Abstract
Background
Achalasia is a neurodegenerative disorder of the oesophagus. Alteration of motor activity induced by oesophageal distension has not been explored.
Objectives
To investigate this function, using high-resolution Manometry.
Methods
This study enrolled 15 healthy subjects, 15 nonobstructive dysphagia (NOD), and 18 achalasia patients successfully treated with pneumatic dilation (six with restored peristalsis). The three groups underwent five rapid (<1 s) intraoesophageal infusions of 20-ml air boluses, followed by eight 5-ml water swallows.
Results
Whereas the response rate to water swallows was similar in the three groups, air infusion induced a lower response rate in achalasia (median, interquartile range: 70%, 40–100%) and, to a lesser extent, in NOD patients (100%, 60–100%) than in healthy subjects (100%, 100–100%;
Conclusions
Motor response to rapid air infusion was variably impaired in achalasia. The role of this alteration in the long-term outcome deserves evaluation.
Introduction
Oesophageal distension triggers a motor response in the oesophageal body, 1,2 which is physiologically important when food or air are retained in the oesophagus after failed primary peristalsis or after reflux from the stomach. Using standard oesophageal manometry, rapid infusion of air into the oesophagus has been shown to consistently induce motor activity in healthy subjects and, less frequently, in patients with nonobstructive dysphagia (NOD). 2,3 Oesophageal motor response to distension is the result of oesophageal reflexes: mechanoreceptors in the oesophagus, stimulated by distension, initiate a reflex that involves both the extrinsic vagal nerve pathways and the intrinsic enteric neural plexus. 4
Achalasia results from a well-documented degeneration of both intrinsic enteric neural plexus and extrinsic vagal innervation. 5 Furthermore, involvement of sensory neural pathways has been recently suggested. 6 –8 It is therefore likely that achalasia patients have alteration in triggering of oesophageal motor response to rapid distension, but objective data in this field are lacking.
The aim of our study is to evaluate triggering and characteristics of distension-induced compared to swallow-induced oesophageal motor response, in NOD and achalasia patients, compared to healthy subjects, using high-resolution manometry (HRM).
Materials and methods
Study population
This study enrolled 15 healthy subjects (six (40%) men, median age 29 years (interquartile range, IQR, 25–32 years) as a control group; they had no gastrointestinal symptoms, no history of upper gastrointestinal tract surgery, and did not use any medications. Fifteen NOD patients (seven (46.6%) men, median age 61 years IQR 46 to 75 years), referred to our centre for oesophageal manometry, were consecutively enrolled. Dysphagia was occasional in 11, daily in three, and at each meal in one patient; two patients had experienced loss of weight; four had also typical gastro-oesophageal reflux symptoms. Twelve patients had weak peristalsis (nine with large breaks, two with small breaks, and one with frequent failed peristalsis) and three had normal peristalsis. Exclusion criteria for NOD patients were: systemic diseases which could affect oesophageal motility (i.e. diabetes, scleroderma), eosinophilic oesophagitis, or the following motility disorders, as defined in the Chicago classification: 9 achalasia, absent peristalsis, oesophagogastric junction outflow obstruction, distal oesophageal spasm, hypercontractile oesophagus, and nutcracker oesophagus. Finally, 18 patients with achalasia (7 (38%) men, median age 62 years (IQR 50 to 73 years), successfully treated with pneumatic dilation and routinely followed up in our centre, were consecutively enrolled. Sixteen of them had type 2 and two had type 1 achalasia at diagnosis, according to the Chicago classification: 9 in six of the type 2 achalasia patients, peristaltic motor activity was restored after pneumatic dilation.
Both NOD and achalasia patients underwent an X-ray barium swallow, and anteroposterior diameter of the oesophageal body was similar in the two groups (median 1.9 cm (IQR 1.6 to 2.0 cm) vs. median 2.2 cm (1.6 to 2.5 cm;
Informed consent was obtained from each patient and healthy subject included in the study. The study was approved by the Human Research Review Committee of the Fondazione IRCCS Ca’Granda – Ospedale Maggiore Policlinico of Milan.
Oesophageal manometry
HRM (Solar HRM, MMS, The Netherlands) was performed using a single-use 20-sensors catheter (MMS G-90500). Catheter lumens were perfused with distilled, degassed water at a rate of 0.1 ml/min. All the lumens were connected to a pressure transducer (DTX Plus TNF-R). The manometric signals were visualized as isobaric contour plots on a dedicated screen and were stored in a computer using a commercial software (MMS Investigation and Diagnostic Software version 8.19 build 2188) for subsequent analysis.
Study design
After an overnight fast, the oesophageal manometry catheter was passed transnasally under topical anaesthesia (Lidocaine spray 10%). The catheter was positioned so that side holes spaced 1 cm apart straddled the lower oesophageal sphyncter. The test was performed in the recumbent right-side position. A second catheter (10 F, Gastric Sump Tube; Medovations, USA) was inserted and the tip positioned at 30 cm from the nares, in order to perform aspiration and to infuse air.
Each test started with rapid (<1 s) 20-ml air injection at mid-oesophageal level, using the second catheter. Five infusions was performed, each one separated by 30 s. Eight 5-ml water swallows were used to evaluate primary peristalsis.
Data analysis
Oesophageal motor response to swallowing and distension
Motor activity occurring after both swallowing and distension was characterized measuring the distal contractile integral (DCI) and contractile front velocity (CFV) and classified as intact, weak, simultaneous, or failed according to the Chicago classification.
9
Lower oesophageal sphincter relaxation was evaluated measuring 4 s integrated relaxation pressure (4 s IRP). In achalasia patients without restored peristalsis, DCI and CFV could not be measured; in those having panoesophageal pressurization in response to water swallowing or air infusion (
According to Schoeman et al.,
2
a motor response to an air bolus or water swallowing was considered to have occurred if a phasic pressure rise (i.e. a pressure wave propagated or simultaneous or a panoesophageal pressurization) of >10 mmHg within 20 s has occurred. Hence, the response rates to water swallows and air infusions, defined as the ratio between number of motor responses and number of water swallows (
Statistical analysis
Results are presented as median (IQR). Swallow-induced and distension-induced oesophageal motor response were compared between each others and within the three study groups using non-parametric statistics. We used Wilcoxon–Mann–Whitney test for pairwise independent comparisons between groups and Wilcoxon signed-rank test for paired comparisons within groups. Correlations between quantitative variables (response rate to air infusion, oesophageal diameter, length of symptomatic disease, severity of dysphagia, age) were analysed using Spearman Rho correlation coefficient.
Results
All patients and healthy subjects completed the study protocol. The basal intraoesophageal pressure was negative in healthy subjects, NOD patients, and achalasia patients: –5.0 mmHg (IQR –7.0 to −3.2 mmHg), −5.5 mmHg (IQR −8.0 to −4.0 mmHg), and −6.0 mmHg (IQR −8.0 to 4.0 mmHg) respectively (NOD vs. healthy subjects
Motor response to swallowing and distension
Oesophageal motility variables in the three groups after water swallowing and air infusions
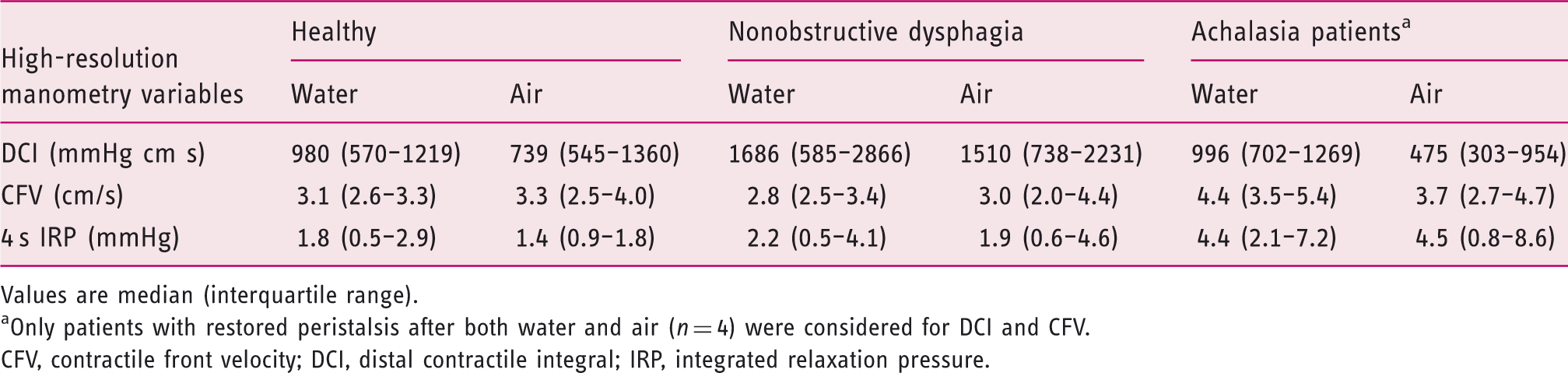
Values are median (interquartile range).
Only patients with restored peristalsis after both water and air (
CFV, contractile front velocity; DCI, distal contractile integral; IRP, integrated relaxation pressure.
Similarly to healthy subjects, in NOD patients DCI, CFV, and 4 s IRP after water swallowing were similar to after air infusion (
With regards to the achalasia group, four of the six patients with restored peristalsis after water swallows showed peristalsis after air infusion and the remaining two showed panoesophageal pressurization. The former four patients had DCI after water swallowing similar to after air infusions: 996 mmHg cm s (IQR 702 to 1269 mmHg cm s) vs. 475 mmHg cm s (IQR 303 to 731 mmHg cm s;
Response rate to water swallowing
Response rate to water swallowing was 100% (IQR 100 to 100%) in healthy subjects and it was similar to response rate of both NOD and achalasia patients: 100% (IQR 100 to 100%) and 100% (IQR 87.5 to 100%), respectively; Response rates to water swallowing (a) air infusions (b) and ratio between response rate to air infusions and water swallowing (c) in healthy subjects (HS), nonobstructive dysphagia (NOD), and achalasia patients.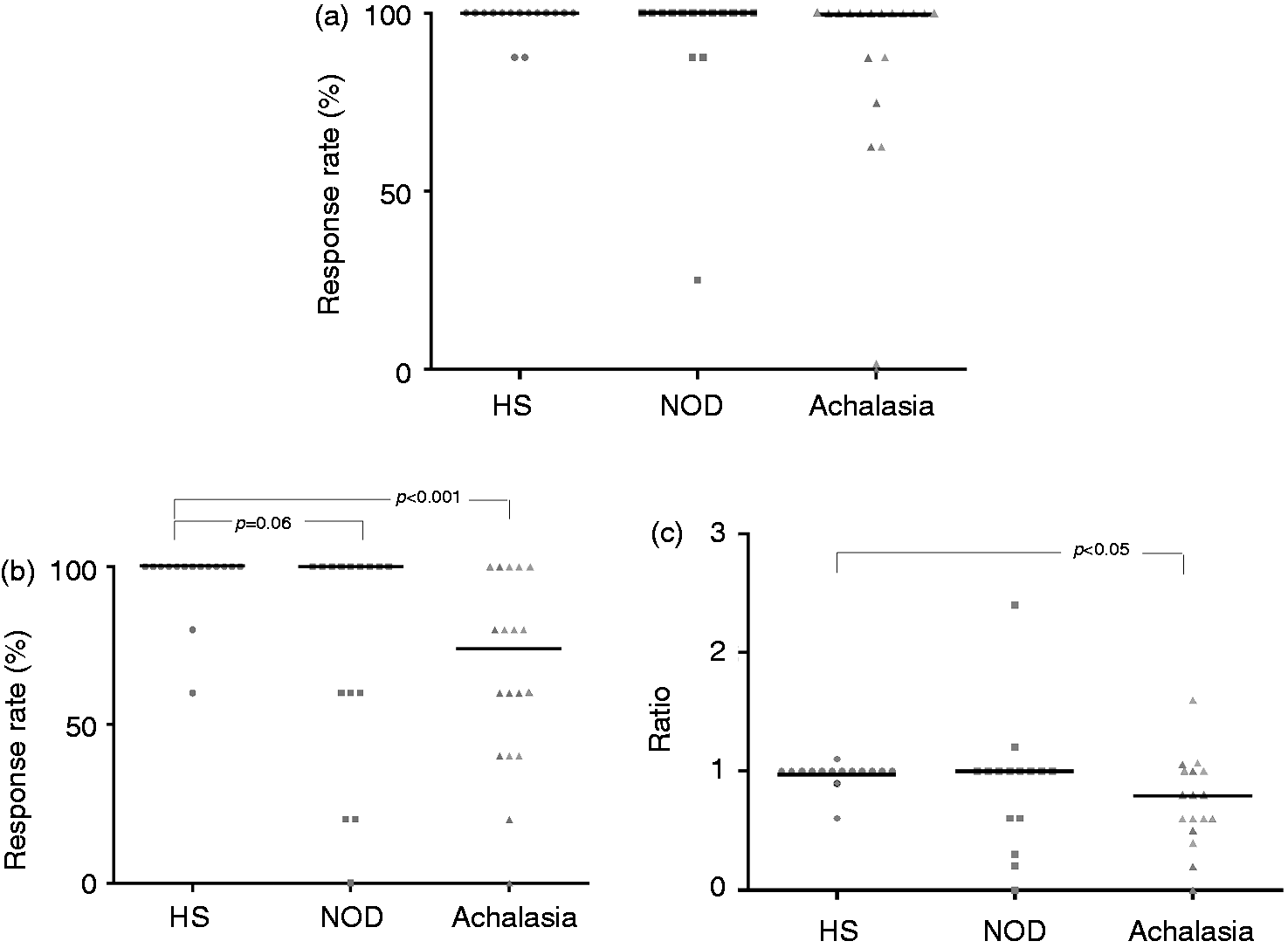
Response rate to air infusions
Response rate to air infusions was 100% (IQR 100 to 100%) in healthy subjects, it was slightly lower in NOD patients (100%, IQR 60 to 100%;
It is interesting to note a heterogeneous response among patients, especially in the achalasia group. Twelve of NOD and 13 of achalasia patients had a rate within the range observed in healthy subjects (>60%). No relationship was found between response rate and oesophageal diameter, duration or frequency of dysphagia, or age in both patients groups. Separating patients according to the post-swallow manometric pattern, the three NOD patients with normal peristalsis had a response rate within the range observed in healthy subjects whereas seven of the 10 patients with the most severe motility disorders (i.e. large breaks or frequent failed peristalsis) had normal response rate and three had low response rate. In the achalasia group, focusing on patients with type 2 disease, the response rate was similar in those with restored peristalsis compared to the ones with panoesophageal pressurization (
Ratio between response rate to air infusions and water swallowing
This variable was used in order to evaluate if impairment of triggering a motor response was selective for the stimulus of rapid air infusion. The type 1 achalasia patient with no motor response to water swallowing was excluded from this analysis. The ratio between response rate to air infusions and water swallowing was 1.0 (IQR 1.0 to 1.0) in healthy subjects, 1.0 (IQR 0.6 to 1.0) in NOD patients ( Examples of water swallow and air infusion in a healthy subject (a) and a type 2 achalasia patient.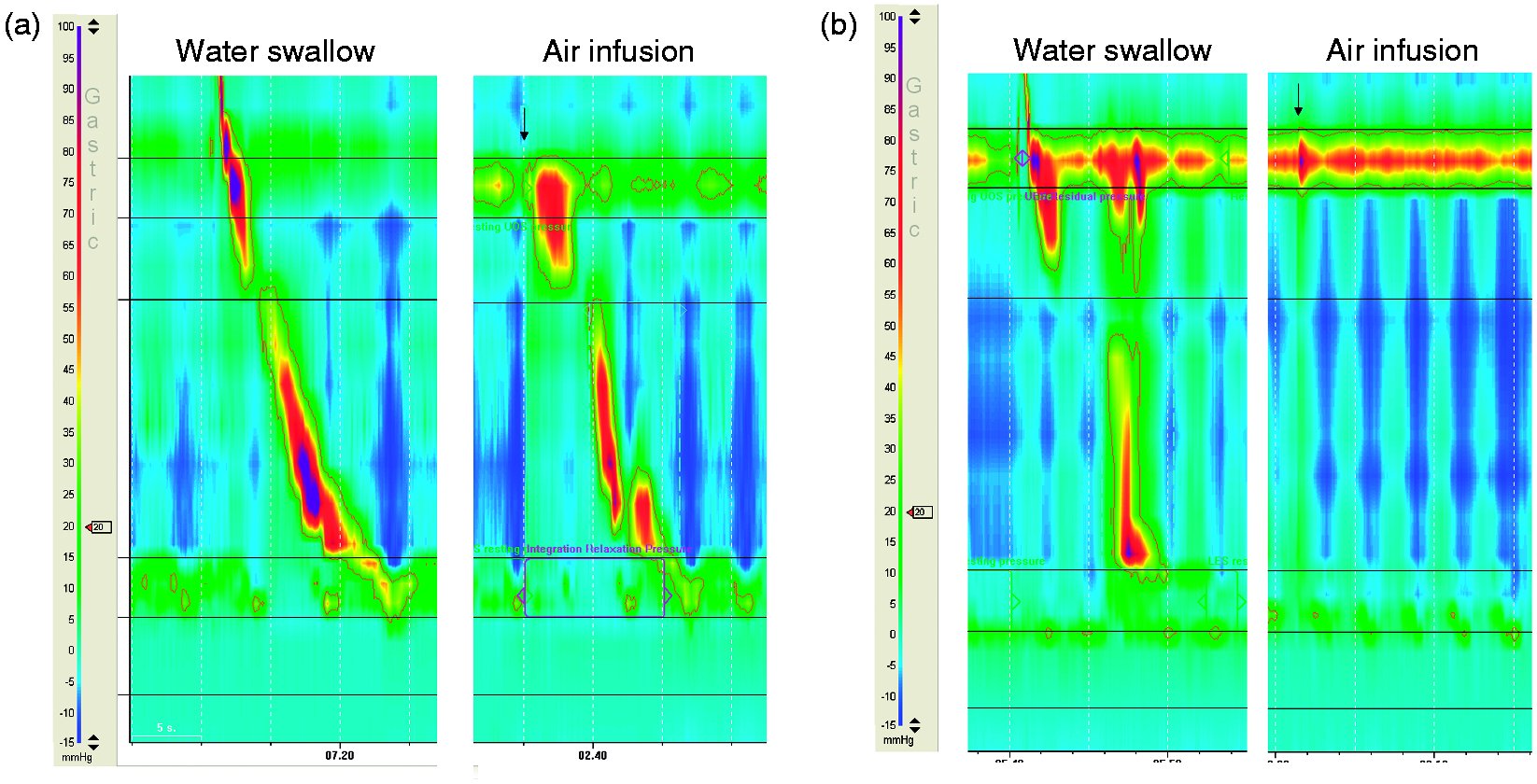
Types of response to water swallows and air infusions
Both healthy subjects and NOD patients generally had intact or weak contractions after both air infusions and water swallowing ( Type of motor response (intact, weak, simultaneous, and failed contractions) to water swallowing and air infusions in healthy subjects (HS) and nonobstructive dysphagia (NOD) patients.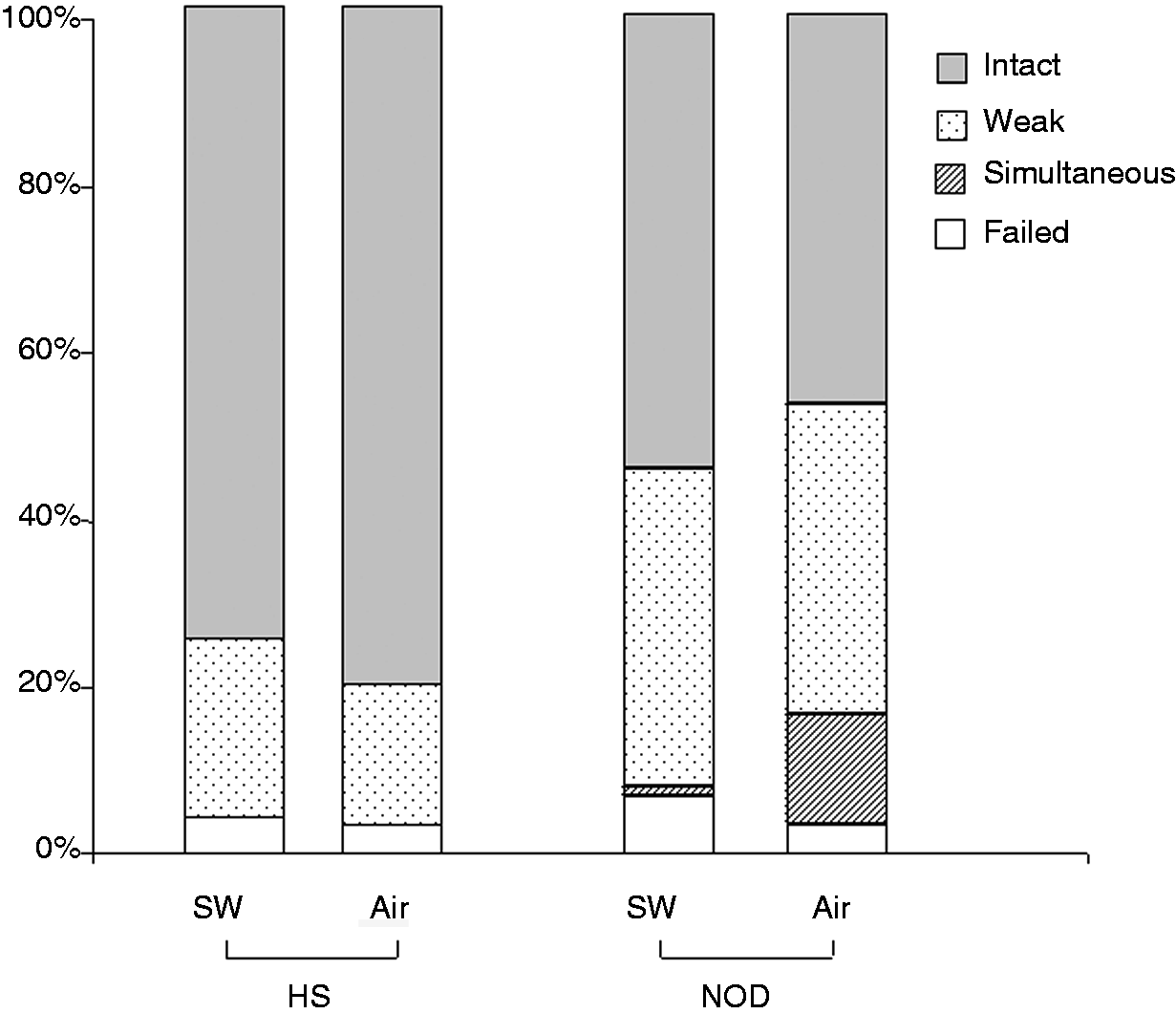
Discussion
Our study has shown that achalasia patients have a lower motor response rate to rapid air infusion into the oesophagus compared to healthy subjects. Furthermore, the motor response reached a maximal pressure lower than after water swallowing. Patients with NOD had only a slightly impaired motor response to rapid air infusion.
The motor response induced by oesophageal distension 11 is a result of a neural reflex: mechanoreceptors, located in the oesophageal mucosa and muscular wall, within the myenteric plexus, are stimulated by oesophageal distension, which is followed by the activation of oesophageal motility. Some authors suggest that the motor response to oesophageal distension follows the same neural pathways of primary peristalsis, 4 while others suggest that it is an intramural mechanism, without any central control. 12
To our knowledge, achalasia patients have not been previously investigated with regards to distension-induced motility. These patients have a well-documented neuropathy (i.e. impairment of inhibitory neural pathways due to degeneration of inhibitory ganglion cells in the myenteric plexus of the oesophagus and degenerative lesions in the vagal nerves and the dorsal motor nucleus). 5,13 –16
Studying oesophageal motility induced by rapid air infusion poses a few challenges in these patients before miotomy or pneumatic dilation is performed. On one hand, the oesophageal body is variably dilated before treatment, making it difficult to standardize distending stimuli; on the other, traditional quantitative variables of motility (i.e. DCI and CFV) can not be applied to achalasia patients because the majority of them only show contraction of the longitudinal muscle layer. 17,18 This is why we studied patients after successful pneumatic dilation who had a non dilated empty oesophagus. Furthermore, when peristalsis was not restored, maximal intraoesophageal pressure was used as a quantitative index of motor response.
Overall, achalasia patients had an impaired response to rapid air infusion. On one hand, their maximal pressure was lower than after swallowing, contrary to what occurred in healthy subjects and NOD patients, where the strength of motor response was similar to after swallowing; on the other, and more importantly, the response rate was lower than in healthy subjects. However, it is interesting to observe a wide range of response rates among these patients independently of presence of restored peristalsis, two-thirds of them having >60% response rate. This may suggest that sensory neural pathways are involved to a variable degree in the neurodegenerative process.
We could not find any relationship between response rate and morphological, clinical, or demographic variables, although our series was relatively small. The observed variable impairment of triggering a distension-induced motor response raises a number of issues, which could need to be addressed in further studies on larger cohorts of patients involving a sufficient number of type 1 and type 3 achalasia as well: first, it could suggest pathophysiological heterogeneity among achalasia patients; secondly, presence of impairment could be a factor predicting a worse outcome of treatment, in view of the evidence that need of pneumatic dilation is quite variable among patients; 19,20 and thirdly, distension-induced motor response may not be stable, but worsen with time.
NOD patients are a heterogeneous group and a variable damage in both inhibitory and excitatory neural pathways has been hypothesized. This is why we have included only NOD patients with normal or weak peristalsis according to the Chicago classification, 9 thus excluding patients with an hypercontractile disorder, in order to decrease pathophysiological heterogeneity. Previous studies have suggested that, in NOD patients with ineffective motility, neuromuscular structures are at least partially preserved and the main nerve damage is at the neural cholinergic stimulation level. 21,22
Our data have shown that a minority of patients have impaired triggering of distension-induced motor response and all of these patients had already evidence of severely altered motility when tested with water swallows. Similarly to what has been discussed with achalasia patients, this alteration could identify a subset of patients with different pathophysiological features, hypothesis to be addressed with a larger cohort of patients.
Schoeman et al. 3 looked at motor response induced by rapid air injection in NOD patients using traditional oesophageal manometry; they observed that primary peristalsis detected an abnormality in 63% of patients, but when distension-induced motor response was also considered, the proportion of patients with abnormal oesophageal motor function increased to 80%. Our study showed a lower contribution of rapid air infusion to establishing altered oesophageal motility; however, our series was different as it included no patients with diffuse oesophageal spasm and only a few with normal swallow-induced motility.
Finally, it could be argued that the younger age of our healthy subjects may have been a source of bias. We are, however, confident that age was not a major variable determining the differences observed among groups. Achalasia patients had the most severe impairment of triggering distension-induced motor response, but the age was similar to NOD patients. Furthermore, in both patients’ groups, the age range was wide, spanning 50 years, and no relationship was found between response rate to air infusion and age.
In conclusion, triggering of motor response by rapid air infusion was variably impaired in achalasia patients. These data should stimulate further prospective studies in larger cohorts in order to evaluate whether alteration of this function has a role in the long-term outcome of achalasia patients.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors declare that there is no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
