Abstract
We present a case of severe and atypical Drug Reaction with Eosinophilia and Systemic Symptoms/Drug-induced hypersensitivity Symptoms (DRESS/DiHS), also known as DiHS, in a 46-year-old Chinese woman, triggered by bortezomib (Velcade), a proteasome inhibitor, commonly used for the treatment of multiple myeloma. Bortezomib-induced DRESS/DiHS syndrome is rare, with this being the second case documented in the literature. The patient presented with severe erythroderma with ichthyosis-like features, hypereosinophilia, and liver injury. Initial response to high dose of solumedrol was followed by relapses upon switching to oral prednisone. Ultimately, the patient responded to dupilumab and maintained complete remission.
Keywords
Introduction
Bortezomib (Velcade®) is a proteasome inhibitor used to treat multiple myeloma, administered in a 3-week cycle.1,2 Common side effects include fatigue, gastrointestinal symptoms, thrombocytopenia, and peripheral neuropathy. 1 Infrequently, it causes cutaneous hypersensitivity reactions.
Drug reaction with eosinophilia and systemic symptoms (DRESS), also referred to as drug-induced hypersensitivity syndrome (DiHS), is a severe and potentially life-threatening idiosyncratic reaction with up to 10% mortality. 3 It is marked by a latency period of 2 to 8 weeks after drug exposure. 4 Cutaneous manifestations are commonly the initial presentation in DRESS/DiHS, characterized by widespread red, raised, and infiltrated papules and plaques, often accompanied by fever. 5 The eruption can involve significant areas, including the trunk and limbs. Facial edema and erythema are highly characteristic. 6 Although dry, cracked lips are typical, mucosal involvement is rare. Lymphadenopathy and multi-organ inflammation, such as the liver or kidneys, may also occur. 5
To date, only one case of bortezomib-induced DRESS/DiHS has been reported. 7 We report a severe and atypical case of ichtyosiform DRESS/DiHS secondary to bortezomib in a patient with multiple myeloma. Despite initial responsiveness to high-dose solumedrol, the patient experienced relapses upon transitioning to oral prednisone but achieved remission with dupilumab, a monoclonal antibody targeting IL-4Rα. We discuss the challenges encountered during treatment and highlight its successful management with dupilumab.
Case report
We report a case of a 46-year-old Chinese woman hospitalized following four DRESS/DiHS flare-ups with severe erythrodermic ichthyosis-like skin eruptions triggered by bortezomib. Diagnosed with multiple myeloma, she underwent treatment with 2 cycles of VRD (Velcade (Bortezomib), Revlimid (Lenalidomide), Dexamethasone) followed by 3 cycles of Cybord (Cyclophosphamide, Bortezomib, and Dexamethasone). Her first episode persisted for 5 months and included a drug-induced hepatitis. Each episode was characterized by elevated hepatic enzymes, hypereosinophilia, widespread rash, and facial edema.
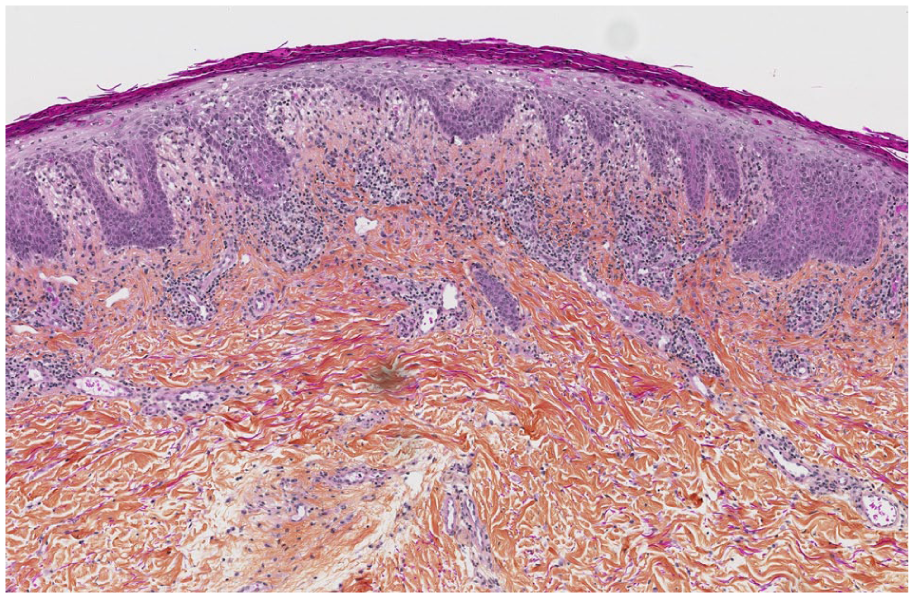
Upon presentation, she exhibited severe erythroderma affecting 97% of her total body surface area (TBSA), sparing her genitals (Figure 1). Erythematous-squamous psoriasiform plaques were observed on her scalp, accompanied by an extensive pan-corporeal ichtyosiform desquamation, particularly in the skin folds (Figure 2). She also had severe facial edema impairing eyelid opening. This atypical DRESS/DiHS presentation has not been previously documented. Biopsies from the left superior arm revealed interface dermatitis with keratinocyte necrosis affecting total epidermal layers, accompanied by a polymorphous inflammatory infiltrate of the superficial dermis, rich in eosinophils (Figures 3 and 4). Another biopsy of the left lower limb revealed psoriasiform dermatitis with spongiosis and multi-stage necrotic keratinocytes compatible with a drug-induced reaction (Figures 5 and 6). Perioral fissures, palmoplantar keratoderma, distal onycholysis, and hyperkeratosis nails were also apparent (Figures 2, 7, and 8). There was no mucosal involvement, lymphadenopathy, and the Nikolsky sign always remained negative. Prior to this hospitalization, she had four previous DRESS/DiHS episodes. The initial episode revealed a drug-induced hepatitis, followed by another hospitalization for another drug-induced hepatitis with deteriorating liver injury (ALT: 500, eosinophilia 2.7 × 109/L). During the third episode, the skin eruption notably worsened. Later, she reconsulted for fever and a progressing eruption. Upon each episode, tapering oral prednisone led to relapses with recurrent symptoms, marked by elevated ALT enzymes, hypereosinophilia, along with extensive deteriorating skin eruption.
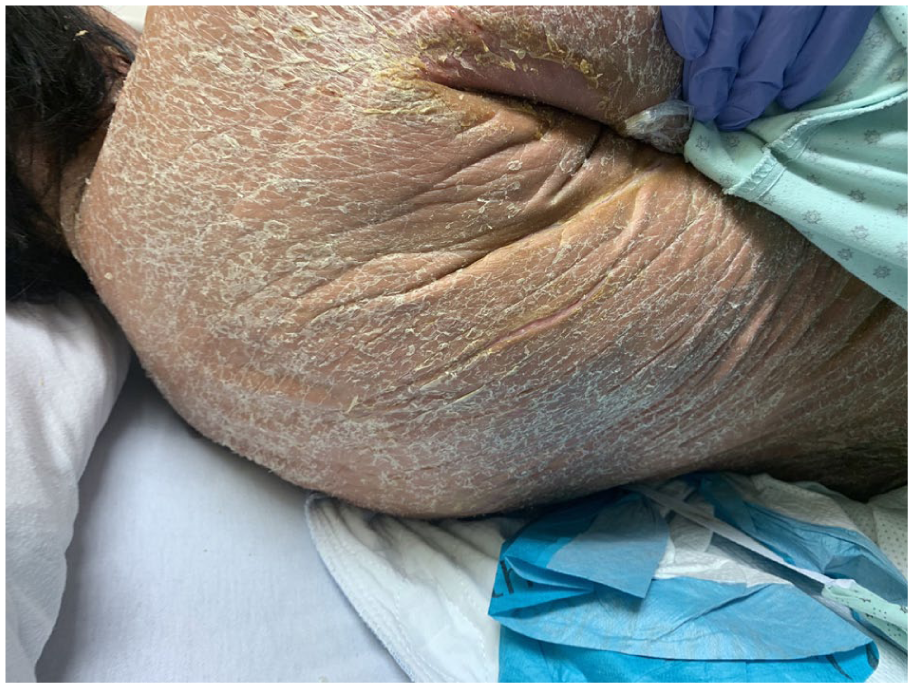
Diffuse ichtyosiform desquamation with severe hyperkeratosis is notable in the skin folds.

Extensive pan-corporeal ichthyosiform desquamation, particularly in the neck skin folds, along with perioral radial fissures, notable facial edema, and impetiginization of the skin folds.

High-power examination reveals interface dermatitis with keratinocyte necrosis involving the full thickness of the epidermis with a polymorphous inflammatory infiltrate.
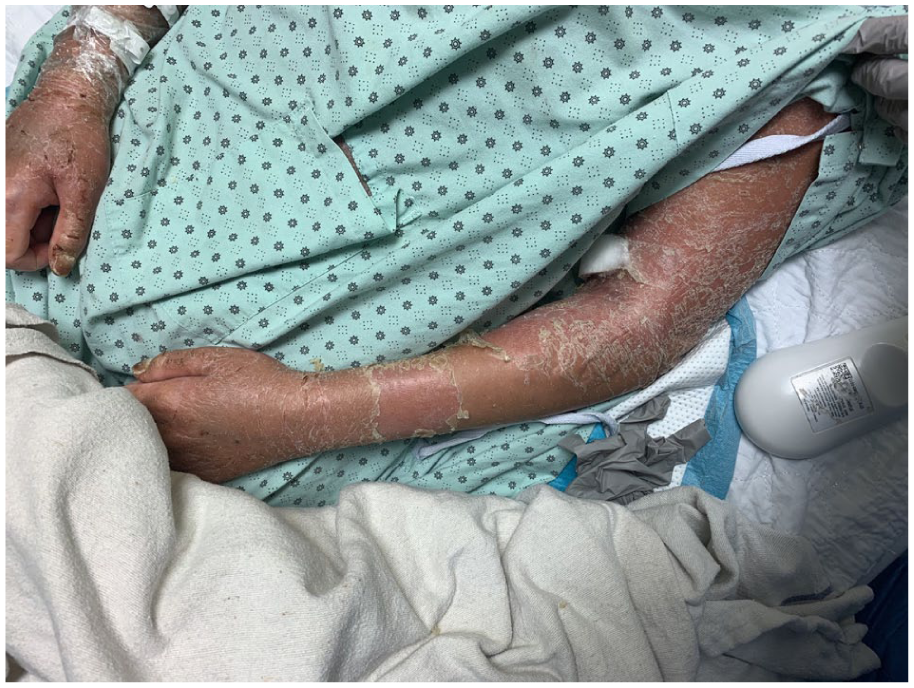
Erythroderma and diffuse desquamation were observed on both arms with visible fissures along the elbow and forearm.
High-power examination reveals a polymorphous inflammatory infiltrate in the superficial dermis, notably rich in eosinophils.
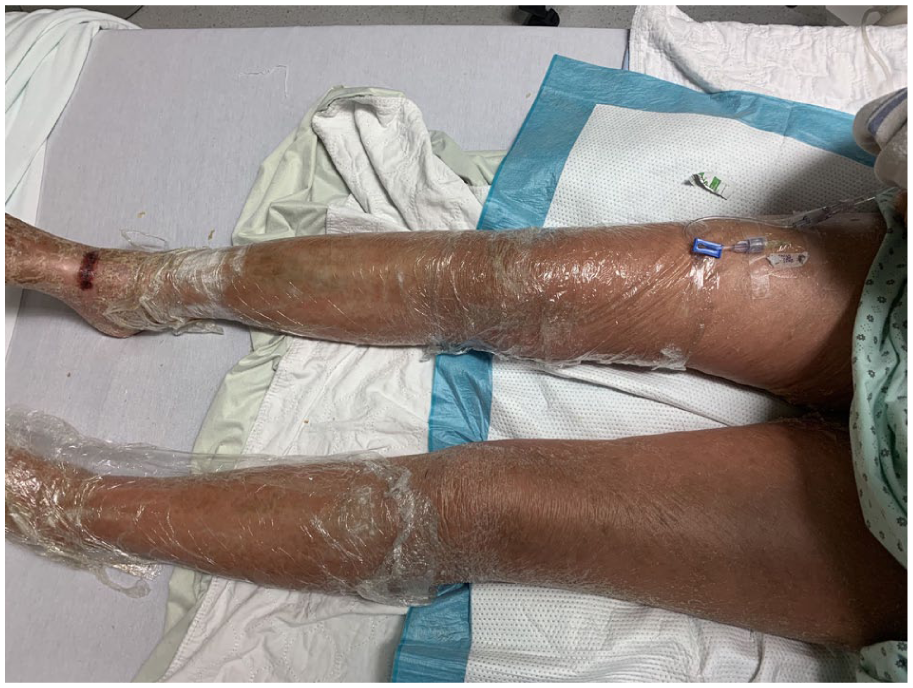
Psoriasiform dermatitis of the lower limbs with widespread erythroderma.
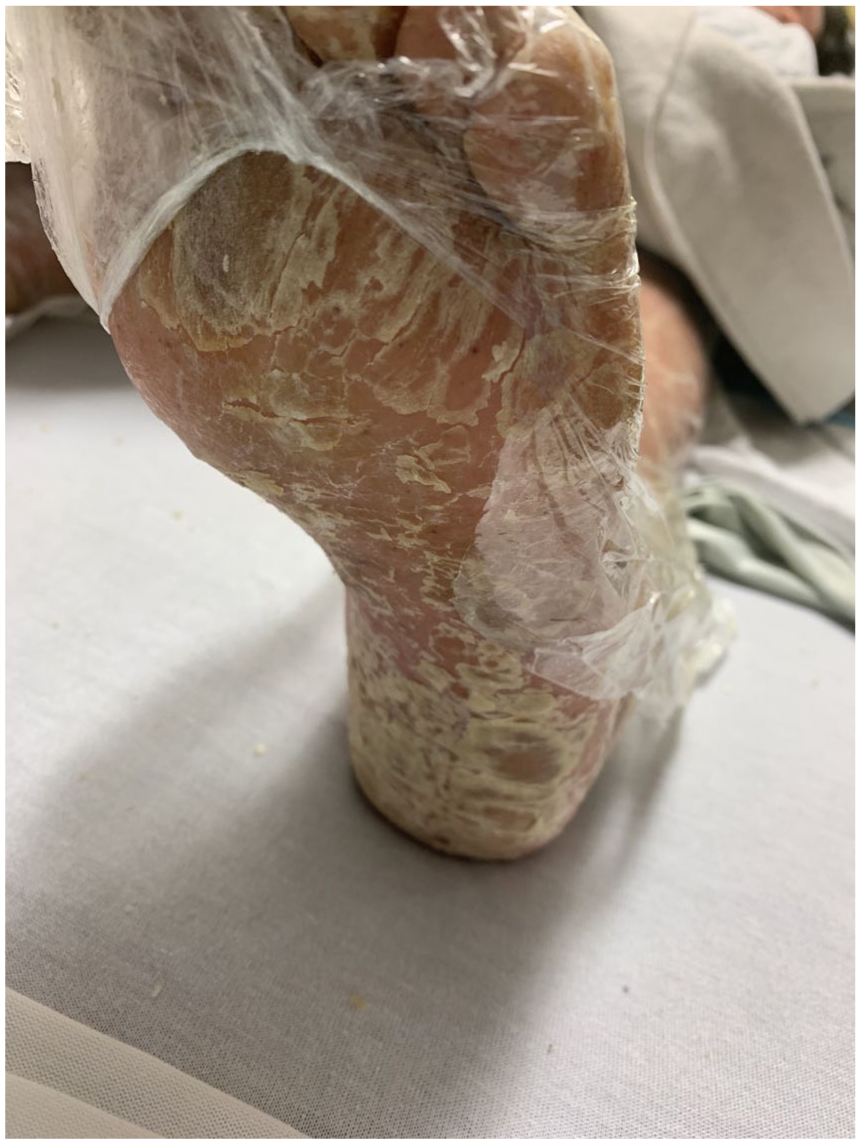
Severe palmoplantar keratoderma with significant desquamation.
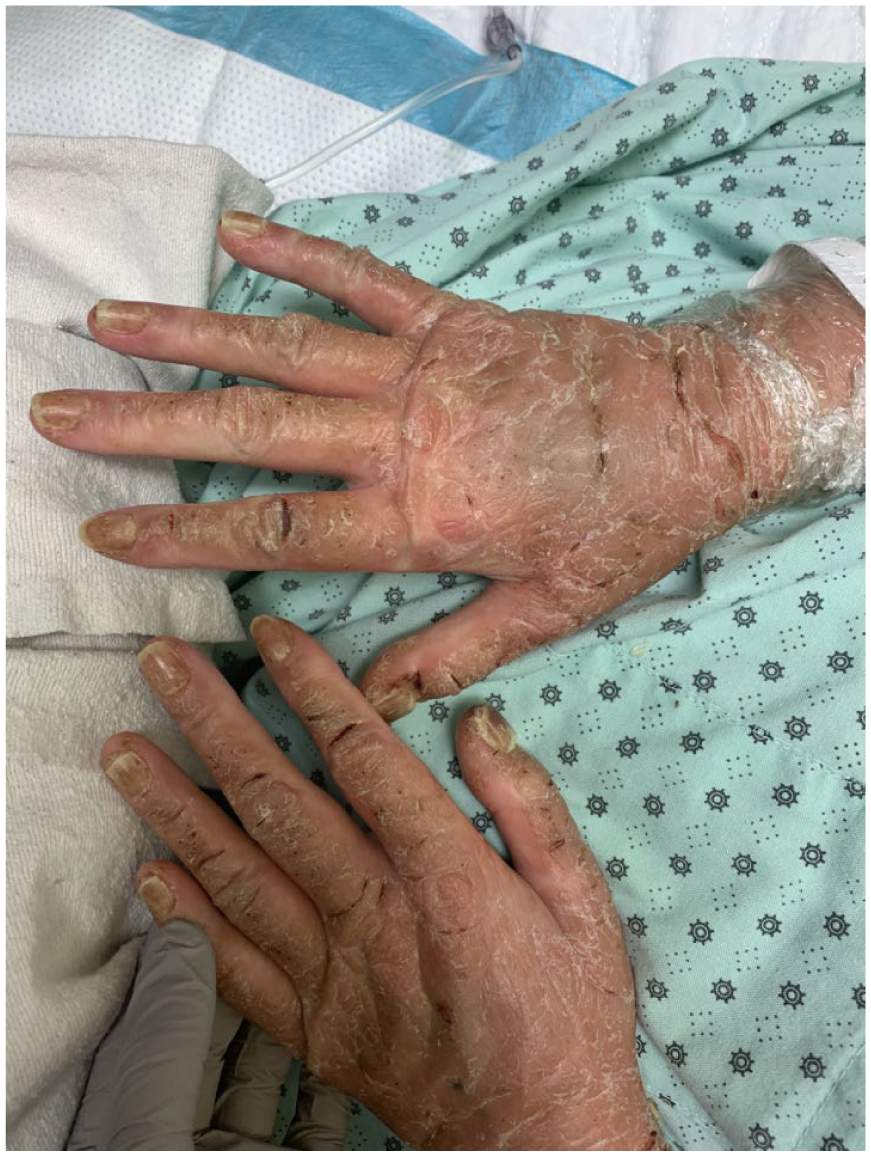
Ichtyosiform desquamation accompanied by distal onycholysis and hyperkeratosis nails.
Upon admission, visceral involvement was absent. Liver function, creatinine levels, and CRP were normal except for hypereosinophilia (2.3 × 109/L). However, as oral prednisone was gradually reduced, she experienced a relapse with a similar presentation as her previous episodes.
Tests for CMV, HHV8, HHV6, and EBV were negative. The RegiSCAR (severe cutaneous adverse reactions) was 5, indicating a probable case of DRESS/DiHS. The acute eruption, eosinophilia, biopsy findings, and newly initiated chemotherapy supported the diagnosis, with bortezomib as the likely culprit.
She responded to IV solumedrol but relapsed after switching to oral prednisone. Therefore, dupilumab was initiated as a long-term corticosteroid-sparing agent. No new medications were introduced to minimize the risk of cross-reactivity. Consequently, there was significant improvement, marked by complete resolution of the erythroderma. There was no recurrence of organ involvement except for a drug-induced hepatitis that developed later due to Levetiracetam (Keppra), which resolved upon switching to Lacosamide (Vimpat). Complete remission was achieved after 14 weeks.
Discussion
DRESS/DiHS may present various forms, including urticated papules, maculopapular/morbilliform erythema, exfoliative erythroderma, or erythema multiforme-like presentations (Table 1).3,8 Common culprits include phenytoin, phenobarbital, carbamazepine, and allopurinol. 9 Our patient exhibited an atypical ichthyosis-like phenotype, marking only the second documented case of bortezomib-induced DRESS/DiHS in multiple myeloma treatment. 7 The first case involved fever, hypereosinophilia, hepatitis, renal dysfunction, alongside diffuse erythematous and hyperpigmented patches on the trunk, upper extremities, and face. Transitioning to an oral steroid taper, the patient experienced worsening rash and further deteriorating liver and renal function, with a RegiSCAR score of 5. 7 He was then discharged on prednisone and triamcinolone ointment wraps and showed improvement as prednisone was tapered over the course of 6 weeks.
Cutaneous morphologies in DRESS/DiHS.

DRESS/DiHS: Drug Reaction with Eosinophilia and Systemic Symptoms/Drug-induced hypersensitivity Symptoms; SJS: Stevens-Johnson Syndrome; TEN: toxic epidermal necrolysis.
Delayed onset of DRESS/DiHS can make the diagnosis challenging. 3 Its symptoms can mimic those of other conditions involving acute rash, lymphadenopathy, fever, and systemic involvement, such as severe cutaneous adverse reactions (SCARs), including Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis, DRESS, and Type IV morbilliform eruptions, but also autoimmune/viral mimics. 10 While systemic involvement is milder in SJS/TEN, DRESS/DiHS presents more extensive cutaneous manifestations. Pathological features are not specific to DRESS/DiHS, but prolonged latency, systemic involvement, hypereosinophilia, and facial edema are key distinguishing features. 11
The patient exhibited erythroderma affecting over 75% of the TBSA, along with pan-corporeal desquamation and facial edema. Laboratory results revealed hypereosinophilia and previous liver injury. Biopsies supported the diagnosis of DRESS/DiHS, and a RegiSCAR score of 5 indicated a probable case.
In our case, diagnostic uncertainty delayed treatment and management. While the patient initially responded to high-dose solumedrol, she relapsed upon switching to oral prednisone. Once dupilumab therapy was introduced, sustained remission was achieved. Dupilumab, a monoclonal antibody against IL-4Rα, inhibits IL-4 and IL-13 signaling pathways. 12 Elevated IL-4/IL-13 producing CD4+ T-cells are associated with active DRESS/DiHS. 13 Dupilumab has also previously been effective in four cases of steroid-refractory DRESS/DiHS, leading to patient recovery.12,14,15
Other biologics include anti-IL-5 agents (mepolizumab and reslizumab) or anti-IL-5 receptor biologics (benralizumab). 16 Another severe case of DRESS/DiHS was treated with a combination of TNF-α antagonist (infliximab) and an IL-6 receptor antagonist (tocilizumab). 17 Our patient’s steroid-refractory DRESS/DiHS prompted biologic therapy.
We report an atypical case of DRESS/DiHS triggered by bortezomib in a patient undergoing treatment for multiple myeloma. The case was characterized by ichthyosis-like erythroderma, not previously described with proteasome inhibitors. Steroid tapering was ineffective, causing dupilumab use, which resulted in remission. Our case highlights the need for greater understanding of DRESS/DiHS phenotypes and the evolving role of biologics in its management.
Footnotes
Consent
The authors obtained consent from the patient for their photographs and medical information to be published in print and online, and with the understanding that this information may be publicly available.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
