Abstract
Objective:
The increasing demand for precision medicine has spurred molecular diagnostic investigations to emphasize the utility of miRNA as significant biomarkers. Recent studies have underscored miRNA’s role as prognostic, diagnostic, and therapeutic biomarkers in managing and monitoring multiple myeloma patients. This review aims to present the latest insights on the potential of circulating miRNA as prognostic, diagnostic, and therapeutic biomarkers in bortezomib-resistant multiple myeloma.
Methods:
For this purpose, a comprehensive thematic literature review from January 2014 and August 2024 was conducted utilizing the databases CINAHL, Pubmed, and Google Scholar. Twenty pertinent studies were meticulously analyzed and categorized into the following sections: Bortezomib (BTZ) resistance in multiple myeloma, the predictive role of miRNAs in BTZ resistance, the impact of circulating miRNAs in multiple myeloma, and the potential of circulating miRNA as prognostic, diagnostic, and therapeutic biomarkers.
Results:
Of note, eight studies identified circulating miRNAs as diagnostic miRNA biomarkers (i.e., miR-744, miR-130a, let-7d, let-7e, miR-34a, etc.). In comparison, nine studies identified several circulating miRNAs that can be used as prognostic biomarkers (i.e., miR-20a, miR-483-5p, mir-1246, let-7a, let-7e, etc.). Moreover, five studies identified circulating miRNAs as promising therapeutic biomarkers (i.e., mir-15a, mir-92a, mir-19a, etc.). This discovery can significantly enhance early detection, accurate diagnosis, prognosis, overall survival rates, and quality of life for patients with multiple myeloma.
Conclusion:
Based on this evidence, exploring circulating miRNAs as a potential noninvasive biomarker for multiple myeloma represents a noteworthy advancement. This is attributed to the abundance of miRNAs in plasma or serum, which exhibits remarkable stability against enzymatic degradation.
Introduction
Multiple myeloma (MM) is a complex and incurable hematological malignancy characterized by the infiltration of abnormal plasma cells in the bone marrow (BM), resulting in the secretion of an overabundance of monoclonal paraproteins. The clinical manifestation of the disease is identified through CRAB features (calcium elevation, renal insufficiency, anemia, and bone lesions). In 2024, it was anticipated that the United States would witness approximately 35,780 new MM cases, with 19,520 cases occurring in males and 16,260 in females. Additionally, MM is projected to be responsible for 12,540 deaths, comprising 7020 fatalities in males and 5520 in females. 1 The incidence rates per 100,000 individuals in the United States are 8.7 for males and 5.9 for females, the highest rates observed among non-Hispanic Black individuals, at 17.1 for males and 13.0 for females. 2 These inequalities are attributed to a complex interplay of genetic, environmental, and cellular alterations among neoplastic cells, which lead to tumor heterogeneity and resistance to therapeutic agents. 3
Traditionally, tissue biopsies have played a pivotal role in cancer diagnosis; however, there is a significant trend toward adopting noninvasive techniques, such as liquid biopsies, for the detection of cancer biomarkers. The utility of noninvasive biomarkers is increasingly recognized in early detection, prognostication, monitoring, and evaluation of treatment responses, thereby contributing to the advancement of precision medicine. 4 Notable biomarker candidates encompass circulating tumor DNA (ctDNA), cell-free DNA (cfDNA), and exosomes. 4 Nonetheless, considerable challenges remain in establishing their clinical utility and validating their effectiveness as reliable biomarkers. 5 A significant challenge with ctDNA/cfDNA is the low levels of peripheral DNA, which affects sensitivity. Also, sample preparation for DNA methylation analysis limits the rigorous clinical application of this technology in everyday use. 6 As the incidence of MM continues to rise, there has been a burgeoning utilization of next-generation biomarkers, including extracellular matrix proteins, angiogenesis markers, miRNAs, cell-free DNA, immune markers, novel proteomics-based markers, and genomic markers, each holding promise in refining management and treatment strategies for individual patients, subsequently leading to improved health outcomes. 7
Nevertheless, despite therapeutic advancements over the past 2 decades, MM remains an incurable disease with a grim prognosis and an overall 5-year survival rate. 8 This is primarily due to frequent treatment failures and disease relapse. 9 Clinical studies have explored the synergistic effects of circulating miRNAs as noninvasive biomarkers for tumor diagnosis and for monitoring anti-MM therapies to enhance patient outcomes. 10 Despite the initial efficacy of Bortezomib (BTZ) treatment, a significant proportion of patients experience drug resistance and subsequent relapse. 10 However, BTZ is a promising proteasome inhibitor (PI) demonstrating a significant effect in MM treatment. BTZ operates by obstructing the activity of PSMC1, a component of the 26S proteasome, crucial for the ATP-dependent degradation of ubiquitinated proteins, particularly those governing the cell cycle and gene regulation.10,11 Cancer cells can upregulate the ubiquitin-proteasome system (UPS), which facilitates the degradation of tumor suppressor proteins, thereby evading apoptosis. 12 BTZ targets cancer cells by inhibiting the 26S proteasome, modulating NF-κB, enhancing NOXA expression, preventing p53 degradation, activating caspases, and inhibiting angiogenesis. 13 Multiple biological mechanisms, including acquired mutations, inducible changes in cell-survival cell signaling, and chromosomal abnormalities, 14 have been identified as crucial in reducing resistance to BTZ in patients with MM. Dysregulated cell signaling is a critical factor in the advancement of MM, occurring through interactions between MM cells and the BM microenvironment. 14 The precise mechanism underlying the development of BTZ resistance in MM remains unclear. The precise mechanism underlying the development of BTZ resistance in MM remains unclear. Numerous studies show that the dysregulation of miRNA is a key feature of cancer profiling. These nonprotein-coding RNAs modulate gene expression posttranscriptionally and significantly influence physiological and pathological processes, including cell differentiation, angiogenesis, apoptosis, tumorigenesis, and drug resistance.14,15 This phenomenon is critical for accurately classifying tumors, their diagnosis, prognosis, and developing targeted therapeutic strategies. 15 This thematic review aims to comprehensively examine existing literature on BTZ resistance in MM, explicitly focusing on the predictive role of miRNAs, the significance of circulating miRNAs, and the potential of utilizing circulating miRNAs as biomarkers for prognostic, diagnostic, and therapeutic purposes.
Bortezomib resistance in MM
Recent advancements in drug therapy, particularly with PIs, have considerably improved the prognosis for patients with MM. 16 Nevertheless, the duration of remission remains limited, often resulting in relapses due to resistance to BTZ. 16 Although BTZ is designed to enhance remission rates, subsequent exposure to the drug does not necessarily lead to improved survival outcomes. 16 BTZ functions by reversibly binding to the β5 subunit of the 20S proteasome, thereby obstructing the degradation of pro-apoptotic proteins that promote cancer cell apoptosis. 17 Its role in managing MM is strategic 18 ; however, complications arising from resistance may result from genetic mutations, epigenetic alterations, and modified metabolic pathways.15,19
Moreover, many anticancer drugs are known to be P-glycoprotein (P-gp) substrates, which can result in increased drug resistance. Interestingly, BTZ is identified as a poor substrate for P-gp, potentially limiting its therapeutic effectiveness in clinical settings. 19 It is imperative to comprehend the intricate mechanisms that underlie acquired resistance to BTZ to refine treatment approaches for MM patients. 20 Studies indicate that such resistance may be attributed to somatic mutation in the PSMB5 gene, which modifies the protein's structure and impedes its binding affinity with BTZ, consequently contributing to drug resistance in malignant cells, particularly in MM.16,21
Extensive research indicates that the overexpression of proteasome maturation protein gene (POMP gene) contributes to resistance against BTZ.16,22 Inhibition of apoptosis is a critical element in developing drug resistance. Furthermore, alterations in drug targets are associated with this resistance, particularly the decreased activity of IRE1/XBP1 and the reliance on the unfolded protein response. 23 Additionally, the reduced efficacy of the UPS can promote resistance by disrupting processes such as autophagy, de-ubiquitination, and the functioning of stress-management chaperone proteins. 24 Chaperones, including Hsp70 and Hsp90, significantly influence autophagy, affecting the survival and apoptosis of MM cells.25,26 Moreover, epigenetic mechanisms, such as DNA methylation and histone acetylation, profoundly impact gene regulation. 27 Hypermethylation of promoter regions within CpG islands may suppress tumor growth by silencing essential genes, including CDKN2B, CDKN2A, CDH1, DAPK1, SOCS1, and SHP1. 28 Additionally, the methylation of the CDKN promoter genes, including MYBPHL, is implicated in myelomagenesis.29,30 The hypermethylation of the DAPK gene at similar rates in both MM and MGUS suggests that it may serve as an early marker of MM pathogenesis.28,31
The deletion of the short arm of chromosome 17, which results in the inactivation of TP53, has been shown to adversely affect the survival rates of patients with MM, with the loss of TP53 serving as a negative prognostic indicator.30,32 Epigenetic mechanisms and miRNAs play a significant role in cancer pathogenesis and treatment strategies. 33 Additionally, the expression of specific miRNAs, particularly miR-125b and miR-50, downregulates the levels of p53, thereby inhibiting apoptosis. 33 In particular, miR-125b modulates the NF-κB, p53, PI3K/Akt/mTOR, ErbB2, Wnt, and various other signaling pathways that govern cellular activities such as proliferation, differentiation, metabolism, apoptosis, drug resistance, and tumor growth immunity. 34 These interactions involve adhesion molecules and soluble factors that activate essential signaling pathways such as NF-κB, PI3K/AKT, and JAK/STAT, which are vital for MM cell proliferation, survival, migration, and the inhibition of apoptosis, ultimately contributing to drug resistance.34,35 Moreover, the imbalance between inflammatory and anti-inflammatory mechanisms plays an essential role in the pathogenesis of MM. Elevated levels of inflammatory cytokines disrupt signaling pathways in MM, promoting the proliferation of malignant cells.35,36 Adhesion molecules initiate signaling cascades that lead to cytokine secretion, activation of osteoclast, a reduction in osteoblasts, and an increase in MM cell proliferation through the upregulation of pathways including PI3K, STAT3, NF-κB, and MAPK.35,36 Furthermore, releasing cytokines, such as VEGF and FGF-2, facilitates neovascularization in MM via the MEK/ERK and PI3K pathways.35,36
BTZ resistance is primarily attributed to abnormal drug metabolism and transport, largely due to hypermethylation of ABC transporters, including ATP binding cassette subfamily B member 1 (ABCB1), ATP binding cassette subfamily B member 2 (ABCG2), and lung resistance-related protein (LRP). This condition contributes to the phenomenon of multidrug resistance. 37 Various miRNAs have been identified as influential factors regulating ABC transporter protein expression by targeting mRNAs or their respective regulators. 38 The reduction of efflux ABC transporter levels may significantly impact substrate disposition, particularly concerning antineoplastic drugs, thereby affecting chemosensitivity. 38 For instance, miRNAs regulate the ABCB1 gene, which encodes P-gp, a critical component of multidrug transport. 38 P-gp serves to detoxify drugs, with many anticancer agents acting as substrates that can contribute to drug resistance. 38 Breast cancer resistance proteins share functional similarities with P-gp, acting as protective mechanisms against toxins within normal tissues, 39 in all the cases such as acute myeloid leukemia, breast cancer, and MM patients undergoing BTZ treatment. 40 Additionally, increased levels of LRP in acute myeloid leukemia, breast cancer, and MM patients undergoing BTZ treatment, 41 also facilitates tumorigenesis and enhance cell survival.42–45 Multidrug-resistant proteins (MRPs), such as MRP1 and MRP2, mediate tumor resistance.46,47 The overexpression of MRP6 has been linked to resistance in tumors exhibiting high MRP1 levels, while MRP7 and MRP8 also contribute to the challenge of anticancer resistance.48,49 MRP transporters are essential for the detoxification process and for mediating glutathione-dependent drug resistance, particularly regarding BTZ. 49 Generally, MRPs confer a higher degree of resistance than P-gp, and both can induce tumorigenesis when overexpressed. 50 miRNAs play a pivotal role in MDR by modulating the expression of various drug-resistant proteins, which constitute a significant cause of treatment failure in cancer therapy.51–53
Glutathione (GSH) plays a crucial role in the detoxification of xenobiotics and has a significant influence on the mechanisms underlying treatment resistance in cancer cells. 54 The elevation of GSH levels enhances drug metabolism, thereby contributing to resistance against chemotherapy. 55 MM dysregulation results from genetic mutations, which include gain-of-function changes in oncogenes such as NRAS, KRAS, BRAF, and cyclin D1 CCND1, along with loss-of-function mutations in tumor suppressor genes like p53, retinoblastoma 1(RB1), DIS3, cyclin-dependent kinase 2A (CDKN2A), and cyclin-dependent kinase 2C (CDKN2C). 56 Furthermore, mutations in the NF-κB and STAT3 signaling pathways significantly contribute to the disease’s pathophysiology. 56 These genetic alterations impair proper cell cycle regulation and the induction of apoptosis. 57 Moreover, the tumor suppressor protein p53 is often mutated, adversely affecting both the cell cycle and apoptotic processes.58–60 Mutations in p53 within MM cells can activate pathways that promote antiapoptotic mechanisms, thus leading to treatment resistance. 61 Notably, the progression of c-Myc on chromosome 8q24 has been linked to aggressive disease progression and resistance to BTZ. 62 Consequently, it enables myeloma cells to evade the effects of bortezomib, often resulting in treatment failure, particularly in advanced cases. Furthermore, gene and protein expression alterations within the BM niche can exacerbate this resistance, diminishing the drug’s efficacy in eliminating cancer cells. 63
The role of miRNAs in predicting bortezomib resistance in MM patients
The rise of drug resistance in addressing patient conditions and gene expression regulation necessitates the incorporation of novel biomarkers, such as miRNAs, to bring about transformative changes. Recent findings have revealed the identification of new circulating miRNAs capable of predicting BTZ resistance in MM patients. These miRNAs are pivotal in modulating gene expression under physiological and pathological conditions. miRNAs are promising clinical biomarkers due to their stability and presence in easily obtainable biofluids from noninvasive methods. Research demonstrates that miRNAs exhibit stability across various biofluids, including blood, CSF, urine, saliva, and lacrimal fluid. These molecules maintain their integrity at room temperature for up to 96 h and can be stored for several months at low (−20°C) and ultralow (−80°C) temperatures.64,65 Moreover, routine regular laboratory freeze-thaw cycles have been shown to have no detrimental effects on the quality of miRNAs. In the context of drug-treated tumor cells, miRNAs have been found to decrease the expression of genes associated with drug resistance, facilitate apoptosis evasion, and promote tumor stem cell development.65,66 Researchers believe combining them with chemotherapy can elevate therapeutic efficacy while mitigating drug resistance.
Consequently, miRNAs have now earned recognition as monitoring markers for anticipating BTZ resistance in MM patients. MiRNAs bolster treatment efficacy by inhibiting tumor growth, reversing chemoresistance, suppressing angiogenesis, inducing apoptosis, enhancing immune response, and ultimately elevating patients’ quality of life. 67 Recent scientific evidence underscores the impact of miRNA dysregulation and its potential as a noninvasive diagnostic, prognostic, and therapeutic biomarker for next-generation liquid biopsy. miRNAs are increasingly recognized as pivotal factors in the pathogenesis of various cancers.
Moreover, circulating miRNAs have been proposed as biomarkers for cancer detection, and genetic mutations of miRNA expression have been linked to tumorigenesis in various cancers. 68 Recent studies have demonstrated the utility of circulating miRNA as prognostic biomarkers for MM. 69 These findings underscore the urgency of identifying prognostic biomarkers for BTZ resistance to enable personalized oncology and precision medicine. Identifying prognostic factors can lead to improved patient survival outcomes, therapeutic strategies, and better stratification of clinical trials.
Identifying patients who are likely to respond to therapy is of paramount importance in optimizing treatment outcomes. Predictive biomarkers play a crucial role in assessing the probable response of patients to treatment in terms of efficacy or toxicity. 70 This information can potentially enhance overall patient health outcomes, encompassing pain management, prevention of bone density, reduction of disability payments, identification of at-risk individuals for MM, and provision of evidence-based personalized care. While several researchers have investigated the role of miRNA as a predictive marker in BTZ resistance, there exists a need for further insight into the mechanisms of drug resistance in MM patients. Our thorough literature review has culminated in identifying such markers, intending to guide researchers and clinicians in comprehending the treatment landscape for MM. Employing a thematic analysis, our study scrutinized published works on circulating miRNA and its role as prognostic, diagnostic, and therapeutic biomarkers associated with BTZ resistance in MM patients. Furthermore, we have synthesized the available miRNA markers to establish a foundation for personalized treatment, subsequently equipping clinicians with the means to make informed decisions for the improved survival of patients with MM.
Methods
To delineate this thematic review, we developed a systematic search strategy to locate pertinent literature to analyze circulating miRNAs as valuable biomarkers in prognosticating BTZ resistance in patients afflicted with MM.
Search strategy
The search strategy was tailored for three databases: CINAHL, PubMed, and Google Scholar, employing the search terms: “microRNA” or “miRNA” and “MMs,” “Myelomas, Multiple,” “Myeloma, Plasma-Cell” “Myeloma, Plasma Cell” “Myelomas, Plasma-Cell,” “Plasma-Cell Myeloma,” “Plasma-Cell Myelomas,” “Myeloma-Multiple,” “Myeloma Multiple,” “Myeloma-Multiples,” “Kahler Disease,” “Disease, Kahler” and “Plasma Cell Myeloma and Cell Myeloma, Plasma” and “Cell Myelomas” and “Plasma and Myelomas, Plasma Cell” and “Plasma Cell Myelomas” and “Myeloma, Multiple” and “Myelomatosis” and “Myelomatoses.” The search was refined to include English articles involving human subjects and encompassed peer-reviewed articles and research reports published between January 2014 and August 2024.
Screening
The inclusion criteria for the papers encompassed studies involving newly diagnosed MM patients and BTZ resistance and the utilization of miRNA derived from serum or plasma specimens. Exclusion criteria accounted for duplicated information, literature letters, comments, case reports, and studies, where miRNA is not obtained from peripheral circulating blood, for example, through animal experiments or biopsies.
The initial search process yielded 143 citations. Subsequently, 22 duplicate citations and 25 irrelevant articles were removed, and 121 unique articles were sought for potential retrieval. After excluding 28 articles with only abstracts, a total of 74 abstracts were subjected to screening for relevant content based on predefined inclusion and exclusion criteria. Among these 74 articles, only 46 were screened as full-text articles containing pertinent information on circulating miRNA associated with BTZ-resistant MM. Another 26 articles were excluded for not being about circulating miRNA, MM, and BTZ resistance. Finally, only 20 articles served as the foundation for thematic analysis, encompassing the role of circulating miRNA in MM, prognostic, diagnostic, and therapeutic discoveries related to circulating miRNA in MM, and methodological strategies for identifying microRNA, as delineated in Figure 1 Prisma flow chart. This culminated in the qualitative synthesis of 20 articles to establish the overarching themes.
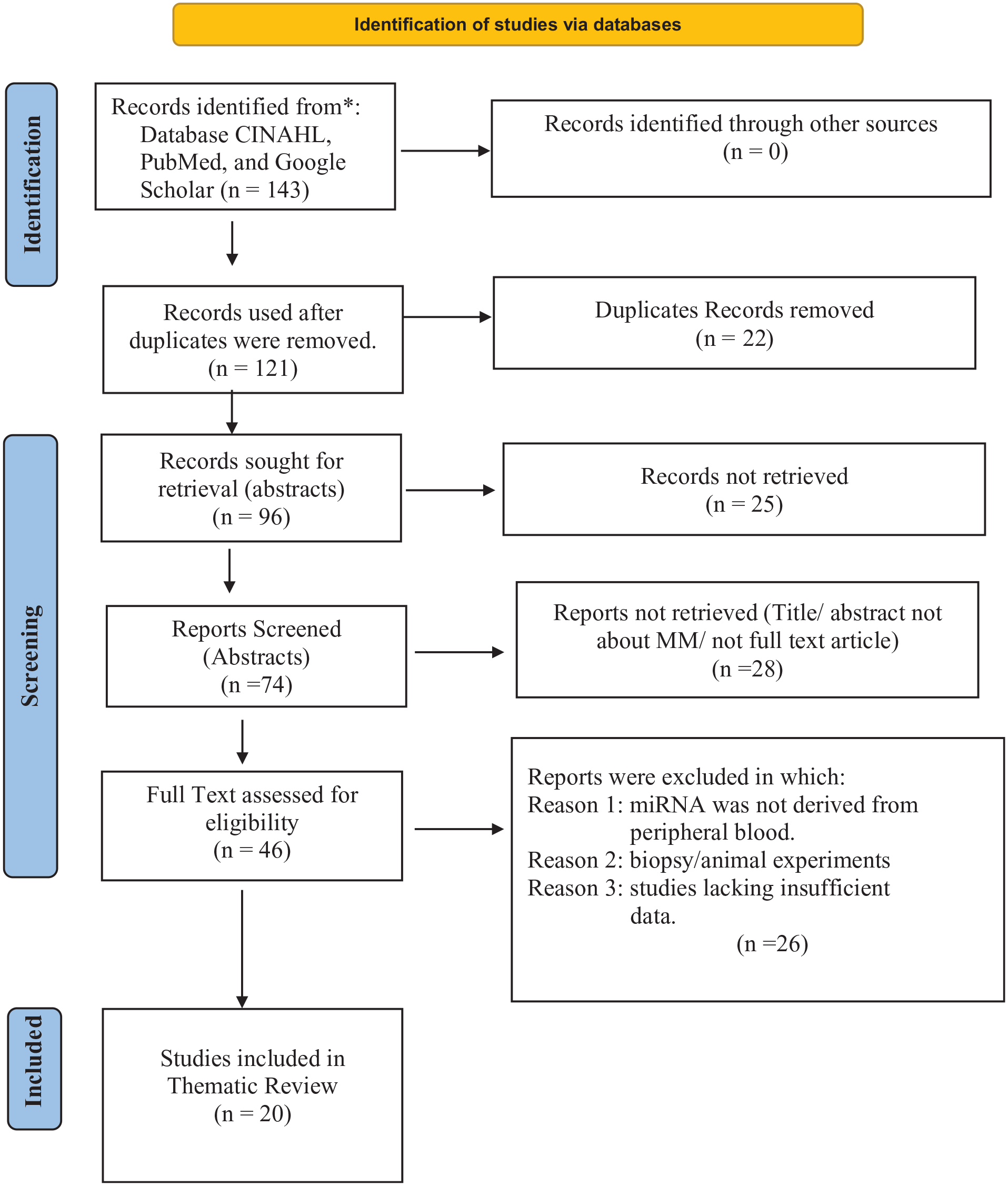
PRISMA 2020 flow diagram for new systematic reviews which included searches of databases.
Results
The results of this thematic review concerning the relationship between circulating miRNA as a biomarker in MM were methodically organized and categorized by PRISMA guidelines. This thematic review meticulously examines the role of circulating miRNAs as biomarkers in MM, adhering to PRISMA guidelines for systematic reviews. As illustrated in Table 1, from an initial pool of 143 articles, 20 articles were deemed eligible and included in the analysis following a comprehensive screening process. The selected studies were categorized into three key themes, providing a structured framework for this investigation.
Circulating miRNAs as diagnostic, prognostic, and therapeutic biomarkers in multiple myeloma disease.

Table 1 presents recent insights into the function of circulating miRNAs in MM, highlighting their potential as diagnostic, prognostic, and therapeutic targets that could enhance clinical management strategies in the future. Among the eight studies focusing on diagnostic biomarkers, several miRNAs were identified as noteworthy. Specifically, miR-774, miR-130a, let-7d, miR-143, miR-144, miR-199, miR-203, let-7d-5p, miR-103a-3p, and let-7e demonstrated significant downregulation in MM patients. Conversely, the upregulated miRNAs in serum from BTZ-resistant MM patients included miR-34a, miR-4449, miR-125b-5p, miR-483-3p, miR-4326, miR-6894-3p, miR-4498, miR-490-3p, miR-7155-5p, miR-937-3p, alongside previously noted miRNAs such as miR-125b-5p, miR-490-3p, miR-143, miR-144, miR-199, miR-203, let-7d-5p, miR-103a-3p, miR-4741, miR-134-5p, miR-107, miR-15a-5p, miR-448, and miR-1246. These findings underscore the intricate roles that circulating miRNAs play in the pathophysiology of MM and their potential as biomarkers for clinical applications.
This review has identified nine studies focusing on microRNAs (miRNAs) and their correlations with the prognosis of MM, as summarized in Table 1. The analysis reveals a range of down-regulated miRNAs, including miR-744, let-7e, miR-20a, miR-483-5p, let-7a, let-7b, let-7i, miR-15b, miR-16, miR-18a, and miR-20a. Conversely, the upregulated miRNAs consist of miR-214, miR-135b, miR-720, miR-1246, miR-125b-5p, miR-8074, and miR-25.
Additionally, Table 1 outlines five articles that provide a list of therapeutic circulating miRNA biomarkers that are downregulated, including miR-19a, miR-4254, miR-15a-5p, miR-16-5p, miR-17-5p, miR-20a-5p, miR-30c-5p, let-7b-5p, and miR-27b-3p, along with miR-497. In the context of BTZ treatment, downregulated BTZ-refractory patients include miR-130a-3p, miR-151a-3p, miR-181a-5p, miR-191-5p, miR-328-3p, miR-376a-3p, miR-409-3p, miR-744-5p, miR-766-3p, and miR-1224-3p. On the contrary, upregulated miRNAs associated with BTZ-refractory patients in MM include miR-16-2-3p, miR-19b-3p, miR-29b-3p, miR-30e-5p, miR-122-5p, miR-143-3p, and miR-148a-3p. However, these studies validated only 215-5p, mir-37c-3p, and miR-181a-5p to predict the response to BTZ treatment.
Discussion
MM is an incurable type of blood cancer that accounts for 10% of hematologic malignancies and primarily affects elderly males. Treatment typically involves several drugs, including BTZ, which is frequently prescribed. However, patients often develop resistance to BTZ therapy rapidly. There is a critical need to understand the molecular basis of BTZ resistance in this population and, more importantly, to identify biomarkers that can predict resistance to BTZ in these patients. This thematic analysis synthesizes current literature investigating the potential of miRNAs as biomarkers for predicting BTZ resistance in MM, aiming to clarify the molecular mechanisms driving this resistance. Specifically, this analysis examines the role of circulating miRNAs and their diagnostic, prognostic, and therapeutic potential in MM.
Role of circulating miRNA in MM
MiRNAs, ranging from 19 to 25 nucleotides in length, are noncoding RNAs that are released into the circulatory system. 69 They function as regulatory noncoding singular stand RNAs and play a crucial role in the modulation of biological functions, cellular differentiation, proliferation, apoptosis, and cancer metastasis. 47 Alterations in miRNA profiles frequently manifest during cancer initiation, progression, treatment response, remission, and relapse. Recent findings have outlined the secretions of miRNAs into body fluids, including serum and plasma, where they remain stable while freely circulating in the bloodstream.
Consequently, the clinical significance of circulating miRNA lies in their noninvasive role as diagnostic, prognostic, and therapeutic biomarkers in MM. 69 Specific miRNAs, including miR-20a, miR-19a, miR-92a, and miR-214-3p, function as oncomiRs, playing pivotal roles in inhibiting apoptosis and promoting cell proliferation, migration, and invasion. 70 Conversely, miRNAs such as miR-15a-5p, miR203, miR144, miR199, miR-483-5p, miR-34a, miR-33b, miR-202, and miR-29a act as suppressors of tumor growth. 70 Specific miRNAs are implicated in developing the BM microenvironment in MM. For instance, miR143 and miR-29a promote angiogenesis and osteoblast differentiation.88,89 Moreover, miR-199 is an antioncogene in BM stromal cells. 88 Additionally, miR-92a regulates oral squamous cell carcinoma cell growth by targeting FOXP1 expression. 90 Its overexpression induces EMT by regulating PI3K/AKT signaling via targeting PTEN, promoting NSCLC cell migration, invasion, and tumor growth. 91 Our analysis underscores the potential utility of circulating miRNA in body fluids for clinical prognostics, diagnostics, and therapeutics. Consequently, the data indicate that specific miRNA expression profiles can serve as biomarkers for monitoring and treatment of disease.
Diagnostic circulating miRNA biomarkers
Eight studies have examined the correlation between circulating miRNA and disease diagnosis, focusing on evaluating the expression levels of miRNA signatures and their diagnostic significance in MM with diagnostic methods and associations, as shown in Table 1. In 2014, Kubiczkova et al. 71 conducted a global analysis of serum microRNA profiles utilizing TaqMan Low-Density Arrays, followed by validation through quantitative real-time PCR using 208 participants (MM patients, n = 121; MGUS patients, n = 57; healthy individuals, n = 30) and discovered five serum-deregulated miRNAs in MM and MGUS: miR-744, miR-130a, let-7d, and let-7e (p < 0.001), all of which were significantly downregulated. 71 Meanwhile, miR-34a (p < 0.0001) was found to be upregulated in both conditions. 71 Their research shows that miR-34a and let-7e can distinguish MM from healthy donors with 80.6% sensitivity and 86.7% specificity, as well as identify monoclonal gammopathy of undetermined significance (MGUS) with 91.1% sensitivity and 96.7% specificity. 71 The analysis indicated that miR-34a and let-7e effectively differentiate MM patients from healthy controls (AUC = 0.898) and those with MGUS (AUC = 0.976). 71 By analyzing the levels of these miRNAs in a patient’s blood, doctors can identify the presence of MM with a high degree of accuracy. Additionally, the overexpression of Let-7e increases the transcription factors Pre-B cell leukemia transcription factor 1 (PBX1) and CCAAT enhancer binding protein alpha (CEBPA), which are associated with MM pathogenesis. 89 Let-7e, located on chromosome 19q13.33, targets genes such as MAPK6, IGF1, MYCN, CDK6, APC2, and TP53 in ALL. 71 In contrast, let-7d expression fell compared to initial diagnosis levels. 71 Let-7d, situated on chromosome 9q22.32, specifically targets the genes APC2, TGFβRI, CDC25A, and TP53, and is associated with AML. 71 A study involving 18 relapsed MM patients revealed a significant increase in the expression levels of miR-34a. This research links miR-34a to myeloproliferative neoplasms via the miRWalk database, identifying several target genes, including MYCN, E2F3, BCL2, and CDK6, all of which are located on chromosome 1p36.23. 71 Furthermore, the upregulation of miR-34a in MM acts as a tumor suppressor by inhibiting cancer cell proliferation, promoting apoptosis, and limiting the properties of cancer stem cells.92,93 MiR-34a modulates target proteins that influence the cell cycle, differentiation, and apoptosis, inhibiting processes essential for cancer cell survival, stemness, metastasis, and chemoresistance. 93 In contrast, Soliman et al. 94 emphasized the role of decreased miR-34a expression in MM, which inhibits apoptosis and promotes prosurvival signaling pathways, primarily by modulating CDK6, BCL2, TGIF2, and NOTCH1. In chronic lymphocytic leukemia (CLL), miR-130a acts as a negative regulator, targeting specific mRNAs (ATG2B and DICER1) and influencing key signaling pathways in disease progression. 92 Patients with extramedullary myeloma exhibited significantly lower levels of circulating miR-130a compared to individuals with newly diagnosed, relapsed, or progressing MM. 69 Moreover, serum levels of miR-130a effectively distinguished MM patients with extramedullary disease from healthy controls and from those with newly diagnosed MM. 95 The miRWalk database indicates that miR-130a is associated with several target genes, including IGF1, CCND2, and TGFβ, all of which are located on chromosome 11q12.13. 71 At the same time, decreased miR-744 through the SOX2/Wnt/β-catenin pathway enhances MM cell proliferation, migration, and invasion, affecting cancer progression. 96 Similarly, mir-744 enhances the tumorigenic potential of pancreatic cancer cells by activating the Wnt/β-catenin signaling pathway. 97
A study found that serum miR-4449 expression levels were observed in 71 MM patients and 46 healthy controls. RT-qPCR indicated that miR-4449 levels were significantly higher in MM patients than in healthy controls (AUC of 0.885, 95% CI: 0.826–0.945), making it a valuable tool for the auxiliary diagnosis of MM compared to other markers. 72 Jiang et al. 73 identified eight upregulated miRNAs in MM patients: miR-125b-5p, miR-483-3p, miR-4326, miR-6894-3p, miR-4498, miR-490-3p, miR-7155-5p, and miR-937-3p serum samples were analyzed using 20 healthy controls and 35 MM patients. Both miR-125b-5p and miR-490-3p demonstrated significant diagnostic accuracy in distinguishing MM patients from healthy subjects, yielding areas under the curve (AUCs) of 0.954 (sensitivity = 86%, specificity = 96%, 95% CI: 0.901–1.006, p < 0.001) and 0.866 (sensitivity = 60%, specificity = 85%, 95% CI: 0.522–0.836, p = 0.028). 73 Elevated levels of miR-125b play a role in MM by lowering p53 levels and blocking apoptosis. 14 While higher miR-125 levels also increase miR-34a expression. 14 It was observed that elevated miR-490-3p increases ERGIC3 expression and decreases PCBP1 activity, promoting multiplication and migration while impairing the immune system response. 98 This research indicates that miR-490 is overexpressed in MM samples, highlighting its potential as an early tumor biomarker. 98
Studies on tumor suppressor miRNAs revealed that the downregulation of miR-143, miR-144, miR-199, and miR-203 are associated with a poor prognosis for MM patients. 20 It was noted that the relative expression of miR-203 on chromosome 14q32.33 targets CREB1 99 and VCAN. 20 The dysregulation of miR-203a expression is implicated in the pathogenesis and progression of various malignancies, including gastric, ovarian, colon, and lung cancers, as well as chronic myelogenous leukemia (CML) and other neoplastic diseases.100–103 Additionally, miR-143, located on chromosome 5q32, targets HDAC7, promotes angiogenesis and osteoblast differentiation. Similarly, miR-144 on chromosome 7q11.2 targets the MEF2A gene, suppressing proliferation and angiogenesis while aiding in apoptosis. 103 miR-199 on chromosome 9q34.11 acts as an antioncogene within the BM microenvironment by targeting downstream signaling VCAN through FAK and STAT3 activation. 20 Zhang et al. 74 conducted a comparative analysis of circulating miRNA levels in serum samples from three groups: patients with clinically manifest MM (n = 20), smoldering multiple myeloma (SMM) (n = 20), and healthy controls (n = 16). 74 Their findings indicated that MM patients showed significantly reduced levels of let-7d-5p alongside increased levels of miR-4741 compared to healthy individuals. In the SMM cohort, there was a notable decrease in miR-103-3p, while miR-4741 levels remained elevated relative to controls. 74 Furthermore, the study identified overexpression of miR-103-3p, miR-140-3p, and miR-425-5p in the MM group. At the same time, a decrease in miR-4505 expression levels was observed in the SMM group. 74 The let-7 miRNA family plays a crucial role in the pathogenesis of MM and the progression from SMM to MM, particularly through its influence on MYC gene overexpression, which is a known driver of malignancy in this context. 74 This ultimately leads to increased cell proliferation and migration and a disrupted cell cycle. 74 This suggests that these specific miRNAs, including the let-7 family, could be potential diagnostic biomarkers or therapeutic targets for MM.
The upregulation of miR-134-5p, miR-107, and miR-15a-5p reveals distinct miRNA expression profiles that differentiate MGUS from MM compared to healthy controls, suggesting their potential as diagnostic markers. Lie et al. validated the diagnostic efficacy of a combined approach using miR-107 and miR-15a-5p alongside hemoglobin (Hb), achieving an AUC of 0.954 (95% CI: 0.890–1.000). 75 This model demonstrated a sensitivity of 91.3% and a specificity of 93.7%, effectively differentiating MM from MGUS. These findings indicate that miR-134-5p, miR-107, and miR-15a-5p may serve as valuable diagnostic biomarkers in the clinical management of MM and MGUS. 75 Moreover, integrating miR-107 and miR-15a-5p with Hb can enhance the differentiation between MM and MGUS, potentially facilitating earlier therapeutic interventions and improving patient prognosis. 75 Notably, miR-15a-5p, residing on chromosome 13q14.2, targets critical regulators such as BCL-2, VEGF-A, PHF19, cyclin D1, cyclin D2, and CDC25A, P13K-Akt pathway thereby inhibiting cell proliferation and modulating apoptosis. 104 The study indicates that specific miRNAs, particularly when combined with Hb levels, can effectively distinguish between MGUS and MM, offering a promising tool for improved diagnosis and potentially earlier treatment.
Another study shows the upregulation of the proliferation of MM, distinguishing newly diagnosed MM patients from healthy donors using ROC analysis, demonstrating 92.90% sensitivity, 87.50% specificity, a 95% confidence interval, and p < 0.001, advocating for its use as a diagnostic biomarker of MM. 76 Additionally, the diagnostic utility of miR-448 expression has been established as markedly higher in MM patients than in individuals without the condition, with varying expression across distinct MM stages. 76 Notably, elevated levels of mir-448 were found in newly diagnosed MM with similarly high concentrations of serum M protein; this aids its potential as diagnostic value. 76
In medical research, specific miRNAs epitomize notable prognostic and diagnostic significance. Notably, in the context of MGUS and MM, miRNAs, such as miR-744 and let-7e, present in circulating serum, have demonstrated potential as markers for diagnostic and prognostic purposes relevant to the survival of MM patients. 71 Furthermore, a study by Zhao et al. 77 has reported substantially higher serum miR-1246 levels in newly diagnosed MM patients compared to the control group, with decreased levels during remission and increased levels during MM relapse (p < 0.05). Recent evidence shows miR-1246 targets CXC motif chemokine receptor type 4 (CXCR4), inhibiting cell migration and invasion in renal cell carcinoma. 105 CXCR4 is linked to accelerated BM dissemination as a G protein-coupled receptor. 106 Thus, miR-1246 may promote extramedullary infiltration in MM by regulating CXCR4. 106 Table 1 details the sample size, objectives, methods, miRNA, results, targets, and references for the studies utilized in this section.
Prognostic circulating miRNA biomarker
The expression of circulating miRNAs in patients with MM has been examined for its predictive value in the context of drug resistance and disease prognosis. 22 This review synthesizes nine studies investigating the interaction of circulating miRNAs that are prognostic circulating miRNA biomarkers in MM, as illustrated in Table 1.
In 2014, Kubiczkova et al. 71 found that lower levels of miR-744 and let-7e correlated with reduced survival and remission rates in myeloma patients. One-year mortality rates were 41.9% for low-expressing miR-744 and 34.6% for low-expressing let-7e, respectively, compared to 3.3% and 3.9% in high-expression groups. 71 The median remission duration was 11 months for patients with low miR-744 and let-7e levels versus about 47 months for those with high expression. 71 These results link altered miRNA patterns in MM and MMGUS to myeloma outcome survival. 71 The dysregulation of miR-744 on chromosome 17p12 results in a poor prognosis. Deletions in 17p13.1, where TP53 is located, further exacerbate the prognosis for MM. 95 Decreased levels of let-7 microRNA are associated with disruptions in the cell cycle and enhanced proliferation of MM due to the upregulation of oncogenes such as RAS, HMGA2, c-Myc, and CCND1.78,107 Let-7 typically suppresses these oncogenes by binding to their 3ʹ UTRs, inhibiting translation. 78 Low let-7 levels remove a vital barrier to cell growth, leading to uncontrolled proliferation. 78 These results emphasize dysregulated miRNAs in MM and MGUS, highlighting the prognostic significance of miR-744 and let-7e for patient survival.
Qu et al. 78 investigated plasma biomarkers in MM and examined the expression levels of miR-483-5p and miR-20a in a cohort of 40 patients compared to 20 healthy controls. The ROC analysis indicated that miR-483-5p produced an AUC of 0.745, with a sensitivity of 58% and specificity of 90%. 78 In contrast, miR-20a yielded an AUC of 0.740, with a sensitivity of 63% and specificity of 85%. 78 Notably, miR-483-5p levels significantly correlated with the patients’ international staging system (ISS) stages. 78 For those with elevated miR-483-5p levels, the median progression-free survival (PFS) was 15 months, significantly shorter than the 21 months for those with lower levels (p = 0.025). 78 Conversely, no significant association was found between miR-20a levels and PFS (p > 0.05). 78 These findings suggest that miR-483-5p may serve as a promising prognostic biomarker for survival outcomes in MM. 79
Some miRNAs have been shown to exhibit oncogenic or tumor-suppressive characteristics depending on their molecular pathways and the silenced genes. 65 For example, in 2015, Wang et al. 79 explicated the critical role of aberrant expression of let-7a, let-7b, let-7i, miR-15b, miR-16, and miR-20a in tumorigenesis and amelioration. More than 60% (11 of 18) of the miRNAs showed significantly lower serum levels in MM patients than controls. 79 The let-7 miRNA family and miR-15b/16 clusters are downregulated in malignant myeloma, suggesting their role as tumor suppressors, thus reducing disease progression. 79 The validated targets of miR-15b include the mRNAs MAP2K4 and MAPKAPK5. 79 In contrast, miR-16 targets BCL2, MCL1, CCND1, and WNT3A. 78 Let-7 miRNAs target key transcripts in oncogenesis and cell cycle regulation, including oncoproteins such as RAS, MYC, HMGA2, and pluripotency factor LIN28—these miRNAs significantly impact myeloma pathogenesis. 108
Hoa et al. 80 reported that patients with bone disease exhibited elevated levels of circulating miR-214 and miR-135b, which correlated with severe lytic bone disruption and were associated with poorer PFS and overall outcome survival. More specifically, higher expression of miR-214 directly correlates with significantly shorter PFS (median: 8 months compared to 22 months) and overall survival (OS) (median: 15 vs 28 months).80,109 This poor prognosis is linked to the activation of the PI3K/Akt pathway and the severe neoplasm.109,110
In addition, a study on newly diagnosed MM patients revealed that the downregulation of miRNAs, let-7b and mir-18a, was linked to poor PFS and OS.3,109 This study included serum from MM (n = 156), which is significantly associated with decreased PFS (HR = 1.9, 95% CI: 1.22–2.94 for miR-18a; HR = 2.76, 95% CI: 1.79–4.26 for let-7b) and OS (HR = 2.83, 95% CI: 1.07–7.5 for miR-18a; HR = 4.52, 95% CI: 1.57–12.98 for let-7b).3,97 They demonstrated that Let-7 miRNAs act as tumor suppressors in MM by inhibiting oncogenes such as CCND1, MYC, and RAS.3,109 Meanwhile, miR-18a induces M1 macrophages by directly targeting IRF2 and NK cells.3,109
Ren et al. 81 reported findings consistent with those of Zhao et al., 77 indicating that miR-1246 and miR-720 were significantly upregulated in MM patients (n = 60) compared to a healthy control group (n = 16). Additionally, the expression levels of these miRNAs showed a positive correlation (rho = 0.621). 81 Importantly, the overexpression of both miRNAs in MM patients was linked to a significant decrease in PFS, with median durations of 12.4 months in the high-expression group compared to 17.8 months in the low-expression group.81,109
Moreover, miR-8074, located on the long arm of chromosome 19 at location 19q13.41, exerts regulatory control over pivotal genes associated with MM pathogenesis, including TP53, MYC, MAPK1, and KIAA. 82 Elevated levels of miR-8074 may indicate disease severity, especially in tandem with other established diagnostic indicators such as stage III Durie Salmon, renal failure, decreased albumin, elevated β2-microglobulin, LDH, and calcium. 82 Higher concentrations of MiR-8074 were linked to shorter PFS (17 months compared to 39 months; HR = 2.67, 95% CI: 1.15–6.20, p = 0.0231) and overall survival (30 months vs not reached; HR = 5.25, 95% CI: 1.38–19.97; p = 0.0155).82,109
In another study, elevated levels of miR-25-3p in the serum of MM patients (n = 69) serve as a noninvasive marker for poorer prognosis. Higher miR-25-3p was linked to lower OS, with a median OS of 25.8 vs 30.7 months.83,109 A multivariate analysis showed a hazard ratio (HR) of 4.1 (95% CI: 0.79–21.32). Still, no correlation was found for PFS.83,109 miR-25 inhibits p53 signaling, reducing apoptosis and cell senescence by targeting TP53 and silencing the histone acetyltransferase PCAF. 111 Additionally, MYC-mediated overexpression of miR-25 is associated with dexamethasone resistance in MM due to its interactions with ULK1 and p27 (CDKN1B).110,112 It also targets PTEN to activate the PI3K/AKT pathway, thereby promoting the proliferation of MM cells while decreasing apoptosis. 112 These studies have linked miRNA dysregulation with disease profiling, thus supporting its potential utility as a prognostic biomarker in MM. Table 1 provides information about the sample size, objectives, methods, miRNA, results, targets, and citations for the studies referenced above.
Therapeutic circulating miRNA biomarkers
Detecting specific circulating miRNAs holds promise for predicting treatment responsiveness in various cancer types, including MM. This thematic review analyzes five studies exploring the interplay between circulating miRNAs and therapeutic strategies in various diseases. The findings indicate that miRNAs function as oncogenes and tumor suppressors, highlighting their dual roles in cancer biology.
Hao et al. 84 observed a reduction in miR-19a levels at diagnosis and a reappearance of lower levels at relapse. This suggests that the dysregulation of miRNAs may influence patient conditions and disease progression. This observation is supported by findings that higher levels of miR-4254 are associated with increased relapse rates. Conversely, MM patients with low miR-19a have enhanced survival outcomes with BTZ-based treatment. 84 Additionally, it has been suggested that combining miR-19a and miR-4254 can be a robust prognostic tool for differentiating MM from healthy donors with high precision. 84 This study also demonstrated a link between 1q21 amplification, drug resistance, and ISS staging. 84 Notably, low levels of miR-19a showed a positive correlation with a 14q32 deletion, indicating that lower levels of miR-19a, combined with 14q32 chromosomal loss, may contribute to better outcomes. 84 Elevated levels of miR-19a were linked to poorer survival rates. 84 Further analysis revealed correlations between miR-19a expression and specific genetic markers such as t(4;14), del(17p), and t(11;14).115 MiR-19a expression was notably higher in patients with t(4;14) while showing reduced levels in those with t(11;14) and del(17p). 84 These patterns indicate a potential prognostic and therapeutic significance of miR-19a expression in patient stratification and the advancement of treatment strategies for patients demonstrating high miR-19a levels expression. 84 Furthermore, the levels of miR-19a expression may offer insights into patient responses, highlighting the significance of its biological pathways, especially the suppression of PTEN and the activation of the PI3K/AKT/mTOR signaling cascade. 113 These findings highlight the necessity for further exploration to validate miR-19a’s role as an essential biomarker for treatment response and prognosis.
Zhang et al. 85 isolated nano-sized exosomes from bortezomib-resistant patients, which showed significantly increased internal RNA content and downregulation of exosomal miR-16-5p, miR-15a-5p, miR-20a-5p, and miR-17-5p. Routine MM workups provide minimal insight into drug resistance prediction. 85 This investigation examines the emerging role of circulating exosome-associated miRNAs in the development of drug resistance (DR) in MM. The study cohort included 204 patients. Drug resistance rates were 36.5% in the bortezomib-containing group, 73.1% in the thalidomide-containing group, and 81.8% in the lenalidomide-containing group. Elevated serum total light chain ratio (⩾100), high C-reactive protein (CRP) levels (⩾20 mg/L), and the use of second-line therapies were identified as risk factors for acquired bortezomib resistance. 83 Among the 68 patients with available genetic data, 1q21 amplification was recognized as a high-risk factor for predicting de novo DR. 83 It was linked to reduced serum cholesterol and LDL-C levels. 83 Circulating exosome-associated miRNAs offer a fresh perspective for investigating in vivo intercellular communication within the MM microenvironment and may help predict drug resistance.
A study by Tian et al. 86 emphasized the significance of miR-497 in inhibiting tumorigenesis by enhancing BTZ sensitivity through targeting B-cell lymphoma 2 (Bcl-2) proteins, which regulate apoptosis and improve cell viability, thereby enhancing treatment efficacy. miR-497 overexpression promoted MM cell apoptosis through cell cycle arrest, decreased colony formation, and cell viability. 86 It also increased the sensitivity of MM cells to bortezomib, suppressing proliferation by targeting Bcl-2 and altering downstream apoptosis-related proteins. 86 Combining miR-497 with bortezomib may enhance drug sensitivity, offering a potential therapeutic method for MM. 86
Robak et al. 10 conducted the first pilot study to identify a blood-based miRNA signature that predicts the efficacy of bortezomib-based therapy in patients with MM. This study is the inaugural investigation revealing differential serum expression profiles of several miRNAs between bortezomib-refractory MM patients and those exhibiting drug sensitivity. These findings could be pivotal for future research focused on addressing and overcoming drug resistance in MM therapies.
This research identified 21 miRNA profiles in MM patients responding differently to BTZ, listed in Table 1 as a panel of differentially expressed miRNAs. More specifically, the collective expression levels of miR-215-5p, miR-376c-3p, and miR-181a-5p indicate a patient's potential response to bortezomib treatment. 10 This study observed higher serum levels of miR-215-5p, miR-143-3p, miR-19b-3p, miR-16-2-3p, miR-122-5p, miR-148a-3p, miR-22-5p, miR-30e-5p, and miR-29b-3p in the BTZ-refractory cohort compared to the BTZ-sensitive group. In contrast, levels of miR-744-5p, miR-151a-3p, miR-376a-3p, miR-191-5p, miR-181a-5p, miR-766-3p, miR-30c-5p, miR-130a-3p, miR-409-3p, miR-1224-3p, and miR-328-3p were lower in the refractory group than in the sensitive patients. Moreover, several studies have linked these miRNAs to the proliferation of MM and refractoriness to BTZ.
To illustrate, the research shows a negative correlation between miR-215-5p and RUNX1 expression in MM BM. 114 MiR-215-5p overexpression enhances MM cell proliferation and inhibits apoptosis by targeting RUNX1 and suppressing the PI3K/AKT/mTOR signaling, suggesting a dual role as an antioncogene, while promoting G1/S progression. 114 MiR-30e-5p differentiates MM with 1q21 gains 115 and varies in expression between patients with and without extramedullary relapse. 10 MiR-29b, a tumor suppressor, plays a role in BTZ resistance by targeting MCL-1 via the lncRNA H19/miR-29b-3p/MCL-1, a promising target for addressing drug resistance in MM treatment. 116 Six upregulated miRNAs (miR-143-3p, miR-19b-3p, miR-16-2-3p, miR-122-5p, miR-148a-3p, miR-22-5p) in bortezomib-refractory patients have not been well studied in MM but are linked to other cancers. MiR-143-3p inhibits proliferation, migration, and invasion by targeting FOS-like antigen 2 (FOSL2) expression in osteosarcoma 117 and proliferation in several cancers.118–120
Upregulation of miR-19b-3p is linked to tumorigenesis, as well as to carcinoma chemoresistance to oxaliplatin-based drugs, by inhibiting SMAD4 in colon cancer, reflecting the upregulation observed in bortezomib-refractory MM. 121 miR-19b-3p suppresses cell growth and reverses cell resistance to saracatinib by regulating PIK3CA in breast cancer. 122 Additionally, it influences radiosensitivity via the regulation of TNFAIP3 in nasopharyngeal carcinoma 123 and serves as a novel prognostic marker in colon cancer. 121 miR-16-2-3p has tumor-suppressive and antimetastatic properties. 124 Downregulation of miR-122-5p serves as a prognostic factor and therapeutic target in cancers, acting as a tumor suppressor that targets LYN, an Src family kinase, which inhibits the proliferation, migration, and invasion of gastric cancer. 125 Also, the downregulation of miR-22-5p contributes to the malignant progression of nonsmall-cell lung cancer (NSCLC) cells by targeting TWIST2. 126 miR-148a-3p, which influences differentiation and B cell development, plays a role in inhibiting the transcription factors BACH2 and MITF. 127
miR-181a-5p has been extensively studied in several cancers, including breast cancer, colorectal cancer, prostate cancer, retinoblastoma, pancreatic cancer, nonsmall cell lung cancer, laryngeal cancer, ovarian cancer, bladder cancer, thyroid cancer, and leukemia myeloma. 128 MiR-181a-5p is a potential therapeutic target for MM. 128 It is associated with lower CDK2, Cyclin E1, Bcl2, and higher p21, Bax, and caspase 3, regulating MM cell proliferation and apoptosis by inhibiting the Hippo/YAP axis. 129 The findings indicate the inhibitory role of miR-181a-5p in MM. 128 In MM, miR-744-5p acts as a tumor suppressor by inhibiting myeloma cell proliferation, invasion, migration, and glycolysis through the SOX12/Wnt/β-catenin pathway. 96 Consequently, reduced levels of miR-744-5p are linked to increased disease progression, highlighting its potential as a therapeutic target. 96 miR-376a is a tumor suppressor inhibiting proliferation and invasion of nonsmall-cell lung cancer by targeting c-Myc. 130 MiR-191-5p promotes tumor progression; higher levels are associated with advanced stages and metastasis of hepatocellular carcinoma. 131 MiR-766-3p downregulates BCL9L, acting as a tumor metastasis suppressor, influencing the β-catenin signaling pathway in osteosarcoma progression. 132 MiR-30-5p is a tumor suppressor and provides a novel therapy by inhibiting the Wnt/β-catenin/BCL9 pathway. 133 It is associated with increased proliferation, survival, migration, drug resistance, and the development of cancer stem cells in MM. 133 MiR-130a-3p regulates tumor progression and metastasis with context-dependent downregulation effects. 134 These findings highlight the differing miRNA expression patterns between refractory BTZ and those sensitive to which is useful for mediating resistance to BTZ in MM.
Interestingly, elevated pretreatment plasma levels with overexpression of the mir-25 gene (7q22.1) have been associated with poor overall and PFS. 83 Mir-25, encoded by the mir-25 gene, obstructs the p53 signaling pathway, averts apoptosis and cell senescence by directly targeting TP53 expression and indirectly silencing the histone acetyltransferase PCAF, a potent coactivator of p53. 111 Furthermore, cancer-suppressive miRNA, such as small RNA, presents the potential for in vivo cell targeting and could yield favorable therapeutic outcomes. 135 A recent study highlighted increased expression levels of let-7b-5p and miR-27b-3p in BTZ-resistant MM patients compared to BTZ-sensitive MM patients, suggesting that these miRNAs could serve as biomarkers for treatment response in MM. 87 Both miRNAs have been shown to regulate proliferation, apoptosis, and drug resistance by targeting specific genes and signaling pathways. 136
Limitations of circulating miRNAs as biomarkers
The utility of circulating miRNA as noninvasive biomarkers in MM’s prognostication, diagnosis, and therapeutic management has garnered considerable attention. Nonetheless, there remains to be contention regarding the specificity and sensitivity of these short miRNA sequences in faithfully representing the dynamics between the serum and the tumor. 137 Complicating therapeutic challenges are plasma cells’ clonal heterogeneity and genomic instability, thereby fostering intrinsic and acquired therapeutic resistance. 138 Furthermore, an evaluation of miRNA overexpression is imperative for discerning the implications of the oncogene in the progression of MM. 10 The potential of miRNAs as therapies lies in their ability to modulate gene expression, inhibit tumor growth, and enhance treatment efficacy. 14
Moreover, concurrent health conditions can, it is speculated, influence the biological properties of circulating miRNA, resulting in differential expression levels among individuals. 137 Additionally, the distinctive nature of testing methodologies, such as RNA sequencing, quantitative reverse transcription polymerase chain reaction (qRT-PCR), northern blot, western blot, and microarray, is subject to intrinsic diagnostic limitations, notably the costly nature of reagents.
In addition, bioinformatics algorithms represent a powerful tool for miRNA analysis, employing computational methods, including mathematical and statistical techniques, to address biomedical queries. 139 The primary objective of the bioinformatics approach is the comprehensive storage of molecular biology data in database formats. 139 Bioinformatics algorithms are poised to impact the identification or validation of individual miRNAs significantly, the prediction of miRNA target sequences, the determination of miRNA expression, the exploration of miRNA signaling pathways and metabolic pathways, the analysis of regulatory networks of miRNAs, the identification of MiR-transcription factor interplay, and the investigation of miRNA links to human disorders. 139 The abnormal expression of miRNAs in tumors can impact various cancer hallmarks, influencing tumor development and progression. 19 Bioinformatics offers a comprehensive tool for identifying dysregulated miRNAs and their associated messenger RNA targets. The human genome database contains over 2588 mature miRNAs, facilitating target predictions through programs such as PITA, Tarbase, rna22, MiRDB, STarMir, microInspector, Mirpath, MaMi, MiRecords, mirDIP, miRGator, Magia, miRTar, and miR, each employing distinct modes. 140 While this technology allows for expedited evaluation of miRNA signatures and targets, it faces challenges due to consistency in accuracy and sensitivity. 141 Computational errors in sequencing may occur, given that the constructions of this evidence-based molecular network rely on experimental data from various sources, including protein–protein interactions, gene regulatory relationships (including miRNAs), and metabolic pathways. These factors collectively play a crucial and enlightening role in understanding cancer biology. Hence, a comprehensive understanding of intricate molecular interactions in cancer can be attained through high-throughput experimental techniques and databases like Gene Ontology, 142 REACTOME, 143 the Kyoto Encyclopedia of Genes and Genomes (KEGG), 144 and Galaxy. 145 These resources contain valuable information that enables a comprehensive analysis of cancer-related molecular dynamics.
Challenges in using miRNA for therapeutics
Employing miRNA for therapeutic applications presents significant challenges, particularly regarding suboptimal delivery mechanisms and associated toxicity, which can trigger adverse immune responses. The accurate identification of specific targets is key to developing miRNA-based therapies. 146 This complexity arises because miRNAs interact with a limited range of sequences, making functional validation labor-intensive and costly. 146 Current methods are inadequate for comprehensively predicting all affected cellular pathways and potential binding sites. 146 Moreover, effective miRNA therapies must avoid interactions with genes crucial for maintaining cellular homeostasis, underscoring the necessity for a thorough understanding of miRNA targets and their operational mechanisms. Notably, a clinical trial was approved using nanocarriers—such as lipid-based and cationic polymer-based carriers—and miR-34 therapy (MRX34) as a monotherapy for patients with MM. However, there were challenges in therapeutic delivery, reducing toxicity, and mitigating off-target effects that were not addressed while preserving the integrity of homeostatic cells, which resulted in its discontinuation. Once these challenges are overcome, the miRNAs will be useful in personalized patient care.
Conclusion
MiRNAs exhibit significant potential as noninvasive clinical biomarkers due to their stability and presence in various biofluids such as blood, cerebrospinal fluid (CSF), urine, saliva, and tear fluid. Numerous studies indicate that miRNAs are essential for continuously monitoring disease progression, staging, and treatment efficacy. This method provides real-time insights that are critical for the early detection of cancer evolution, thereby enhancing effective management strategies. This methodology offers real-time insights that are critical for the early detection of cancer evolution, thereby enhancing effective management strategies. Extensive research underscores the pivotal role of miRNA dysregulation in MM, influencing key signaling pathways, apoptosis, and cell proliferation. This thematic review is systematically organized into the following sections: BTZ resistance in MM, the predictive role of miRNAs in BTZ resistance, the impact of circulating miRNAs in MM, and the potential of circulating miRNAs as prognostic, diagnostic, and therapeutic biomarkers. Nine studies suggest that specific circulating miR-15b could function as prognostic biomarkers. Specifically, reductions in let-7a, let-7b, let-7i, miR-15b, miR-16, and miR-20a are correlated with tumorigenesis, while increases in miR-214 and miR-135b are associated with myeloma bone disease and the development of MM. Furthermore, eight studies have identified specific circulating miRNAs as potential diagnostic biomarkers, targeting distinct genes. For instance, the downregulation of miR-16 influences AKT3, MAPK, rp-S6, NF-kB, and VEGF, inhibiting cell proliferation and growth in MM.
In contrast, upregulated miR-1246 significantly impacts critical genes such as TP53, MYC, MAPK1, and KIAA, indicating a poor prognosis. In addition, five articles have recognized circulating miRNAs as promising therapeutic biomarkers. For example, the downregulation of miR-19a and miR-4254 has significantly improved survival outcomes in bortezomib-based therapies and may facilitate the monitoring of myeloma progression. These findings strongly support the incorporation of miRNA into personalized medicine, enhancing capabilities for early detection, precise diagnosis, prognostication, and overall survival rates. Considering this evidence, exploring circulating miRNAs as potential noninvasive biomarkers for MM signifies a noteworthy advancement in MM’s prognostic, diagnostic, and therapy. This assertion is bolstered by the abundance of miRNAs in plasma or serum, demonstrating remarkable stability against enzymatic degradation.
Footnotes
Acknowledgements
Not applicable.
Ethical considerations
Not applicable.
Author contributions
SB-C wrote the first draft of the manuscript and analyzed the data; JS and ZAK conceptualized, edited, and finalized the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
