Abstract
The proteasome inhibitor bortezomib is widely used in the treatment of multiple myeloma. While peripheral neuropathy and gastrointestinal effects are well-documented adverse reactions to bortezomib administration, cutaneous side effects in patients with multiple myeloma are less commonly reported. We present a patient with IgG kappa multiple myeloma who developed a spider-like and digitate eruption at the site of subcutaneous bortezomib injection. This report expands the spectrum of bortezomib-associated dermatologic toxicity and emphasizes the importance of clinician awareness for prompt diagnosis and management.
Introduction
Multiple myeloma (MM) is a hematologic malignancy characterized by clonal proliferation of plasma cells producing monoclonal immunoglobulins (M-protein). 1 Although MM accounts for 1%–2% of cancers globally, incidence has increased significantly in recent years. 2 The treatment landscape for myeloma was relatively stagnant from the 1970s to 1990s, but over the past 25 years, several important therapies have emerged. 3 Specifically, bortezomib (Velcade) was the first proteasome inhibitor approved by the U.S. Food and Drug Administration as a first-line agent for patients with MM in 2008. 4 The proteasome is responsible for degrading senescent proteins and is essential to cell homeostasis. Bortezomib reversibly binds the chymotrypsin-like subunit on the 26S proteasome, preventing degradation of pro-apoptotic factors and leading to cell cycle arrest in cancer cells. 5 This novel therapy has been a major advance in the treatment and prognosis of patients diagnosed with MM, improving 5-year survival rates increased from 30%–35% to 50%–55%. 2 Although these advances in therapy have changed the landscape of therapy for MM, there are known reported adverse events associated with bortezomib. The most cited and clinically relevant toxicities of bortezomib include painful peripheral muscle weakness, neuropathy, nausea, vomiting, and diarrhea. 6 When first introduced, bortezomib was given intravenously, but over time, subcutaneous administration, which lowers the risk of neuropathy without reducing efficacy, has become standard of care. Subcutaneous bortezomib injections have been reported to trigger cutaneous adverse reactions in 10%–24% of patients.7,8 These reactions may include maculopapular eruptions, vasculitis, neutrophilic eruptions, and serious cutaneous adverse reactions such as Steven–Johnson syndrome and toxic epidermal necrolysis.7–15 We present a unique case of a patient with MM who developed a progressive digitate and spider-like injection site reaction following treatment with bortezomib.
Case report
A 56-year-old male diagnosed with IgG kappa plasma cell MM was initiated on a treatment protocol consisting of lenalidomide, bortezomib, and dexamethasone. Within this protocol, bortezomib is given as a subcutaneous injection on days 1, 8, 15, and 22 and repeated on a 28-day cycle. Following the first bortezomib injection, this patient noted a cutaneous reaction at the injection site. He described a significantly pruritic and erythematous plaque that progressively expanded outward around the site of injection with finger-like projections. The patient reported that the reaction would progress over 2–3 days, and during this time, the erythema was noted to track caudally in linear streaks from the site of injection. Resolution of the eruption would begin with improvement of the pruritus followed by the erythema. Similar reactions occurred following each bortezomib dose, despite rotation of injection locations, and the patient noted that each reaction was larger than the last. The patient otherwise felt well following the injections and denied other symptoms associated with bortezomib hypersensitivity.
Prior to this presentation, the patient had no history of skin disease and had never had a reaction or hypersensitivity to medications. He did not take any regular medications other than the myeloma treatment protocol. He was actively smoking and had a 40 pack-year smoking history.
The patient was systemically well on presentation to clinic and was noted to have a sharply demarcated erythematous plaque on his left lower abdomen at the most recent injection site (Figure 1). Interestingly, it was noted that there were linear digitate projections tracking caudally along the abdomen. Examining the sites of previous injections, there was similar post-inflammatory hyperpigmented patches at each site. Full cutaneous examination revealed no evidence of other primary skin disease, and the rest of the body and mucosa were spared.
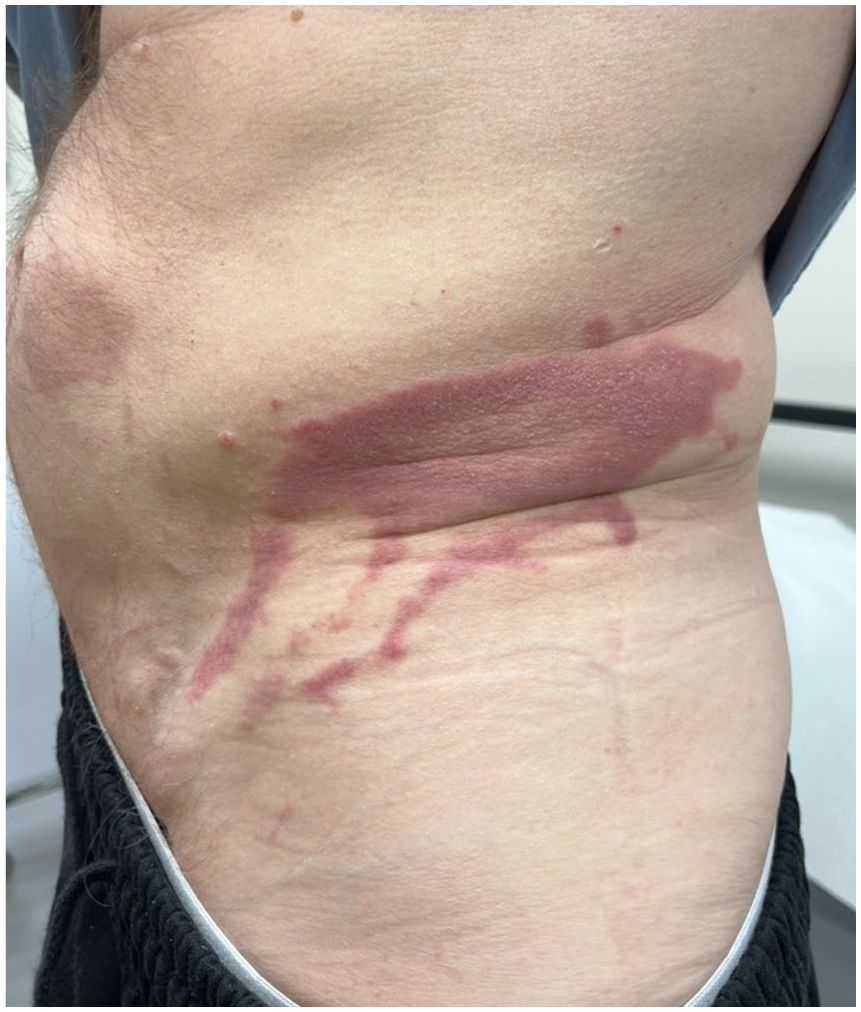
Erythematous and pruritic plaque with digitate projections spreading caudally at the site of recent bortezomib injection.
Management of the injection site reaction was initiated with a topical compound containing 0.5% menthol and 0.5% camphor in betamethasone valerate 0.1% cream to be applied daily as needed for symptomatic treatment of the pruritus and inflammation. Bortezomib was continued following discussion with his hematologist, and recommendations were given to reduce injection site reactions. This included administration of a non-sedating antihistamine prior to injection and application of ice at the site following injection.
The patient was reassessed 6 weeks later, and the patient noted that subsequent injection site reactions were mild and tolerable with the measures implemented. There was ongoing post-inflammatory hyperpigmentation that was subtle but noticeable at the site of the previous reaction but no active reaction elsewhere on his skin (Figure 2). He continued application of the compounded cream after the injections for symptomatic relief.

Subtle post-inflammatory hyperpigmentation at the site of previous digitate injection site reaction.
Discussion
Cutaneous adverse drug reactions to bortezomib are increasingly recognized, particularly with the transition from intravenous to subcutaneous administration. While subcutaneous delivery reduces the risk of peripheral neuropathy, it has been associated with a higher incidence of dermatologic reactions.1,2 These reactions have been reported to include maculopapular eruptions, vasculitis, neutrophilic eruptions, and serious cutaneous adverse reactions including Steven–Johnson syndrome and toxic epidermal necrolysis.7–9
The pathogenesis of bortezomib-induced skin reactions is not yet fully understood. One hypothesis proposes the upregulation of pro-inflammatory cytokines, particularly interleukin-6 and tumor necrosis factor-alpha, which may amplify cell-mediated immune responses and contribute to hypersensitivity-type eruptions. 3 These cytokines, which are already elevated in MM, may be further potentiated by bortezomib’s immunomodulatory effects, increasing the risk of cutaneous toxicity. There have been very few reported cases of similar spider-like or digitate injection site reactions following bortezomib injections.10,13 It has been hypothesized that the digitate or spider-link spread of the plaque may be related to diffusion of medication into local capillaries and lymphatics, which may also be the case for this patient. 13
This rare presentation emphasizes the need for heightened clinical awareness to recognize atypical dermatologic presentations of toxicities from cancer therapies. A pooled safety analysis has shown that 22% of patients discontinue bortezomib therapy due to toxicity, posing a major clinical challenge. 16 Injection site reactions may lead patients to discontinue therapy if they are symptomatic or significantly distressing to the patient. 17 Because bortezomib is a commonly used, a standard of care therapy for MM, especially in high-risk disease, early identification of adverse cutaneous responses is critical. Prompt dermatologic evaluation, diagnosis, and appropriate management including consideration of corticosteroids, antihistamines, or modification of the treatment regimen can prevent progression and improve patient outcomes.
This case of a digitate plaque following subcutaneous bortezomib injection expands the spectrum of bortezomib-related skin reactions and highlights the importance of reporting novel presentations. Further studies are warranted to elucidate the underlying pathophysiology of cutaneous adverse reactions to proteasome inhibitors, such as bortezomib. Recognition and prompt management of cutaneous toxicities to novel immunotherapies allows patients to remain on treatment and optimize outcomes.
Footnotes
Consent for publication
Patient consent was obtained for the publication of this manuscript.
Author contributions
Rachel Creighton – manuscript writing and editing. Tamryn Eglington – manuscript editing, patient care. Luke Y. C. Chen – manuscript editing, patient care. Ashley E. Sutherland – manuscript writing, editing, patient care.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
