Abstract
Multiple eruptive dermatofibroma is a rare variant of dermatofibroma and often occurs in association with systemic disease, particularly involving immune dysregulation. We present what may be the first reported case of multiple eruptive dermatofibroma in association with monoclonal gammopathy of undetermined significance, a premalignant plasma cell dyscrasia. A 66-year-old male with stable IgM monoclonal gammopathy of undetermined significance developed over 50 asymptomatic papulonodular lesions over two years. Histopathology confirmed dermatofibromas, the timeline of lesion development was consistent with multiple eruptive dermatofibroma, and laboratory evaluation revealed elevated IgM and a monoclonal paraprotein band. This case highlights a novel systemic association between multiple eruptive dermatofibroma and monoclonal gammopathy of undetermined significance. Given the frequent co-occurrence of immune dysregulation in patients with multiple eruptive dermatofibroma, evaluation for underlying systemic immunologic conditions in affected individuals is warranted. Recognition of such associations may facilitate earlier diagnosis and surveillance of systemic disease.
Keywords
Introduction
Dermatofibromas (DFs) are common, focal, benign fibrohistiocytic tumors of the dermis, typically arising between the second and fifth decade of life with a female predominance. Classically, a DF presents as solitary, firm papule or nodule <1 cm in size, most often located on the lower extremities. 1
Multiple eruptive DF (MEDF) represents a rare subtype of DF. MEDF is defined as the development of five to eight DFs within a four-month period. MEDF is strongly associated with various underlying diseases, including autoimmune diseases, hematological malignancies, myeloproliferative disorders, infectious diseases, familial cases, and other miscellaneous associations.2,3
Herein, we report a rare case of MEDF occurring in the context of monoclonal gammopathy of undetermined significance (MGUS), a premalignant plasma cell disorder, which to our knowledge has not been previously documented.
Case
A 66-year-old male presented with a two-year history of spreading, nonpainful, nonpruritic lesions that started on the lower extremities and progressed to the trunk and upper extremities (Figure 1(A)–(F)). His condition worsened sub-acutely over two months, after which he subsequently continued to develop one to two additional lesions every few months. He endorsed habitual picking of some of the lesions. He was treated with topical triamcinolone twice daily for two months, topical clindamycin twice daily for one month, topical chlorhexidine washes twice weekly, and a two-week course of oral doxycycline with little to no improvement. His past medical history was notable for aortic dissection complicated by heart failure and IgM MGUS incidentally diagnosed at the onset of the cutaneous eruption, which remained stable under observation. His medications included metoprolol, sacubitril-valsartan, dapagliflozin, furosemide, and spironolactone. On physical examination, over 50 hyperpigmented, hyperkeratotic macules were scattered on the trunk, upper back, forearms, and legs. They demonstrated a nodular component with a central keratotic core. Of these lesions, two, identified on the right upper abdomen, were mildly excoriated papulonodules and a few pink, violaceous papules on the shins demonstrated hemorrhagic crusting. A punch biopsy of a recently developed, clinically representative lesion on the right upper abdomen revealed epidermal hyperplasia with compact hyperkeratosis and focal erosion, overlying a proliferation of uniform spindle cells in the dermis (Figure 2(A)). The lesional cells were arranged in a storiform pattern within a fibrotic stroma, with the characteristic feature of entrapped collagen bundles at the periphery of the lesion (Figure 2(B)). These histological features are diagnostic of DF. A perilesional biopsy for direct immunofluorescence studies did not detect specific immunoreactants for IgA, IgG, IgM, C3, or fibrinogen. Laboratory evaluation identified a serum protein electrophoresis band consistent with paraprotein, and an elevated IgM level of 275 mg/dL (normal IgM: 40–230 mg/dL). Thyroid-stimulating hormone, erythrocyte sediment rate, and C-reactive protein were normal. Autoimmune and infectious workups were unremarkable, including negative antinuclear antibody, double-stranded DNA antibody, human immunodeficiency virus (HIV) fourth generation assay, bacterial culture, varicella-zoster virus PCR, and herpes simplex virus PCR. A diagnosis of MEDF was made, and the patient opted for clinical monitoring.

(A) Multiple brown papules and macules on the lower extremities, (B) upper extremities, (C-E) abdomen, and (F) back. (E) Some papules are secondarily excoriated.
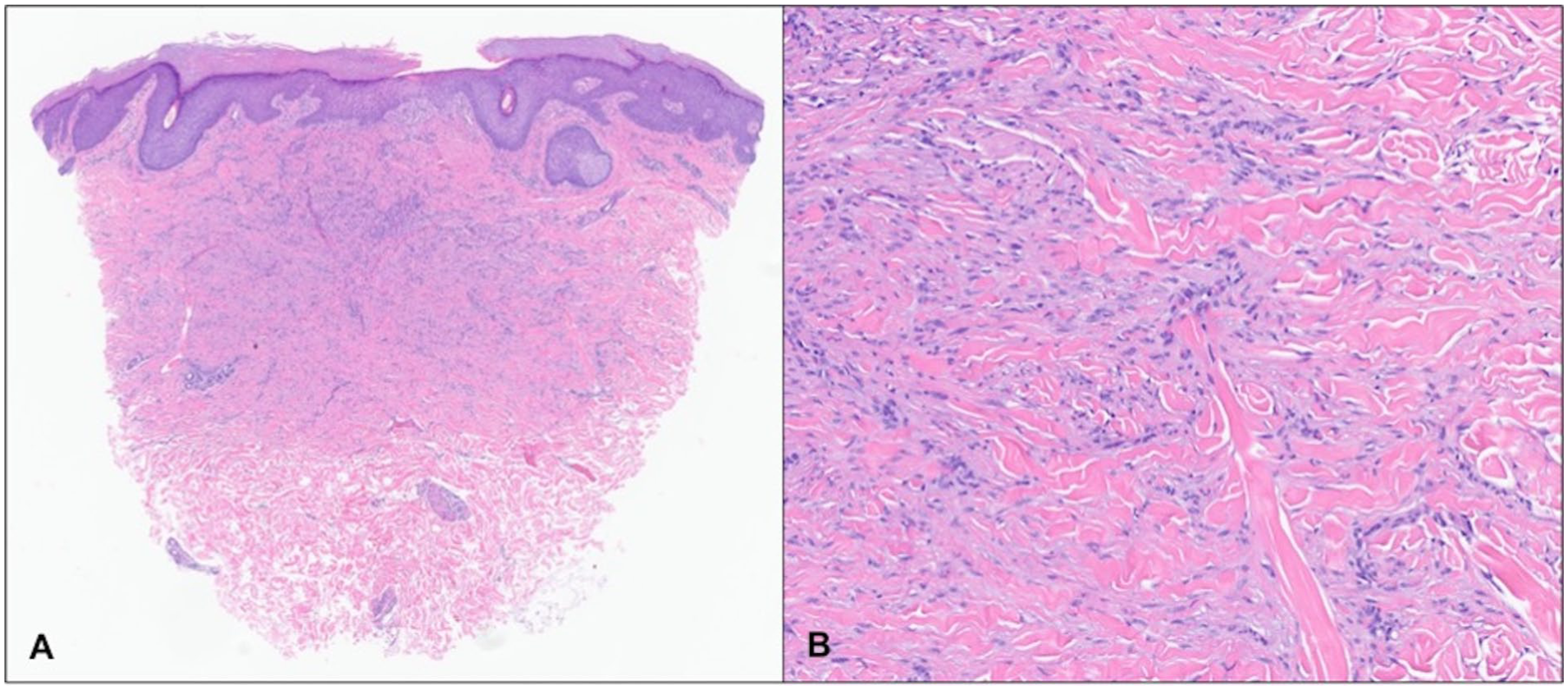
A punch biopsy from right upper abdomen reveals epidermal hyperplasia with compact hyperkeratosis and focal erosion, overlying a dermal spindle cell proliferation (A, 20×). The spindle cells are arranged in a storiform pattern, exhibit uniform cytological features, associated with fibrotic stroma with entrapped collagen bundles at the periphery, consistent with dermatofibroma (B, 100×).
Discussion
This report describes the first documented case of MEDF associated with MGUS. MGUS is a premalignant proliferative disorder of plasma cells seen in 4.2% of individuals over age 50. The clonal expansion of B-cells in MGUS marks severe immune dysfunction, including increased susceptibility to infection, impaired vaccine response, and increased risk of developing multiple myeloma. 4 This immunosuppressive state aligns with the hypothesis that MEDF arises in contexts of immunologic compromise.
MEDF is rare, accounting for only ~0.3% of all DF cases. 3 Diagnosis relies on clinical and histopathologic correlation. Clinically, MEDFs resemble classic DFs – that is, asymptomatic, firm, brown, papules, and nodules that often exhibit a positive dimpling sign. Histologically, MEDF is characterized by dermal nodules composed of spindle-shaped fibroblasts and histiocytes with collagen trapping. 5 Immunohistochemical staining shows positivity for factor-XIIIa and negativity for CD34.
MEDF may be associated with an underlying systemic condition in 50% to two-thirds of patients.2,3 A review of 36 patients with MEDF found that an overwhelming 91% had underlying diseases, most commonly systemic lupus erythematosus (SLE), HIV, or hematologic malignancies. A slight female predilection and a mean age of 43 years was also noted. 6 Another review of 67 patients found that 69% had an underlying associated disease, of which 83% were related to immunodeficient states including SLE, HIV, dermatomyositis, myasthenia gravis, and leukemia. 3 Other uncommon associations have also been reported including pregnancy, Down syndrome, and pityriasis lichenoides et varioliformis acuta.7–9 Similarly, drug-induced MEDF has been linked to immunosuppressive therapies, including corticosteroids, cyclophosphamide, azathioprine, and methotrexate. 10 Notably, no correlation has been established between the severity of the underlying disease and the development of MEDF or its symptoms.2,6
As is the case with solitary DFs, the pathogenesis of MEDF is poorly understood. Classic DFs are thought to involve unregulated fibroblast inflammation in response to trauma. 6 In association with immune-mediated disease, MEDF may represent a more widespread immunoreactive process, where dermal dendritic cells are hypothesized to present an unknown antigen to infiltrating T cells in the dermis, triggering a persistent and proliferative inflammatory response.2,10 In immunosuppressive states, the ability to terminate this cascade may be impaired, leading to the development of MEDF.
Dermatologists should be attentive toward identifying systemic associations in patients presenting with MEDF, as MEDF may be indicative of immune dysregulation or immune-mediated disease which may require further workup. Following workup for underlying conditions, treatment of MEDF is primarily symptomatic and may include topical therapies or surgical excision, although recurrence is common. 5 Future research of MEDF in association with immune-mediated disease and the pathophysiology leading to risk of diffuse development of these lesions can provide insight to streamline clinical workup and possibly diagnose systemic disease earlier in its course.
Footnotes
Consent for publication
Consent for the publication of recognizable patient photographs or other identifiable material was obtained by the authors and included at the time of article submission to the journal stating that all patients gave consent with the understanding that this information may be publicly available.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
