Abstract
Midline peripheral intravenous catheters are widely used for long-term peripheral venous access due to ease of placement and a lower complication profile than central lines. To date, gas embolism has not been reported in association with midline catheters. We report the case of a 59-year-old man with COVID-19-related acute respiratory distress syndrome, who developed sudden coma and acute hypoxemia. Bedsides, echography revealed gas embolism in all cardiac chambers, and body-CT scan showed cerebral air emboli and hepatic portal venous gas. The event followed disconnection of a midline catheter infusion set while the patient was spontaneously breathing, hypovolemic and seated. Neurological sequelae persisted despite prompt hyperbaric oxygen therapy and supportive care. This is the first reported case of cerebral gas embolism related to a midline catheter. The case highlights the importance of individualized catheter length, secure line fixation, and risk factor awareness to prevent air embolism, even with peripheral lines.
Keywords
Introduction
Midline peripheral intravenous catheters are increasingly utilized worldwide for long-term peripheral vascular access. 1 Their growing popularity is due to easier placement (often under ultrasound guidance), reduced complication rates, and decreased reliance on central venous catheters (CVCs), which are associated with serious risks including gas embolism.2,3
To date, no cases of gas embolism have been linked to midline catheter use.1,2,4 Although rare (The estimated incidence of iatrogenic gas embolism is ~2.65/100,000 hospital admissions. 5 ), gas embolism is a potentially fatal complication6,7 involving the entry of gas bubbles into the bloodstream via venous or arterial access. 8
Clinical manifestations can vary widely, ranging from asymptomatic cases or subtle neurological deficits to life-threatening hemodynamic instability. 9 Among survivors, severe neurological or systemic sequelae are reported in 9%–35% of cases.8,10 Mortality rates can reach up to 42% when cerebral embolism occurs, 10 underscoring the need for preventive strategies even in peripheral catheter management. In fact, early recognition and aggressive management seems to significantly influence prognosis and functional outcomes, 11 highlighting the critical need for timely diagnosis in high-risk patients. 9
We present the first documented case of paradoxical cerebral gas embolism associated with a midline catheter in a hypovolemic ICU patient.
Case presentation
A 59-year-old man (Mr. J.) with a history of Poppema-type paragranuloma requiring rituximab infusions was admitted to the ICU for severe COVID-19 pneumonia complicated by acute respiratory distress syndrome, requiring mechanical ventilation.
During his ICU stay, the patient developed ventilator-associated pneumonia (Enterobacter cloacae, Escherichia coli, and methicillin sensitive staphylococcus aureus) and experienced an ischemic cerebellar stroke. Due to prolonged weaning, a tracheostomy was performed.
Spontaneous ventilation through a tracheostomy hygroscopic condenser humdifier was initiated 18 days after admission. A few days later, the patient was weaned from mechanical ventilation and continued breathing through a cuff-deflated tracheostomy cannula.
After 21 days in ICU, the CVC was removed and replaced with a 20 cm midline catheter inserted in the mid-arm.
At day 25, in the morning, patient was described as mildly polypneic and confused. A ventilator-associated pneumonia was suspected and antibiotic therapy was initiated.
At day 25, at 12:00, during the nurse’s visit, patient was seated in a chair, fully alert (Richmond Agitation Sedation Scale score 0), hemodynamically stable without vasopressor support, and receiving low-flow oxygen therapy. He was always being monitored.
On day 25, at 14:00, while seated and breathing spontaneously, the patient developed sudden coma and severe hypoxemia with sudden drop in oxygen saturation at 26% on the monitor. He became cyanotic and tachycardic, hypotensive with signs of right heart failure and mottled skin. Alerted by the monitor’s alarms, the patient was immediately taken care. The infusion line was found disconnected from the proximal end of the midline catheter.
Emergency cardiac ultrasound revealed gas bubbles in all four heart chambers. The patient was sedated, re-intubated, and mechanically ventilated with protective strategies and neuromuscular blockade. Norepinephrine was initiated.
CT imaging in emergency, revealed Multiple small gas emboli in the left temporo-parietal brain cortex. It also highlighted pneumomediastinum, pneumopericardium, and COVID-19 sequelae (ground-glass opacities, architectural distortion). Above all, the CT revealed a significant hepatic portal venous gas.
The patient was urgently transferred for hyperbaric oxygen therapy (HBOT), receiving three sessions. Following the first HBOT’s session, he developed generalized tonic–clonic seizures. EEG confirmed bifrontal focal status epilepticus, resolved with clonazepam twice 1 mg, then levetiracetam 1000 mg twice a day and deep sedation by midazolam between 0.08 and 0.12 mg/kg/h during 48 h.
Brain MRI showed new bilateral parieto-occipital and superior frontal cortical lesions not present on previous imaging.
Neurologically, the patient improved gradually. Seizures were controlled with clobazam 10 mg every 8 h and levetiracetam 1000 mg twice a day for a total of 7 days. Right hemiparesis (Medical Research Concil Scale 3/5 in the leg) and mild frontal syndrome persisted. Behavioral disturbances improved. Hemodynamic support was withdrawn.
Transthoracic and transesophageal echocardiography showed preserved left ventricular function, right heart dilatation, and no patent foramen ovale (PFO).
The patient was weaned from ventilation by day 33 but remained on 4 L/min oxygen via tracheostomy. CT follow-up showed progression to pulmonary fibrosis.
He was discharged to rehabilitation after 52 days of hospitalization in intensive care units.
Discussion
Gas embolism: Mechanisms and risks
Gas embolism refers to the obstruction of blood vessels by gas bubbles,2,3,8 most commonly introduced iatrogenically. It can occur via several routes 5 : Venous entry, typically resulting in pulmonary complications; Arterial access, which may lead to ischemic injury in end organs, or paradoxical embolism, where gas crosses from the venous to the arterial circulation through intracardiac or intrapulmonary shunts. The clinical manifestations of air infiltration into the vascular system seems to depend on the location, volume, and flow rate of injection, which determine the path taken by the air. 5 Beyond retrograde cerebral involvement, venous air emboli can migrate via the inferior vena cava to the liver or to the right heart and pulmonary circulation. Large gas volumes (>50 cm3) may exceed pulmonary clearance, causing a right ventricular “air lock” and resulting in decreased cardiac output, elevated central venous pressure, and systemic hypotension. Accumulated microbubbles can obstruct the pulmonary microvasculature, leading to vasoconstriction and increased pulmonary vascular resistance. Some bubbles may cross the pulmonary capillaries, causing arterial air embolism and organ ischemia. In the present case, a venous gas embolism, favored by the proximal disconnection of the infusion line, progressed to a paradoxical embolism despite the absence of a PFO diagnosed by the immediate presence of a large quantity of gas bubbles in all four heart chambers. This is likely due to transient right heart overload, leading to an intracardiac temporary right-to-left shunt. During an acute gas embolism, a sudden and transient reversal of the intra-atrial pressure gradient can functionally open the foramen ovale, allowing bubbles to pass from right to left cardiac chambers as observed on echocardiography. Once pressures normalize, detecting a PFO becomes more challenging, especially when left atrial pressure is elevated. this may explain the absence of PFO detection in subsequent examinations.
Furthermore, in this case of severe gas embolism, the pneumomediastinum arises from alveolar rupture due to acute pulmonary overdistension caused by massive intravascular gas load obstructing pulmonary capillaries and precipitating a pressure gradient across the alveolar-capillary membrane. Free air dissects centripetally along bronchovascular sheaths toward the pulmonary hilum—a process termed the “Macklin effect.” 12
Recent studies suggest that patient-specific variables and procedural characteristics may critically modulate clinical outcomes. Advanced computational modeling has demonstrated that microbubbles can interact with blood flow dynamics in a manner that exacerbates endothelial injury and initiates inflammatory cascades, 5 indicating that even low-volume air emboli may provoke disproportionately severe systemic responses—particularly in critically ill patients.
Predisposing factors identified
In our case, multiple risk factors were presents and can occur the occurrence of gas embolism. In first, the patient was in hypovolemia (tachycardia, ~10 kg recent weight loss after diuretics, BMI 16.78 kg/m2). Before the gas embolism accident, it was noted a tachypnea at 26 breaths/min with signs of increased work of breathing. Patient was in upright posture (seated position). The infusion line was unsecured, and we found it disconnected, maybe because of the patient’s agitation, allowed air entry. Finally, we noted a catheter malposition. In fact, the midline catheter was full length inserted (20 cm), possibly reaching central vein. The catheter tip was positioned within the subclavian vein, and we didn’t positioned an antireflux valve.
Negative intrathoracic pressure during spontaneous inspiration likely favored air entry. The patient’s clinical status, combined with technical factors, led to embolism.
Midline catheters and air embolism risk
Midlines are peripheral devices placed in upper arm veins with the tip ideally terminating at or just before the axillary vein. Their long subcutaneous path and narrow lumen (typically 4 Fr) theoretically reduce embolism risk. However, if mispositioned or poorly secured, they can exceptionally pose the same risks as central lines, particularly in vulnerable patients. To our knowledge, this is the first reported case of paradoxical cerebral gas embolism originating from a midline catheter.
Preventive measures and recommendations
To mitigate the risk of gas embolism in midline catheter use in our critical care unit, we decide in first to tailor catheter length to patient anatomy. We avoid full-length insertion without measurement and all midline input must be cut to put the extremity at the humero-brachial vein junction. We must ensure secure connection between the catheter and infusion tubing and add an extension tubing to increase external segment length. Finally, we consider anti-reflux devices, and we monitor for early signs of agitation and risk of disconnection with air ingress in spontaneously breathing, not only with hypovolemic patients.
Conclusion
Midline catheters are effective tools for long-term peripheral access and generally safer than central lines. However, they are not immune to serious and exceptional complications like gas embolism. This case underscores the importance of proper insertion technique, secure line connections, and clinical vigilance particularly in hypovolemic, spontaneously breathing ICU patients. Clinicians should ensure that midline catheter tips do not extend beyond the brachio-axillary junction, and protocols should address risks specific to this device class (Figures 1–5).
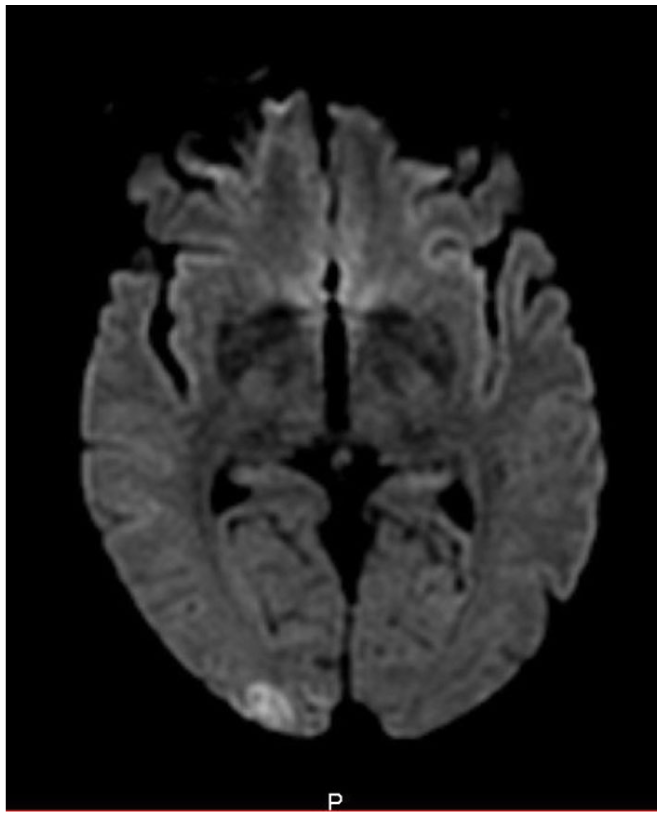
MRI showing multiple cortical lesions of the superior frontal and bilateral parieto-occipital regions – picture 1.

MRI showing multiples cortical lesions of the superior frontal and bilateral parieto-occipal regions – picture 2.
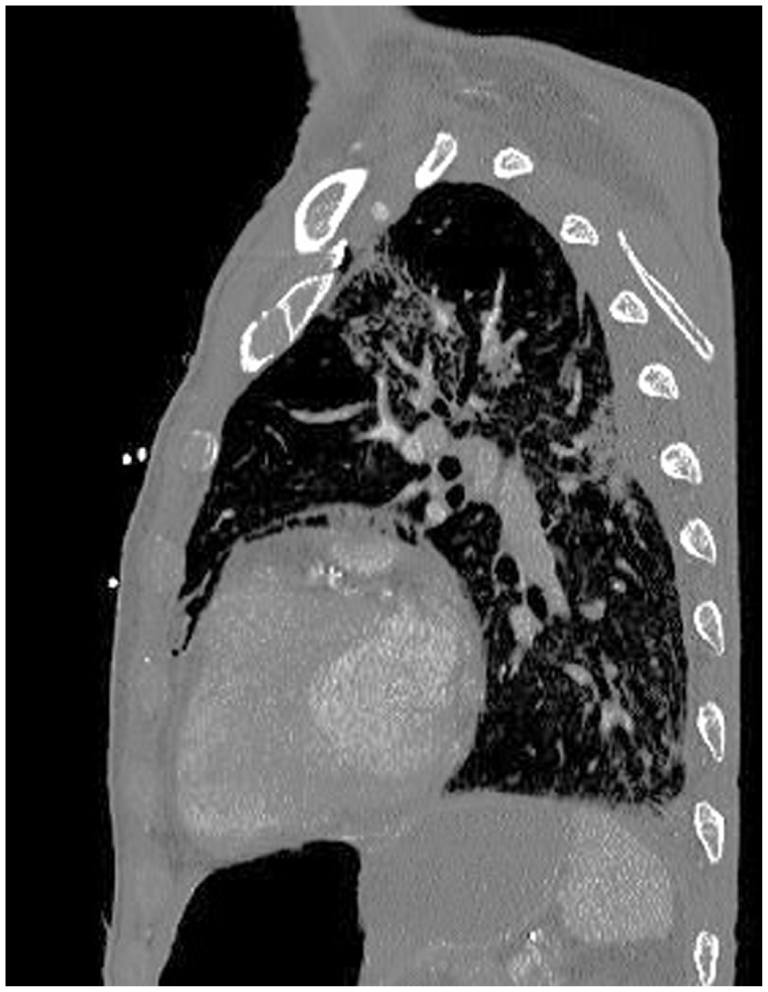
Computerized tomography scan showing pneumomediastin.

Computerized tomography scan showing hepatic portal veinous gas.

X-ray showing the midline in the left subclavian vein.
Footnotes
Consent for publication
Written informed consent for publication was obtained from the next of kin.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
