Abstract
Background
Achalasia is a primary oesophageal motility disorder. Although aetiology remains mainly unknown, a genetic risk variant, rs28688207 in HLA-DQB1, showed strong achalasia association suggesting involvement of immune-mediated processes in the pathogenesis. High-resolution manometry recognises three types of achalasia. The aim of our study was to perform the first genotype-phenotype analysis investigating the frequency of rs28688207 across the high-resolution manometry subtypes.
Methods
This was a cross-sectional retrospective study. Achalasia patients from tertiary centres in the Czech Republic (
Results
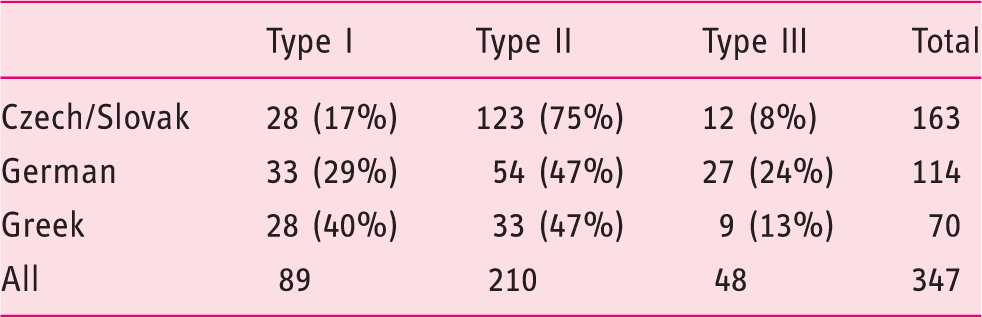
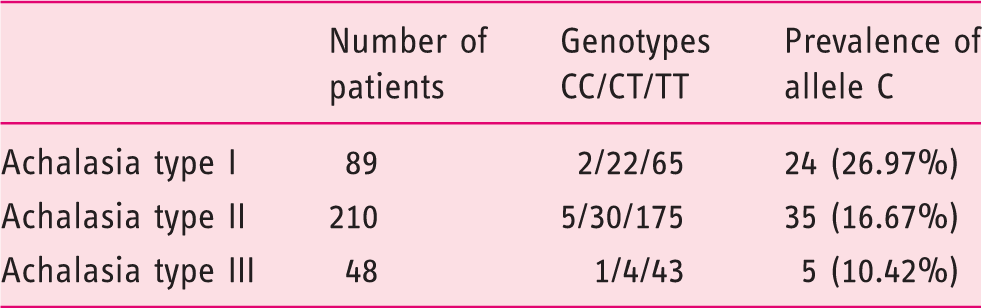
A total of 347 achalasia patients (type I – 89, II – 210, III – 48) were included. The overall frequency of the rs28688207 was 10.3%. The distribution of the insertion was significantly different across the high-resolution manometry subtypes (
Conclusion
The frequency of the HLA-DQB1 insertion differs among high-resolution manometry achalasia subtypes. The insertion is most prevalent in type I, suggesting that immune-mediated mechanisms triggered by the insertion may play a more prominent role in the pathogenesis of this subtype.
Key Summary
1. Summarise the established knowledge on this subject • Autoimmune mechanisms are involved in the pathogenesis of achalasia. • The genetic risk variant rs28688207 in HLA-DQB1 is associated with achalasia. • Achalasia presents in three different manometric subtypes. 2. What are the significant and/or new findings of this study? • The frequency of the HLA-DQB1 risk variant differs among the three manometric subtypes of achalasia. • The different frequency among the high-resolution manometry subtypes might suggest a different extent of immune-mediated processes involvement in the pathogenesis.
Introduction
Idiopathic achalasia is a rare motility disorder of the oesophagus with a lifetime prevalence of 1:10,000. 1 It is characterised by impaired deglutitive relaxation of the lower oesophageal sphincter (LES) and disturbed peristalsis of the oesophagus. Clinically, achalasia presents as dysphagia, regurgitation of ingested food and chest pain. The motility dysfunction is due to the loss of inhibitory neurons in the myenteric plexus within the muscular layer of the oesophageal wall, which causes the excitatory neurons to outbalance. 2 However, histopathologic studies have demonstrated heterogeneity in the extent of neuronal loss among achalasia patients ranging from mild reduction to complete agangliosis, as well as in other histopathologic features such as grade of inflammatory infiltration and fibrosis.3–7 This heterogeneity is also reflected in the manometric findings and in the high-resolution manometry (HRM) classification of motility disorders, the Chicago Classification (CC), which distinguishes three distinct manometric subtypes (I–III) of achalasia.8,9 Moreover, the HRM subtypes of achalasia also vary in clinical presentation and responsiveness to treatment, 10 thus representing different phenotypes of the disease.
On the aetiological level, achalasia is a multifactorial disease where environmental and genetic factors determine disease presentation. 1 Recently, a relatively penetrant genetic risk variant for achalasia has been identified. It is, so far, the most significant achalasia-associated risk variant and is represented by the single nucleotide polymorphism (SNP) rs28688207 in the gene HLA-DQB1 on chromosome 6. 11 The variant leads to an alternative splicing of exon 5 of HLA-DQB1 and thereby to an eight-amino-acid insertion in the cytoplasmic tail of HLA-DQβ1. 12 On the protein level HLA-DQβ1 forms a subunit of the HLA-DQ receptor, which is present on the surface of antigen-presenting cells (APCs). 12 This finding supports the hypothesis that immune-mediated processes are involved in the pathogenesis of the disease. Furthermore, the HLA-DQβ1 insertion showed different frequencies among European populations with a geospatial north-south gradient. 13
The main aim of the present study was to perform a genotype-phenotype (GxP) analysis testing whether the frequency of the HLA-DQβ1 insertion differs among HRM subtypes of achalasia (as defined by the CC). Since our study comprised achalasia samples from populations in Central and Southern Europe, that have not been analysed so far, we also intended to replicate the achalasia association of the HLA-DQβ1 insertion in independent case-control samples and to investigate whether the HLA-DQβ1 insertion indeed displays a geospatial north-south gradient among Europeans.
Material and methods
Study design
This was a retrospective, multicentre, population-based, cross-sectional, genotype-phenotype association study analysing frequency of the HLA-DQB1 variant in achalasia patients with regard to the HRM subtype.
Patients
Between March 2015–August 2016 patients aged over 18 years with idiopathic achalasia referred to tertiary centres in the Czech Republic (Department of Hepatogastroenterology, Institute for Clinical and Experimental Medicine, Prague, Department of Internal Medicine, University Hospital Plzen, Department of Gastroenterology, Hospital Kolin), Slovak Republic (University Hospital of Trnava, 1st Department of Internal Medicine, Louis Pasteur University Hospital, Kosice), Germany (Department of Visceral, Transplant, Thoracic and Vascular Surgery, University Hospital of Leipzig) and Greece (Foregut Surgery Department, 1st Propaedeutic Surgical Clinic, Hippokration General Hospital of Athens) were offered to participate in the study. The diagnosis of achalasia was based on HRM, timed barium oesophagogram (TBE) and upper gastrointestinal (GI) endoscopy findings. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a prior approval by the institution's human research committee. The study was approved by the Ethical Committee of Institute for Clinical and Experimental Medicine and Thomayer's Hospital on the 12 March 2015 and all participants signed an informed consent form.
To test whether the frequency of the HLA-DQβ1 insertion displays a geospatial north-south gradient among Europeans, we excluded the samples from Germany, since most of the patients were already included in our initial study. 13 In addition, we used control samples from the Czech/Slovak Republic and Greece in order to confirm the achalasia association of the HLA-DQβ1 insertion in both populations. The Czech/Slovak control group was enrolled from consecutive blood donors from the blood bank at Thomayer's Hospital in Prague and included 221 (75 females, 146 males). As for Greek controls we used 594 population-based samples (300 females, 294 males) from the THISEAS (The Hellenic study of Interactions between Single nucleotide polymorphisms and Eating in Atherosclerosis Susceptibility) cohort. 14 This sample has been originally enrolled to study demographic and clinical characteristics as well as biochemical indices and lifestyle habits of the population in order to identify risk factors for coronary artery disease.
HRM
All patients with achalasia underwent the oesophageal HRM. The ManoScan 360 High-Resolution Manometry device by Given Imaging with a solid-state catheter equipped with 36 circumferential sensors spaced 1 cm apart was used at all participating centres. A standardised examination protocol of 10 wet swallows of 5 ml of water in the supine position was applied. All HRM studies were analysed manually by experienced physicians using ManoView analysis software (Given Imaging, Los Angeles, California, USA) and achalasia subtype was determined based on the criteria postulated in the CC v3.0.15 In HRM metrics, the impaired oesophago-gastric junction (EGJ) relaxation, which is a hallmark of achalasia, is characterised by elevated median integrated relaxation pressure (IRP) > 15 mm Hg, for type I achalasia the cut-off was set at 10 mm Hg as suggested in the current version of CC. Besides the elevated IRP, type I achalasia was defined by 100% failed peristalsis, type II achalasia by 100% failed peristalsis and pan-oesophageal pressurization of ≥ 30 mm Hg in ≥ 20% of swallows (if the pressurizations were 15–29 mm Hg the patient was classified as having type I achalasia) and type III achalasia by presence of spastic contractions in ≥ 20% of swallows.
Genotyping and imputation
Whereas the genotypes of rs28688207 (encoding for the insertion in HLA-DQβ1) have been obtained previously from German patients, 13 within the present study rs28688207 was genotyped in cases and controls from the Czech/Slovak Republic and from Greece using a custom designed TaqMan Genotyping Assay (Applied Biosystems). For the Greek THISEAScontrol cohort we used genome-wide genotyped SNPs 14 and information of linkage disequilibrium (LD) between SNPs from a reference cohort. Although rs28688207 was not directly genotyped in the THISEAS sample, we imputed this variant into the Greek control cohort using IMPUTE2 16 and 1000 Genomes 17 as reference samples. For the case-control comparison in the Greek sample we only used genotypes from rs28688207 with an InfoScore of > 0.99. Previously, we have demonstrated that the imputation of the insertion shows a high accuracy (concordance rate = 99.7%). 11
Biostatistics
For the GxP analysis we compared all three achalasia HRM subtypes with regard to carrying the risk allele of rs28688207 using the Kruskal–Wallis test. The Student's
To test whether the risk allele of rs28688207 displays a geospatial north-south gradient among Europeans we used Armitage trend test for comparison of genotype distribution in patients and controls in each population.
Results
Patients' characteristics and HRM results
Distribution of achalasia high-resolution manometry (HRM) subtypes in the studied populations.
GxP analysis
Allele C (rs28688207) distribution in high-resolution manometry (HRM) subtypes of achalasia.
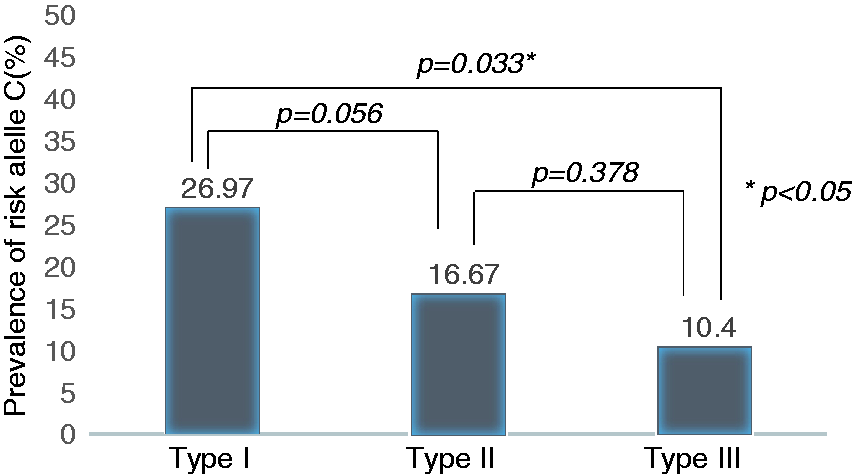
Association of the risk allele C (carrying the HLA-DQβ1 insertion) with high-resolution manometry (HRM)-subtypes of achalasia.
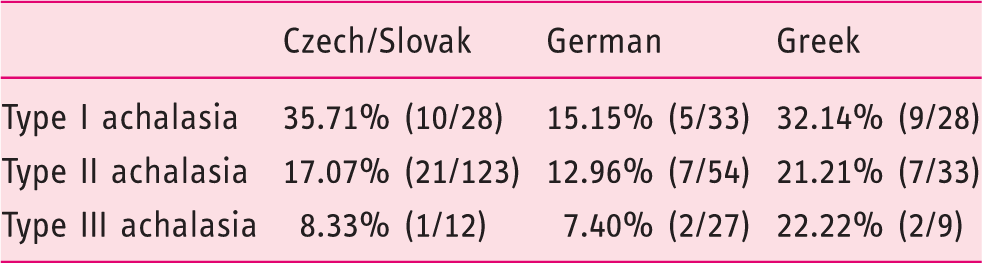
Prevalence of risk allele C (carrying the HLA-DQβ1 insertion) in each high-resolution manometry subtype of achalasia across the studied populations.
Replication and population frequency
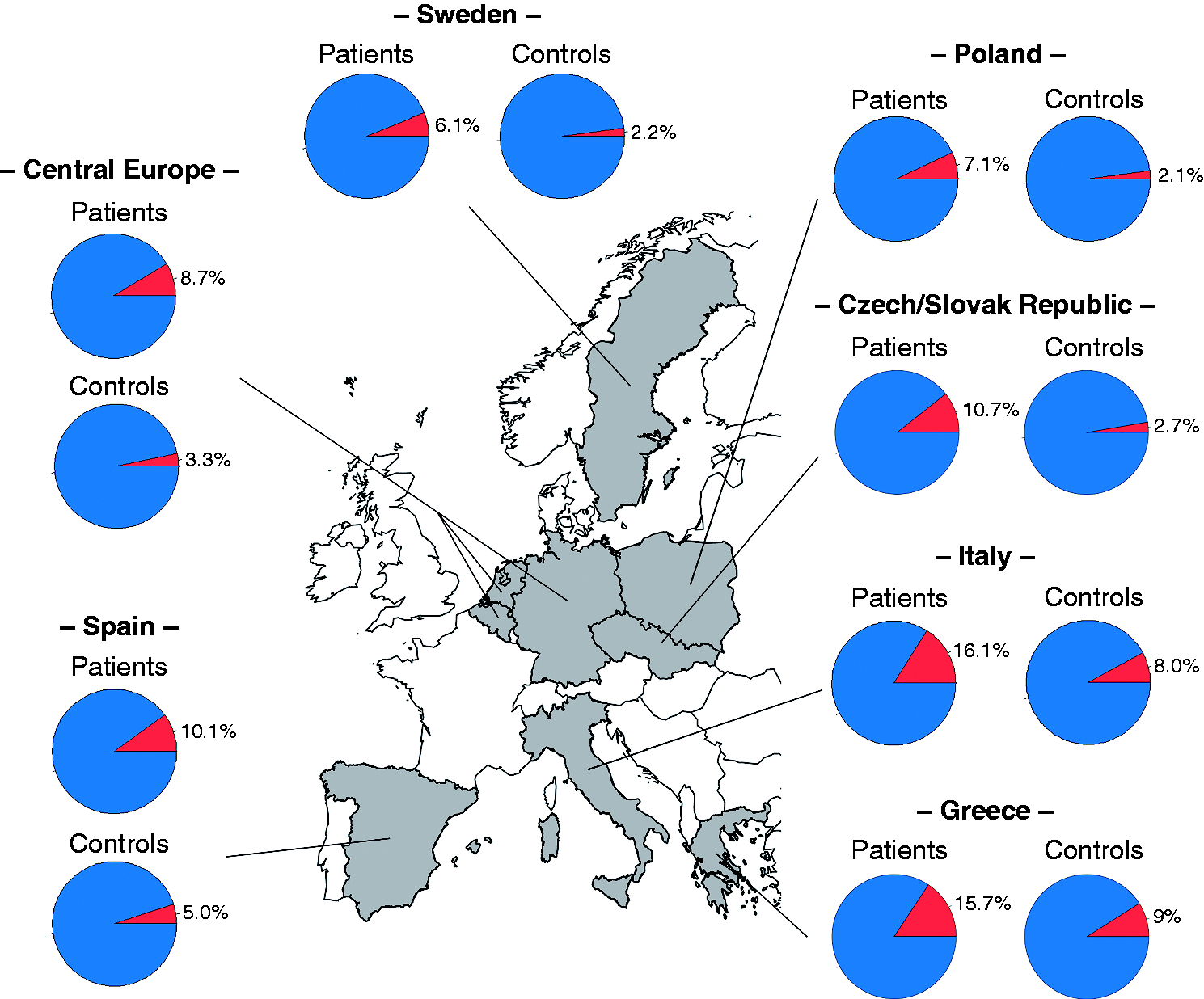
As expected, analysis of rs28688207 in the Czech/Slovak case-control sample confirmed a significant association of allele C with achalasia ( Geospatial north-south gradient of the frequency of the HLA-DQβ1 insertion in the studied populations and other European countries as has been published previously.
13
Discussion
We present the first GxP association study in achalasia based on the recent discovery of the strongest known genetic risk variant rs28688207 located in HLA-DQB1 region 11 and the HRM classification of achalasia into three distinct subtypes.
The GxP analysis revealed statistically significant frequency differences of the HLA-DQβ1 insertion among the HRM-subtypes of achalasia. We have shown that patients with type I are significantly more affected by the insertion compared with type III and also type II (here the statistical significance was borderline). Our findings suggest, that in type I achalasia, which is manometrically characterised by complete absence of oesophageal contractility as a consequence of myenteric agangliosis 19 and clinically represents a more severe or progressed phenotype of the disease, the insertion may play a more prominent role in the pathogenesis.
At the molecular level the HLA-DQβ1 insertion causes a structural alteration to the cytoplasmic tail of the HLA-DQ receptor. The HLA-DQ as a MHC class II molecule displays antigens on the surface of antigen-presenting cells (APCs) which leads to activation of CD4+ T lymphocytes and initiates the adaptive (both cell- and antibody-mediated) immune response. 19 Moreover, the cytoplasmic tail (CT) of the HLA-DQ receptor is involved in controlling multiple aspects of the immune response such as intracellular trafficking and activation of certain signalling pathways. 20 In carriers of the rs28688207 risk allele, the HLA-DQβ1 insertion represents a connecting peptide motif that drives the intracellular cyclic adenosine monophosphate (cAMP) signalling pathway, which is essential for activation of B-cells and antibody production in the context of immune response. 20 In contrast, in non-carriers the cAMP-signalling motif is replaced and the cAMP-signalling pathway is impaired. 20 Thus, individuals carrying the HLA-DQβ1 insertion might have, hypothetically, an enhanced immune response compared with non-carriers.
As the frequency of the risk allele varies among the achalasia subtypes, the intensity of the immune response may vary too. This hypothesis is supported by findings of several (but sparse) pathological studies that have shown that the extent of typical histopathologic features found in muscularis propria specimens from achalasia patients, such as inflammatory infiltration, depletion of ganglion cells and fibrosis is not uniform in all cases.3,4,21–23 Goldblum et al. reported that the loss of myenteric neurons in patients with unresponsive end-stage achalasia (corresponding to HRM type I) is nearly complete. 3 Moreover, the same authors showed in a small study of myotomy specimens from 11 patients with early-stage achalasia that in six out of 11 patients ganglion cells, although markedly diminished, were present. And maybe more interestingly in all three patients with ‘vigorous’ achalasia (corresponding to HRM type III) that were in this study the number of ganglion cells was within the normal range. 4 In addition, Sodikoff et al. analysed the heterogeneity of histopathologic findings in the context of manometric subtypes and showed that there was a significantly greater proportion of agangliosis as well as a greater degree of ganglion cell loss in the muscularis propria specimens from type I achalasia patients compared with type II. 23 Taken together, type I achalasia might represent a more severe phenotype where progression of the destructive autoimmune assault to the myenteric plexus seems to be accelerated and – together with other factors – this might be triggered by increased immune response due to the HLA-DQβ1 insertion.
We may further speculate that in patients with a more severe phenotype (achalasia I, especially patients carrying the insertion), altered immune response may trigger increased inducible nitric oxide synthase (iNOS) expression, which may lead to high levels of nitric oxide (NO) and eventually contribute to impairment of the normal function of nitrergic neurons accelerating their selective degeneration. As it has been shown that the functional polymorphism (CCTTT) of NOS2 promoter may be associated with achalasia, likely by an allele-specific modulation of NO production. 25
In addition, our study confirmed that the HLA-DQβ1 insertion displays a geospatial north-south gradient among Europeans. Similar gradients have been already observed for other immune-mediated diseases (such as Crohn's disease, Type 1 diabetes or IgA nephropathy) where genetic variation at the HLA locus has an important role in disease pathogenesis.24,25 The reasons for such latitudinal gradients are most probably evolutionary mechanisms such as genetic drift or natural selection in the context of different environmental conditions.
There are two main limitations of our study. Firstly, although the association of the HLA-DQB1 insertion and achalasia has been confirmed, the insertion itself cannot explain the pathogenesis of the disease (the majority of achalasia patients do not carry the insertion). Nevertheless, the insertion might play an influential pathogenetic role in at least some cases. Our study mainly focused on revealing whether the insertion frequency has a predominance in any of the manometric subtypes of achalasia. Even though a clear clinical (as well as manometrical) distinction, especially between type I and II, is sometimes disputable, an ongoing hypothesis is that type I is evolving from type II, while it is the type III that might be distinct. Our results did not particularly follow such a hypothesis but showed a distinction between type I and type III, and not between type II and III with regard to the insertion frequency. On the other hand, only a small number of patients with type III achalasia were included which might have caused an erroneous estimation of the frequency of the risk allele in this type. The second limitation is the lack of detailed phenotypic characteristics regarding the clinical course of the disease, especially the age of the first manifestation and disease duration in the studied subjects as these data were insufficient in a considerable number of patients (a retrospective study). In our study we only found that the patients with type I were significantly younger than patients with types II and III, but we did not confirm such an association when comparing the carriers and non-carriers of the risk allele within the group of one manometric subtype of achalasia. Precise clinical data would be valuable in characterising the disease progression across the different genotypes, although in a previous study by Becker et al. multiple disease characteristics have already been tested with regard to the genetic risk variant and it was only pregnancy at the time of achalasia onset that was revealed to be more common in female risk allele carriers of rs28688207 compared with nonrisk carriers. 18
In conclusion, our study confirms the HLA-DQβ1 insertion to be a strong genetic risk factor for idiopathic achalasia and for the first time we have shown that the frequency of this insertion differs among achalasia HRM subtypes, being most prevalent in achalasia type I. Nevertheless, the exact role of the genetic risk variant in altering the immune processes engaged in pathogenetic pathways of achalasia still awaits to be elucidated.
Footnotes
Declaration of conflicting interests
The authors have no financial conflicts of interest to disclose.
Ethics approval
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a prior approval by the institution's human research committee. The study was approved by the Ethical Committee of Institute for Clinical and Experimental Medicine and Thomayer's Hospital on the 12 March 2015.
Funding
No special funding was received, the costs were covered from the clinics' budgets.
Informed consent
All participants signed an informed consent form before entering the study.
