) is reference.
After excluding polypectomies from the analysis, the relative frequency of bleeding was 0.13% and that of perforation was 0.10%. The relative frequency of bleeding after a colonoscopy varied from 0.022% to 0.21% in the counties, while that of perforation varied from 0.00% to 0.25%. When polypectomies were excluded, the Pearson correlation coefficient between the relative frequency of bleeding and relative frequency of perforation at the county level was 0.814 (p < 0.001).
Discussion
Limited endoscopic technical skills may be reflected in an increased AE frequency, especially during diagnostic procedures. Technical skills may also reflect the ability to manage and recognise AEs in the endoscopy room. Bielewska et al. showed that colonoscopies performed by surgeons and endoscopists of unknown specialty had higher rates of perforation than those performed by gastroenterologists (odds ratio, 2.00; 95% CI, 1.30–3.08). They suggest that differences in an endoscopist’s specialty may reflect how his or her education is structured and that gastroenterologists may perform more colonoscopies than other endoscopists. 8 This is consistent with several studies reporting that gastroenterologists are more effective than surgeons or primary care physicians in terms of preventing CRC via colonoscopy screening. 9–13 The positive impact of education on colonoscopy quality, as measured by the adenoma detection rate (ADR), is shown in two randomised, controlled trials (RCTs). The ADR is commonly used as a measurement of colonoscopy quality for CRC screening programs. Training colonoscopists improves ADR and is superior to only receiving feedback regarding individual performance.14,15 The importance of training as it relates to AEs is not known. In Sweden, endoscopies are performed by surgeons, gastroenterologists and, in rare cases, radiologists. Historically, more surgeons than gastroenterologists performed colonoscopies, though this is changing over time. The number of colonoscopies performed per endoscopist varies from only a handful yearly to more than 1000 procedures per year. There are no national standards regarding how to organise training for or evaluate trainees and specialists.
The varying availability of equipment, such as high-resolution screens, carbon dioxide insufflations and flush pumps, as well as the availability of paediatric endoscopes when needed, may help explain the above-mentioned differences. In addition, differences in procedural routines, such as the bowel preparation regimen, how and when to stop anticoagulants before the procedure, the use of clips and the use of cold or hot snares, may be important. In our multivariate analysis, we have attempted to control for the development of equipment and routines by comparing two periods (2001–2006 and 2007–2013). There are lower, but not significantly lower, RRs of bleeding and perforation in the latter period (see Table 2).
Geography may matter. Sweden is a country of long distances and relatively few inhabitants, especially in the northern region. Remote areas provide endoscopies for inhabitants despite low expected volumes; otherwise, patients would have to travel long distances. Colonoscopy unit size varies from large centres with full-time endoscopists in urban regions to small centres with one or two part-time endoscopists. In a small unit in a sparsely populated area, the possibility for peer review and help from colleagues may be limited, and therefore, there is a higher likelihood of continuing with a difficult procedure despite a lack of help. Otherwise, a new endoscopic procedure must be scheduled for a patient who must travel long distances. There is also a possibility that in cases in which the cecum is not reached, instead of re-examination, computed tomography (CT) of the colon is more likely to be an option. The use of CT of the colon instead of re-examinations may decrease the potential risk of bleeding and perforation, as defined in this study, but on the other hand, it may increase the risk of missing small premalignant or malignant lesions.16,17 There is also a possibility that in colonoscopy units with less experience, surgical resections of advanced or malignant polyps, rather than endoscopic procedures (endoscopic submucosal/mucosal dissections and polypectomies), may be performed in higher numbers.
Rabeneck et al. showed that those who underwent colonoscopies performed by low-volume endoscopists were three times more likely to experience bleeding or perforation than those whose colonoscopies were performed by a high-volume endoscopist (range 379–1225, median of 417 procedures annually). The risk of AEs was increased under a threshold of approximately 300 colonoscopies per year. 13 In our cohort, we do not observe any correlation between the numbers of colonoscopies performed yearly and the relative frequencies of perforation or bleeding at the unit or county levels. However, in our study, it was not possible to measure the number of colonoscopies per endoscopist. Variations in individual endoscopists’ experience and volume may help explain the differences revealed by our study.
The strength of this study is that it is nationwide and a large number of colonoscopies were studied. Swedish patient registers have high validity. For example, Ludvigsson et al. showed that the positive predictive value of the diagnoses in the Swedish inpatient register has a high validity, estimated at 85%–95%. 18
Our study has certain limitations. Firstly, Blanks et al. showed that polyp size, patient American Society of Anaesthesiologists (ASA) grade and cecal polyp location significantly affect the risk of bleeding. 19 Our model considered whether polypectomy was performed, not how many polyps were removed or the size of such polyps. An ASA grade was not available, but we considered heart disease and diabetes mellitus, which may reflect ASA classifications to some extent. Secondly, Hamadi et al. showed that lower body mass index (BMI) resulted in a higher risk of perforation. 20 Data on BMI are not available in the registers. Thirdly, we did not control for the use of acetylsalicylic acid, warfarin or novel anticoagulants (NOACs). Forsberg et al. controlled for the use of acetylsalicylic acid and anticoagulants (warfarin or NOACs) in an analysis of the present cohort for the 2006–2013 time period. Without controlling for anticoagulants, they reported slight differences in RRs between the northern and southern parts of Sweden. After controlling for the use of acetylsalicylic acid and warfarin, this slight difference was no longer observed. 2
Colonoscopy AEs are difficult to capture because they often become apparent after the patient leaves the endoscopy unit. We analysed national datasets for codes of bleeding and perforation and linked these with colonoscopies performed up to 30 days before the AE became apparent. We cannot be sure there is a linkage to colonoscopy. Theoretically, there could be another event explaining major bleeding or perforation within 30 days of a colonoscopy, for example, ulceration of another origin or perforation due to the penetration of the colon for other reasons. We think it is likely that such events were limited in their extent. Another possibility is that patients who underwent colonoscopy were admitted to the hospital and codes for perforation and bleeding were registered incorrectly. We also consider this to be extremely rare.
As mentioned above, procedures performed by nurses are not registered, and therefore, data about nurse endoscopies are not available or easily objectified. This will be a considerable drawback to future studies because the number of nurse endoscopies has increased markedly in the last three to four years in Sweden. In terms of the studied time period (2001–2013), our perception is that there has been a recent increase from almost zero in the first years to a very small proportion of all colonoscopies in 2013. (The first nurse-performed colonoscopies began in 2006 in Sweden.) In the two counties with the most inhabitants, up to 50% of colonoscopies are not registered because organisations have large numbers of private caregivers and local agreements. This issue cannot be solved in this retrospective study. However, we think that the large total number of colonoscopies in the cohort makes the analyses relevant.
The risk of perforation is significantly increased when general anaesthesia is employed (RR 2.38, p < 0.001; see Table 3). This is in line with other studies. 21 Colonoscopies in the oldest age groups are also afflicted with an alarmingly increased risk of perforation (RR 3.46 at age 70–80 years and RR 5.25 above 80 years, both with p < 0.001). This may have consequences for the choice of endoscopist in examinations with anaesthesia and in the most fragile patients; in these cases, it should be stipulated that the most experienced colleagues are needed. Differences in RRs based on comorbidity, anaesthesia and age in the studied cohort were analysed in a study by Forsberg et al. 2
In summary, there are noticeable differences between the counties regarding bleeding and perforation rates, and these differences persist even after considering comorbidities, sex and age. Our data demonstrate a significant, positive correlation between the relative frequency of bleeding and that of perforation at the county level. These differences and this same positive correlation are still observed after excluding polypectomies from the analysis. This suggests that bleeding and perforation rates reflect colonoscopy performance rather than differences in therapeutic colonoscopy rates or complexity of therapeutic procedures across counties. One potential approach to balancing these inequalities would be to improve colonoscopy skills. Swedish administrative healthcare, with its division into self-governing counties, constitutes a good basis for future research regarding the factors that improve colonoscopy quality.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This work was supported by a regional agreement on medical training and clinical research (ALF) between Stockholm County and the Karolinska Institutet, as well as by the Swedish Society of Medicine, the Capio Research Fund and the Swedish Cancer Society.
Informed consent
Not applicable as anonymised data was used in the article.
Ethics approval
This study was approved by the local ethical committee at Karolinska Institutet (Dnr: 2015/690-31/2) and conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in prior approval by the institution’s human research committee.
