Abstract
Ataxias, a group of disorders characterized by impaired coordination, often lack effective treatments. Recent evidence suggests hyperbaric oxygen therapy (HBOT) may promote neuroplasticity and improve brain function. However, its impact on the core symptom of ataxia remains largely unknown. We present two cases of patients with chronic ataxia of different etiologies who received hyperbaric oxygen therapy. Our findings provide preliminary insights into the potential benefits of hyperbaric oxygen therapy for individuals with chronic ataxia. Two patients, aged 73 and 57, were presented with ataxia characterized by impaired static and dynamic balance, uncoordinated gait, and reliance on walking aids following stroke and brain injury, respectively. Pre and post-treatment evaluation included neuroimaging, gait analysis, and physical/functional assessment. Hyperbaric oxygen therapy treatment was administered for 5 days/week, consisting of 100% oxygen at two atmospheres of absolute pressure. Concurrently, twice-weekly physical therapy sessions were integrated into the treatment regimen. Post-treatment evaluation revealed significant improvements in gait, coordination, and balance, which correlated with perfusion changes in peri-lesional cerebellar tissue as measured by perfusion magnetic resonance imaging. This study presents a pioneering case series exploring the therapeutic potential of hyperbaric oxygen therapy in patients with cerebellar ataxia. Our findings suggest that hyperbaric oxygen therapy may enhance brain perfusion within the peri-lesional region, which correlates with neurophysical improvements. Given the novel nature of these findings, further randomized controlled trials with larger sample sizes and control groups are essential to validate and generalize these findings, ensuring a comprehensive understanding of hyperbaric oxygen therapy’s effects on various brain injuries.
Introduction
Ataxia, a neurological condition characterized by impaired coordination, represents a diverse group of disorders that can significantly impact an individual’s quality of life. While ataxia does not involve motor paralysis or altered consciousness, it markedly compromises voluntary movements, affecting their direction, force, and speed.1,2 This impairment can hinder an individual’s ability to perform daily tasks, leading to a functional challenge. The etiology of ataxia is multifaceted, including hereditary and acquired causes. The prevalence of acquired ataxia is noted to be higher than that of hereditary ataxia; cerebellar stroke 3 and traumatic brain injury 4 are significant causes of acquired ataxia encompassing a spectrum of pathologies. Understanding the diverse causes and manifestations of ataxia is crucial for accurate diagnosis and the development of targeted therapeutic interventions.
Ataxias, characterized by impaired coordination and balance, are broadly classified into cerebellar, proprioceptive, and vestibular categories based on the affected region. 2 Cerebellar ataxias often manifest with dysmetria, particularly hypermetria (exaggerated movements). 2 The lesion location within the cerebellum further dictates specific impairments: hemispheric lesions primarily affect coordination, vermis lesions lead to gait imbalance,1,2 and thalamic lesions can cause unilateral hemiataxia, potentially compounded by sensory deficits. 5 Proprioceptive ataxia, a neurological condition primarily linked to spinal cord diseases, is characterized by impaired coordination due to sensory deficits. This type of ataxia can also arise from lesions in the brainstem, thalamus, or basal nuclei and characterized by near-normal coordination when visual feedback is available. 2 Lastly, vestibular ataxia typically presents with vertigo, slow nystagmus with or without positional changes, and a tendency to veer toward the ipsilateral side during ambulation. These symptoms are often associated with positional changes or inner ear disturbances. 2 Ataxia, regardless of its underlying etiology, leading to impairments in activities of daily living, diminished social participation, and a marked decline in overall quality of life.2,3 While it is essential to address any underlying reversible or progressive causes, the primary focus of treatment lies in mitigating further functional decline and optimizing patient outcomes. Comprehensive rehabilitation programs emphasizing balance, gait, and muscle strengthening have consistently demonstrated their effectiveness in enhancing motor function, trunk stability, gait patterns, and the performance of activities of daily living. 4
Hyperbaric oxygen therapy (HBOT) involves the administration of 100% oxygen at a pressure exceeding one atmosphere absolute, leading to increased tissue oxygenation. Emerging evidence from preclinical and clinical studies suggests that innovative HBOT protocols may stimulate neuroplasticity and enhance brain function, even in the context of chronic injury. 6 By increased oxygen delivery to ischemic tissues, the hypoxia-induced neuronal damage reduces. 7 Additionally, studies indicate that HBOT fosters angiogenesis and neurogenesis, contributing to the repair of damaged neural networks. 8 Furthermore, HBOT has been investigated for its potential to target secondary mechanisms following brain injury, such as oxidative stress, inflammation, and neuronal.9,10 It has been shown to mitigate inflammation and edema by inhibiting the production of pro-inflammatory cytokines, 11 while simultaneously promoting neuroprotective factors, including vascular endothelial growth factor. 12 HBOT also attenuates oxidative stress by enhancing the activity of antioxidant enzymes and decreasing free radical production, thereby preventing further neuronal damage. 13
Moreover, HBOT has been shown to reduce apoptosis by modulating apoptotic pathways, such as by inhibiting caspase activation, a key mediator of cellular death following brain injury.14,15 Given the potential overlap in pathophysiological pathways between brain injury and ataxia, it suggests a rationale for exploring HBOT’s effects on ataxia’s symptoms. This case series presents preliminary evidence suggesting potential clinical benefits of HBOT in patients with chronic ataxia resulting from acquired brain injury.
Case presentation
This is a retrospective case series investigating the effects of HBOT in two patients with chronic ataxia. The patients were treated at the Sagol Center for Hyperbaric Medicine and Research, Shamir Medical Center, Israel, between January 1, 2020 and December 31, 2021. The HBOT protocol included 60 daily HBOT sessions over 12 weeks in a multi-place hyperbaric chamber (HAUX-Life-Support GmbH, Karlsbad-Ittersbach, Germany). Each 90-min session involved 100% oxygen exposure at two absolute atmospheres with 5-min air breaks every 20 min, delivered via tight-fitting masks. Concurrently, patients receive twice-weekly, 30-min physical therapy sessions focusing on static and dynamic balance exercises and gait training. The patients were assessed at baseline, before starting the HBOT treatment. Baseline evaluations were compared to post-treatment assessments conducted 2–3 weeks following the final HBOT session.
Clinical assessments are essential for evaluating functional mobility, balance, and quality of life, informing rehabilitation planning and outcome assessment in ataxia. These assessments include the Romberg Test, which evaluates postural stability by observing a patient’s ability to maintain balance with eyes open and subsequently closed for a standardized duration, typically 30 s. This test assesses the integration of proprioceptive, vestibular, and visual inputs for postural control. Patients with ataxia may exhibit instability within ~6 s, reflecting deficits in proprioceptive, and cerebellar functions impacting postural control. 16 A Timed Up and Go (TUG) test completion time exceeding 14 s indicates increased fall risk and potential impairments in mobility and balance. 17 This prolonged time frequently reflects underlying deficits in gait velocity, postural stability, and motor coordination. Conversely, healthy adults typically demonstrate TUG completion times under 10 s, signifying efficient mobility and balance control. 17 Utilizing the 10-m walk test and its dual-task (DT) variant, quantitative gait analysis demonstrates a significant reduction in walking velocity in individuals with ataxia compared to healthy adults. Healthy adults typically exhibit a gait speed of 1.2–1.5 m/s, whereas individuals with ataxia often present with velocities below 0.4 m/s, indicative of impaired gait patterns and diminished motor coordination. 18 A minimal detectable change (MDC) of 0.06 m/s and a minimal clinically important difference (MCID) of 0.14 m/s have been established for this metric. 18 The 30-s sit-to-stand (STS) test, assessing functional lower limb strength and endurance, quantifies the number of repetitions completed within a 30-s interval without upper extremity support, an MCID of two repetitions has been reported. 19 The motor component of the Functional Independence Measure (FIM) assesses a patient’s physical performance across activities of daily living, encompassing domains such as self-care and locomotion. Each item is scored on a seven-point ordinal scale, ranging from 1, indicating total assistance, to 7, signifying complete independence. 20 The Short Form-36 (SF-36) is a validated, self-administered instrument designed to quantify health-related quality of life. It evaluates eight distinct health domains: physical functioning, role limitations due to physical health, bodily pain, general health, vitality, social functioning, role limitations due to emotional problems, and mental health. Domain scores are normalized to a scale ranging from 0 to 100, where higher scores reflect better health status. Clinically significant changes in domain scores are typically defined as a 5–10-point variation, indicating meaningful shifts in perceived health. Larger changes, exceeding 10 points and potentially reaching 15 points, may suggest more substantial improvements in functional capacity and emotional well-being. 21 See Supplementary Figure S1 for additional data.
In addition to the functional assessments, we used the Zebris FDM-T Treadmill (Zebris Medical GmbH, Isny, Germany) for gait analysis – gait spatiotemporal parameters were acquired using the Zebris FDM-T Treadmill (Zebris Medical GmbH). This instrumented treadmill features a force-sensitive mat of 10,240 miniature sensors capturing foot-ground interactions at 120 Hz. Participants walked at their self-selected comfortable speed for 1 min, following an adaptation period. The following parameters were extracted: velocity, cadence, step time, step length, stride length, center of pressure (CoP) trajectories, and gait line.
Finally, magnetic resonance imaging (MRI) scans were performed on a MAGNETOM VIDA 3 T scanner, configured with 64-channel receiver head coils (Siemens Healthcare, Erlangen, Germany). The MRI protocol included T2-weighted, 3D fluid attenuated inversion recovery, dynamic susceptibility weighted imaging, pre- and post-contrast high-resolution MPRAGE 3D T1-weighted, and dynamic susceptibility contrast for calculating whole-brain quantitative perfusion maps. Briefly, preprocessing of images was performed using the SPM software (version 12, UCL, London, UK) and included motion correction, co-registration with MPRAGE T1 images, spatial normalization, and spatial smoothing with a kernel size of 6 mm full width half maximum. Whole-brain quantitative perfusion analysis was performed as described in previous studies. 22 MR signal intensity was converted to Gadolinium concentrations, arterial input function was determined automatically, fitted to the gamma variate function, and deconvolved on a voxel-by-voxel basis to calculate brain perfusion maps.
Case 1 was a 73-year-old Romanian female with a history of hypertension, atrial fibrillation, and a cerebellar stroke on December 25, 2019. Evaluated 1 year post-stroke, MRI perfusion demonstrated reduced cerebral blood flow in the left cerebellar lesion and the surrounding left hemisphere (Figure 1). Significant balance impairment limited her activities of daily living; she required caregiver assistance for indoor ambulation and used a wheelchair outdoors. Clinical examination revealed left limb muscle strength of 4/5, full range of motion (ROM) in all limbs, a motor FIM score of 13/21 (Table 1), and a positive Romberg test. The patient was unable to independently complete the STS, 10-m walk, 10-m walk with DT, and TUG tests (Table 2). Post-HBOT, the patient reported improved balance, enabling increased independence in daily activities. Ambulation progressed from wheelchair-bound to walker use with close supervision indoors and caregiver assistance outdoors. Clinical examination demonstrated significant improvements, including increased motor FIM score (Table 1), ability to perform three STS repetitions, maintain Romberg balance for 30 s with supervision, and complete the previously insurmountable 10-m walk, 10-m walk DT, and TUG tests (Table 2). MRI perfusion demonstrated increased cerebral blood flow in both left and right cerebellar hemispheres without changes in the lesion core (Figure 1).

Case 1 MR Fluid-attenuated inversion recovery (FLAIR) sequence and perfusion changes.
Functional independence score.

HBOT: hyperbaric oxygen therapy.
Outcome measure.

HBOT: hyperbaric oxygen therapy; TUG: timed up and go; MW: minute walk; DT: dual-task; NA: not available; reps: repetition; STS: sit-to-stand.
Case 2 was a 57-year-old Yemeni male with a history of hypertension and a traumatic brain injury in 2017, resulting in cerebellar atrophy as confirmed by MRI (Figure 2). Despite previous surgical intervention, he experienced persistent cerebellar ataxia, manifesting as gait instability, falls, and difficulties with activities of daily living. Clinical examination revealed bilateral lower limb weakness (4/5), full ROM, a motor FIM score of 19/21 (Table 1), and impaired balance (Romberg test). Functional assessments demonstrated relatively preserved performance on the STS, 10-m walk, 10-m walk DT, and TUG tests (Table 2). Post-HBOT, the patient reported improved functional independence, including the ability to dress and shower without assistance. While muscle strength remained unchanged, clinical assessments revealed significant improvements. Motor FIM scores increased (Table 1), the Romberg test became negative, and STS repetitions doubled to 12 (Table 2). MRI perfusion revealed increased cerebral blood flow in both cerebellar hemispheres without changes in cerebellar volume or atrophy (Figure 2).
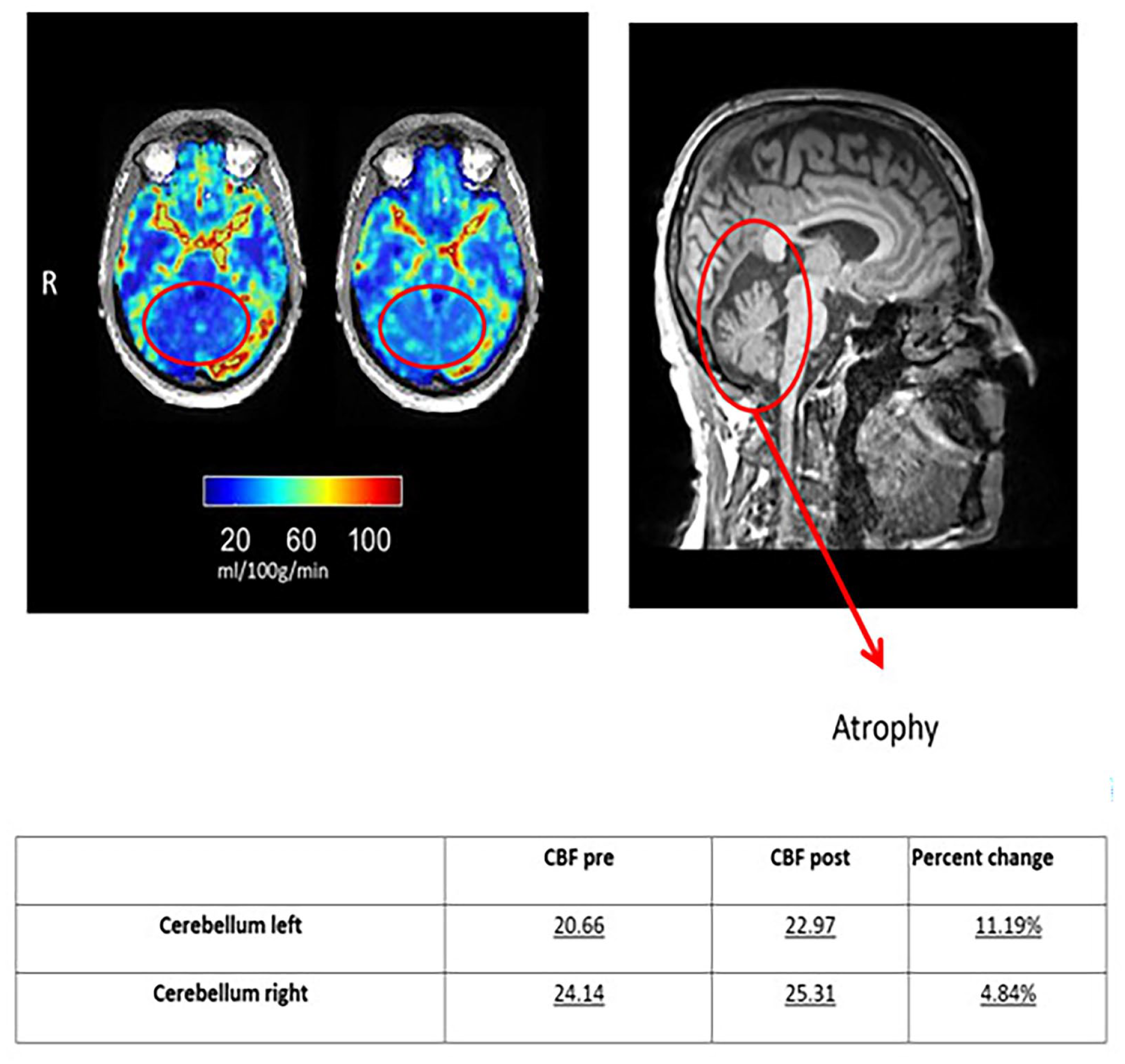
Case 2 MR Fluid-attenuated inversion recovery (FLAIR) sequence and perfusion changes.
Spatio-temporal gait parameters (Table 3) indicated clinically significant improvements (⩾10%) across most variables in both cases, however, differential trends were observed between spatial and temporal parameters. Spatial parameters, specifically step and stride length, exhibited substantial increases (12% and 24% for cases 1 and 2, respectively), although remaining below normative values (step length: 70–80 cm; stride length: 140–160 cm). Conversely, initial temporal parameters (step time: case 1 – right 0.58 s, left 0.60 s; case 2 – right 0.54 s, left 0.59 s), which were at the lower limit of normal (0.6–0.8 s), showed a 10% improvement in case 1 but minimal change in case 2. Notably, cadence exhibited divergent patterns: case 2 maintained a normal cadence (107–109 steps/min; normative range: 100–120 steps/min) while demonstrating spatial improvements, whereas case 1 showed a decrease in cadence (102–92 steps/min) concurrent with improvements in both spatial and temporal parameters. Pre HBOT, CoP, and gait line reflect inconsistent and incoordination, while after treatment, improvement in both the CoP and gait line was observed (Figures 3 and 4).
Gait parameter.

HBOT: hyperbaric oxygen therapy.
⩾10% improvement.

Case 1 gait parameter (gray – pre, white – post).

Case 2 gait parameter (gray – pre, white – post).
Analysis of SF-36 questionnaire data revealed a modest change in physical function scores. However, clinically significant improvements (⩾15 points) were observed in the emotional and social domains for case 1. Both patients reported substantial enhancements in quality of life across multiple SF-36 parameters, with particularly pronounced improvements in the emotional domain (Table 4).
Quality of life SF-36 score.

SF-36: short form-36.
Score range 0–100.
Clinically significant changes: 5–10 points.
Substantial improvements >10.
Discussion
We present a case series demonstrating the efficacy of HBOT in patients with chronic cerebellar ataxia. Participants exhibited profound ataxia characterized by impaired static and dynamic balance, incoordination, and reliance on walking aids. Following a combined regimen of HBOT and physical therapy, patients experienced significant improvements in gait, coordination, and balance. These clinical gains correlated with increased perfusion in peri-lesional cerebellar tissue.
Significant advancements have been made in the management of cerebellar ataxia over the past three decades, with diverse therapeutic approaches demonstrating varying degrees of efficacy. 4 Intensive physical therapy regimens targeting balance, gait, and core stability have shown promise in improving motor function.23–25 Additionally, non-invasive brain stimulation of the cerebellum has emerged as a potential therapeutic avenue for enhancing motor function,26,27 gait,24,25 and potentially augmenting the effects of physical therapy. Nonetheless, the efficacy of these interventions requires further investigation through controlled trials. Our study presents pioneering data on the potential synergistic benefits of HBOT when combined with physical therapy in chronic ataxia patients. Previous studies have demonstrated the efficacy of HBOT in enhancing cognitive function among patients with chronic stroke and traumatic brain injury.6,28,29 Our group has also reported significant improvements in overall neurological scores among 74 chronic stroke patients treated with HBOT. 30 Similarly, Schiavo et al. observed enhanced upper limb motor function in chronic stroke patients following HBOT. 31 However, most of these studies focused on patients with cerebral stroke, often characterized by mono- or hemiparesis. However, the impact of HBOT on cerebellar ataxia, gait, and balance remains largely unexplored.
Our two patients underwent a novel HBOT protocol involving repeated intermittent hyperoxic exposures, known to induce the “hyperoxic hypoxic paradox.” 32 This approach exploits the cell’s sensitivity to oxygen fluctuations, mimicking hypoxia without its harmful effects. Consequently, it activates gene expression and metabolic pathways crucial for regeneration, including immune modulation, increased cerebral perfusion, angiogenesis, restored mitochondrial function, and neurogenesis. 32 While previous studies by our group demonstrated HBOT efficacy in cortical and basal ganglia strokes, 33 the impact on atypical stroke locations and functional outcomes beyond cognition remained unexplored.
This study extends previous findings by demonstrating significant functional and physical improvements in patients with cerebellar ataxia following HBOT. The improvements in FIM scores (Table 1), reflect enhanced performance in activities of daily living and reduced dependency, correlated with observed improvements in mobility as measured by the TUG, STS, and gait velocity tests (Table 2). Quantitative gait analysis further substantiated these findings, revealing improvements in postural stability and increased gait symmetry (Figures 3 and 4).
Intriguingly, while overall gait patterns demonstrated improvement, step width, a critical measure of lateral stability, remained persistently elevated (>10 cm) and outside the established normative range (7–9 cm). This observation is notable given the concurrent enhancements observed in coordination and directional control, as evidenced by the CoP trajectories and gait line analyses presented in Figures 3 and 4. This discrepancy suggests a potential dissociation between improvements in dynamic control and the restoration of optimal lateral stability, warranting further investigation into the neuroplasticity and HBOT effects.
This observation supports the hypothesis that functional improvements in gait velocity and stability in individuals with ataxia may occur through compensatory mechanisms rather than a complete restoration of typical gait patterns. Observed enhancements in the clinical assessments and gait parameters (Tables 2 and 3) demonstrated significant correlations with improvements in balance-dependent functional tasks. These findings suggest the potential for ameliorating functional deficits and facilitating following HBOT and increased community reintegration. The significant improvements, beyond MDC and MCID thresholds, despite baseline heterogeneity in the time since injury, ataxia etiology, and initial functional scores, suggest a potential for HBOT efficacy that is not strictly dependent on these factors. However, the inherent limitations of a heterogeneous cohort necessitate cautious interpretation.
The SF-36 assessment revealed clinically significant improvements exceeding 15 points in vitality, bodily pain, and particularly, mental health domains following HBOT in patients with ataxia. Given the established relationship between subjective well-being and motivational states, these findings raise the possibility that HBOT may confer positive effects on psychological health and quality of life, in addition to observed enhancements in physical performance within this patient population. Finally, the convergence of observed neuroimaging changes with corresponding clinical improvements provides compelling evidence supporting a causal relationship between HBOT and functional recovery in cerebellar ataxia, thereby underscoring its therapeutic potential.
While the preceding discussion presents a rationale for HBOT in ataxia, a rigorous, systematic evaluation of its efficacy across diverse etiologies and ataxia phenotypes is imperative to elucidate underlying mechanisms and optimize clinical outcomes. In addition, the absence of follow-up data precludes assessment of the sustained effects; therefore, prospective studies incorporating extended follow-up periods are essential to assess the long-term stability and durability of observed treatment effects.
Conclusions
This case series offers compelling preliminary evidence supporting the potential efficacy of HBOT as a treatment for cerebellar ataxia. The neurophysical improvements correlated with increased cerebral perfusion within the peri-lesional parenchyma, implying a putative mechanism of action. While these findings offer initial insights into HBOT’s potential benefits for neurological insult, further investigation is warranted. Randomized controlled trials with larger sample sizes and control groups are essential to validate and generalize these findings, ensuring a comprehensive understanding of HBOT’s effects on various brain injuries.
Supplemental Material
sj-docx-1-sco-10.1177_2050313X251337996 – Supplemental material for Effect of hyperbaric oxygen therapy on ataxia: A case report
Supplemental material, sj-docx-1-sco-10.1177_2050313X251337996 for Effect of hyperbaric oxygen therapy on ataxia: A case report by Vicktoria Elkarif, Amir Hadanny, Ruth Tuck Harpaz, Efrat Sasson and Shai Efrati in SAGE Open Medical Case Reports
Footnotes
Ethics considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent for publication
Written informed consent was obtained from the patients for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Author contributions
V.E.: wrote the first draft of the article and the final version. Collected and analyzed the patient’s data. S.E. and A.H.: reviewed the first draft and the final version. R.T.H.: treated the patients who were presented. E.S.: analyzed the MRI results.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: V.E., A.H., and S.E. works for AVIV scientific LDT. E.S. is a shareholder at AVIV scientific LDT.
Data availability Statement
The datasets analyzed during the current study are available from the Sagol Center for Hyperbaric Medicine, Shamir Medical Center, Israel, on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
