Abstract
Nephrotic syndrome and monoclonal gammopathy of undetermined significance are thought to be associated with venous thromboembolism. However, the association is thought to be weak and is often ignored by clinicians. We present a rare case of sudden-onset bilateral pulmonary emboli with lower extremity deep vein thrombosis in a patient diagnosed with both minimal change disease and immunoglobulin M (IgM) kappa monoclonal gammopathy of undetermined significance. No previous report has been published describing venous thromboembolism in a patient with plasma cell dyscrasia and minimal change disease. This case establishes the importance of considering a diagnostic workup for both disorders in patients with venous thromboembolism. Furthermore, venous thromboembolism risk in patients with both of these diseases is significant. Benefits of prophylactic anticoagulation in these patients are still controversial.
Keywords
Introduction
VTE represents the third leading cause of vascular disease with approximately 10 million cases per year globally. 1 Mortality and morbidity of PE has decreased over the years but continues to have devastating impacts on the population and remains fatal for a subset of the population. 2 Nephrotic syndrome is characterized by proteinuria of ⩾ 3.5 g/24 h, albuminemia < 3.0 g/dL, peripheral edema, hyperlipemia, and increased thrombotic risk. 3 Minimal change disease (MCD) is a cause of idiopathic nephrotic syndrome and is typically seen in 10%–15% of adults presenting with nephrotic syndrome. 4 MGUS is a clinically asymptomatic disorder characterized by < 3 g/dL monoclonal protein concentration in serum, <10% monoclonal plasma cell infiltration of the bone marrow, and absence of end organ injury. 5 Glomerular and tubulointerstitial lesions associated with plasma cell dyscrasias are well known to include amyloid, cast nephropathy, monoclonal immunoglobulin deposition disease, cryoglobulin-associated glomerular disease, and fibrillary immunotactoid glomerulopathy. 6 The association between MGUS and VTE as well as nephrotic syndrome and VTE have been described in multiple large cohort studies.7,8 However, preventive measures are not routinely taken. Cases presenting with both plasma cell dyscrasias and minimal change disease are rarely reported.9,10 To our knowledge, there has not been a single published case report describing VTE in a patient with MGUS and MCD. We present a rare case of sudden-onset bilateral PE with lower extremity DVT in a patient diagnosed with both MCD and MGUS.
Case presentation
A 50-year-old Caucasian male with osteoarthritis and obstructive sleep apnea presented to the emergency room with diffuse, progressive anasarca including lower extremities, upper extremities, and abdomen. History was pertinent for a travel 1 month prior to initial presentation. Patient had a weight of 93 kg and body mass index of 29.4 kg/m2. He was afebrile and hemodynamically stable. Physical exam revealed regular heart rate and rhythm, breath sounds clear to auscultation, abdominal ascites with fluid shift, and lower extremity 2+ pitting edema. Labs were significant for an albumin of 1.9 gm/dL and globulin of 3.5 gm/dL. Urinalysis showed 3 + proteinuria, quantified as 1015 gm/dL. Urine creatinine was 165 mg/dL and urine protein: creatinine ratio was 6.15. Lipid panel showed total cholesterol of 363 mg/dL and low-density lipoprotein cholesterol of 262 mg/dL. Computed tomography (CT) of the abdomen and pelvis showed anasarca. Bilateral lower extremity venous dopplers were negative for DVT and CT angiography of the chest was negative for PE, aortic dissection, or aortic aneurysm. Given hypoalbuminemia, elevated urine protein: creatinine ratio, hyperlipidemia, and generalized edema, he was presumptively diagnosed with acute nephrotic syndrome and thus, 24 h urine protein was not collected. During admission, he was treated with intravenous (IV) furosemide 40 mg twice a day, enalapril 2.5 mg daily, and atorvastatin 80 mg at bedtime. Additional workup included negative antinuclear antibody (ANA), negative cytoplasmic ANca antibody, negative anti-myeloperoxidase antibody, complement C3 of 104 mg/dL, complement C4 of 33.7 mg/dL, and an anti-double strand DNA antibody of 6.0 IU/mL. Serum protein electrophoresis (SPEP) with immunofixation resulted as monoclonal IgM kappa chain gammopathy. Left renal biopsy as well as quantitative immunoglobulins were obtained with results pending. He was deemed to be clinically stable and discharged on oral furosemide, enalapril, and atorvastatin to follow-up closely with nephrology for kidney biopsy results and hematology/oncology for outpatient bone marrow biopsy.
Four days after discharge, he presented back to the hospital with complaints of right lower extremity pain, chest pain, and shortness of breath. He was hemodynamically stable with an oxygen saturation of 97% on room air. Physical exam was remarkable for + 1 pitting edema of the right lower extremity with positive Homan sign. Labs were similar to prior admission. Lower extremity venous dopplers showed an occlusive thrombus in the right posterior tibial vein (Figure 1). CT pulmonary angiography (CTPA) of the chest showing filling defects in the distal main bilateral pulmonary arteries extending into segmental branches in both upper and lower lobes consistent with bilateral pulmonary emboli (Figure 2). He was monitored in the intensive care unit overnight and started on weight-based full-dose low-molecular weight heparin (LMWH). At this time, left kidney biopsy pathology reports resulted. Electron microscopy showed effacement of foot processes of the visceral epithelial cells with effacement in 70% of the foot processes (Figure 3). Glomerular basement membranes were of normal thickness without immune complexes in peripheral capillary walls, mesangial cells, or tubular basement membranes. Light microscopy identified up to nine glomeruli with no segmentally or globally sclerosed glomeruli. The glomeruli were normocellular (Figure 4(a)) with thin peripheral capillary walls and open capillary spaces. There was mild thickening of the walls of arterioles/small size arteries and mild intimal fibrous in larger arteries (Figure 4(b)). There was minimal, focal interstitial fibrosis without evidence of tublitis or vasculitis (Figure 4(c)). Periodic acid-Schiff (PAS) staining was negative. Immunofluorescence stains were negative for IgG, IgM, C3, C1q, albumin, and globulin. Immunofluorescence demonstrated 2 + IgA, 2 + kappa, and 2 + lambda staining of tubular casts without glomerular or vascular staining. These findings were consistent with MCD and nonspecific mild vascular sclerosis associated with minimal interstitial fibrosis. Serum quantitative IgG, IgA, and IgM values were 469, 161, and 527 mg/dL, respectively. Kappa light chain and lambda light chain values were 39.3 and 15.8 mg/dL, respectively, with an elevated kappa/lambda ratio of 2.49. Our patient was started on high-dose steroid therapy with prednisone 60 mg daily. Furosemide was switched to oral bumetanide 1 mg twice daily with metolazone 5 mg daily. Bone marrow biopsy was obtained, showing a normocellular bone marrow with less than 10% of plasma cells consistent with MGUS.

Occlusive right posterior tibial vein with no venous blood flow.
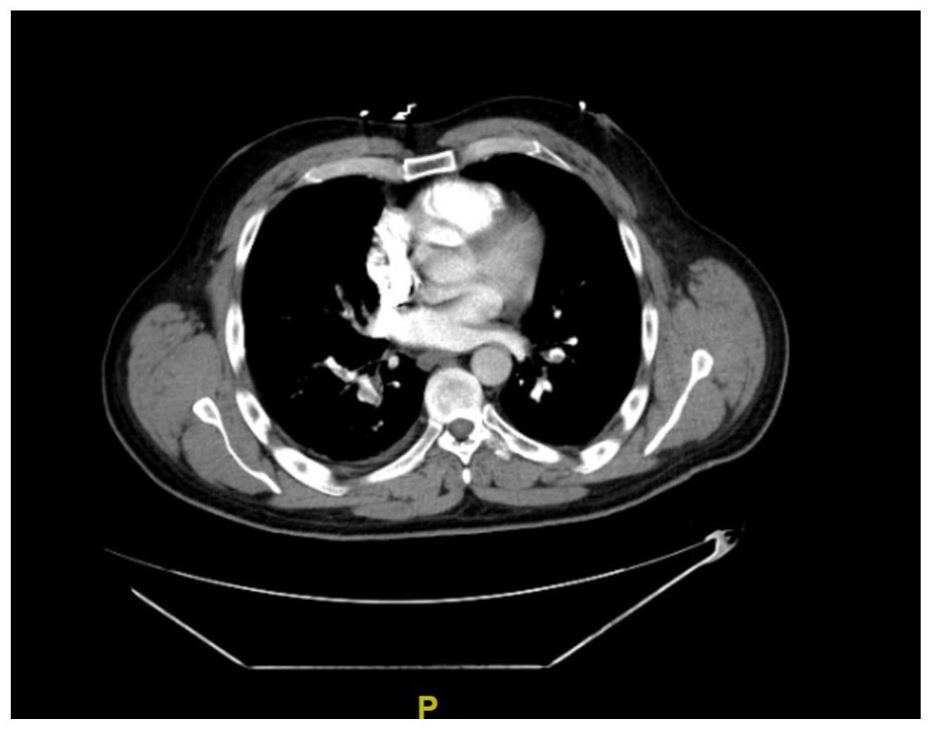
Computed tomography angiography of the chest: bilateral pulmonary emboli.
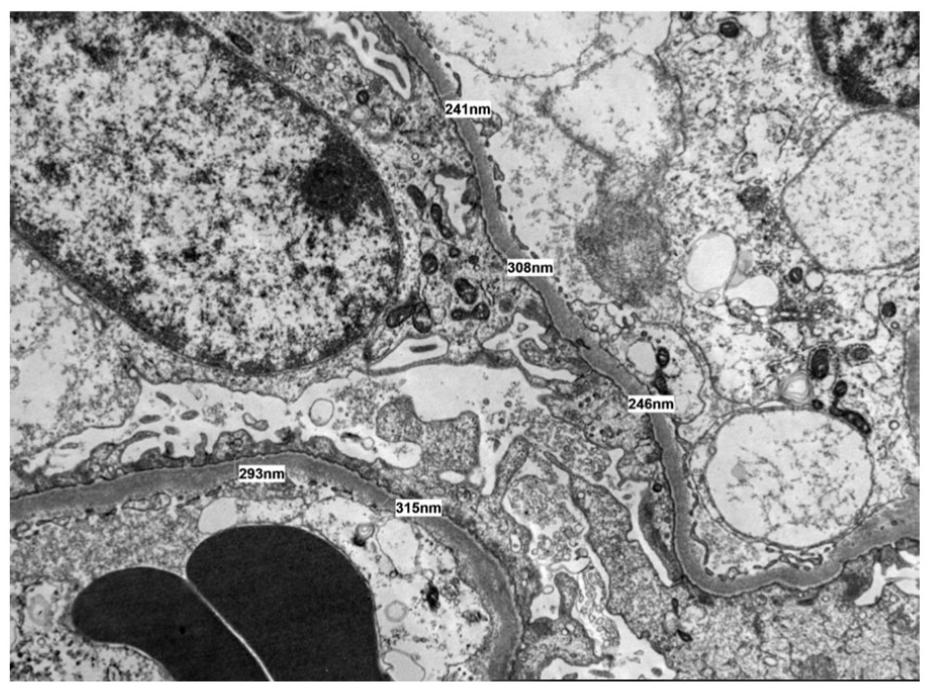
Electron microscopy of left kidney biopsy showing minimal change disease: effacement of foot processes, normal glomerular basement membranes, and no immune complexes.

Light microscopy of left kidney biopsy: (a) normal glomerulus, (b) normal interstitium with mild vascular sclerosis, and (c) mild intimal fibrous thickening.
With anticoagulation and targeted treatment for nephrotic syndrome, our patient improved clinically and was determined to be stable for discharge. He was discharged with apixaban, bumetanide, metolazone, prednisone, and sulfamethoxazole/trimethoprim prophylaxis. Outpatient hypercoagulable workup including antithrombin III, protein C, protein S, lupus anticoagulant-1, Factor V Leiden, prothrombin gene, and MTHFR were all unremarkable. The patient was diagnosed with multiple VTEs in the setting of both MCD and IgM kappa MGUS.
Discussion
Occurrence of VTE occurs with vascular endothelial injury, stasis of blood flow, and a hypercoagulable state. VTE includes both deep vein thrombosis (DVT) and pulmonary emboli (PE). Symptoms of lower extremity DVT include unilateral leg swelling, redness, or pain and symptoms of PE include pleuritic chest pain, shortness of breath, and during a massive PE, patients may even present with hemodynamic instability and syncope. 11 Patients with suspected VTE should undergo a close examination for signs and symptoms as well as assessment of risk factors including hospitalization, cancer, previous VTE, immobilization, and recent surgery. All these factors are then typically used to analyze clinical probability of a DVT or PE through the Wells criteria. 12 When DVT is initially suspected and there is unlikely clinical probability, a d-dimer test can be used as the first-step in diagnostic workup. If the d-dimer returns negative in a low or clinically unlikely patient then DVT can be ruled out. If the d-dimer returns positive, then guidelines recommend further imaging. Compression ultrasonography is the diagnostic imaging test of choice for a DVT and a negative result with a whole-leg ultrasound rules out DVT. 13
The diagnostic approach for PE follows a different algorithm. If PE is clinically unlikely and d-dimer is negative, then no further testing is required. If there is a suspicion for both PE and DVT then lower extremity ultrasound should be obtained first. If there is a high clinical probability for PE or a high d-dimer value then CTPA of the chest is the next indicated exam. If pretest clinical probability is high, physicians can consider chest CTPA results as diagnostic. 14 An alternate diagnostic option for PE is a ventilation-perfusion (V/Q) scan, mostly indicated for patients where CTPA is contraindicated such as renal insufficiency, pregnancy, or severe contrast allergy. No further treatment is indicated if V/Q scan is normal. Treatment with anticoagulation is recommended if V/Q scan shows a high probability of PE. If V/Q scan is non-diagnostic and a PE is unlikely, serial US is sufficient but if V/Q scan is non-diagnostic and PE is likely, CTPA should be considered. 15
The goal of treatment for VTE is to help improve symptoms, prevent recurrent future events, and avoid further extension of the blood clot. Patients who have symptoms of a massive DVT, PE with a risk for cardiorespiratory deterioration or requirement of supplemental oxygen, major comorbidity requiring in-hospital care, or at high-risk for anticoagulant-related bleeding should be considered for inpatient hospitalization. 16 Initial anticoagulation refers to systemic anticoagulation administered immediately following the diagnosis of VTE while a decision regarding long-term anticoagulation is being made. Options typically include LMWH, unfractionated heparin (UFH), fondaparinux, oral factor Xa inhibitors or direct thrombin inhibitors. Long-term anticoagulation is administered beyond the initial days for 3 to 6 months. Options include oral anticoagulants such as factor Xa inhibitors, direct thrombin inhibitors, and warfarin and parenteral subcutaneous agents such as LMWH and fondaparinux. Recent studies have shown higher rates of efficacy and fewer deaths with LMWH in comparison to UFH. 17 Our patient was successfully initially treated with LMWH and then transitioned over to an oral anticoagulant for long-term anticoagulation.
It is known in literature that both nephrotic syndrome and plasma cell dyscrasias are associated with an increased risk of VTE. The association of nephrotic syndrome and VTE has been reported in multiple cohort studies18–20. Medjeral-Thomas et. al found that VTE occurs in 7% to 40% of patients diagnosed with nephrotic syndrome. 18 Kayali et al.’s study evaluated 925,000 patients with nephrotic syndrome and 898,253,000 patients without nephrotic syndrome. They found a relative risk of 1.39 for PE and 1.72 for DVT comparing patients with nephrotic syndrome to those without. 19 Out of 512 patients evaluated by Zhang et al., 153 patients had PE. 20 The cause of hypercoagulability in nephrotic syndrome is not completely well understood but the hypoalbuminemia seen in nephrotic syndrome contributes to increased hepatic synthesis of fibrinogen, favoring thrombus formation. In addition, damage to the glomerular membrane results in increased filtration of antithrombin III, plasminogen, protein C, and protein S, increasing coagulability. 21
The association of MGUS and VTE has also been reported in cohort studies.7,8 On the basis of 2374 MGUS cases, Kristinsson et al. found MGUS to be associated with a threefold increased risk of DVT. 7 Gregersen et al. included more than 5000 MGUS patients and found an excessive risk of venous and arterial thrombosis in patients with MGUS, approximately 37% higher incidence compared with the general population. 8 Furthermore, a case of extensive lower extremity DVT has been published as the initial presentation of MGUS. 22 The etiology of this association is unclear, but studies have identified an increase in von Willebrand factor (vWF) activity, FVIII levels, and whole-blood viscosity in patients with MGUS.23,24
Upon initial presentation, our patient met the diagnostic criteria for nephrotic syndrome as well as monoclonal gammopathy. Workup for VTE at that time was negative. Shortly after discharge, kidney biopsy results confirmed MCD. Four days after, he was readmitted with significant VTE. At this time, a bone marrow biopsy was obtained that was consistent with MGUS. The relation between MGUS and nephrotic syndrome is notably well-established. The pathophysiology of renal involvement in MGUS involves two mechanisms: immunoglobulin deposition in tubular and glomerular cells after the protein has been excreted in the urine and immunoglobin acting as a local antibody.25,26 Interestingly, it was determined that our patient’s nephrotic syndrome likely had no correlation with his MGUS as his kidney biopsy showed MCD without any focal immunoglobulin or immune complex deposition, ruling out amyloidosis. Furthermore, since our patient had IgM MGUS, it is important to consider IgM is much larger than IgA and IgG particles as IgM is a pentamer and can cause viscosity issues in patients.
As mentioned earlier, both MCD and MGUS have been associated with an increased risk of VTE. However, the most effective method of VTE prophylaxis is unclear. 27 According to the 2012 Kidney Disease: Improving Global Outcomes (KDIGO) guidelines, prophylactic anticoagulation should be considered in nephrotic syndrome patients if serum albumin < 2.0-2/5 g/dL, proteinuria > 10 g/d, body mass index > 35 kg/m2, genetic disposition for thromboembolism, heart failure New York Heart Association class III or IV, recent orthopedic or abdominal surgery, or prolonged immobilization. 28 However, it must be acknowledged that existing data are limited and direct-acting oral factor Xa inhibitors (DOAC) have not been systematically studied for VTE prophylaxis in nephrotic patients. Medjeral-Thomas et al. completed a retrospective analysis of 143 high-risk patients initiated on prophylactic anticoagulation, aspirin 75 mg daily if albumin levels 2–3 g/dl and low molecular weight heparin or warfarin if albumin < 2.0 g/dl. Over a 5-year period, no symptomatic VTE occurred in patients established on prophylaxis for at least 1 week. This study did limit confounding factors such as malignancy and thus concluded that this particular regimen in nephrotic patients with underlying causes requires further investigation. 28 According to the International Myeloma Working Group (IMWG) and National Comprehensive Cancer Network (NCCN), in patients with myeloma who are treated with a combination chemotherapy regimen, the routine use of VTE prophylaxis is recommended.29,30 No clinical studies have evaluated the benefit of prophylactic anticoagulation in patients with MGUS or on patients not on treatment for multiple myeloma.
Even though our patient had significant thromboembolism, he responded well to anticoagulation. The aim of this case report was to increase awareness for multiple issues. First, we add to known literature of how MGUS and MCD play a significant role in the increased risk of VTEs. We also establish how workup for both nephrotic syndrome and monoclonal gammopathy should always be considered in the differential diagnosis when VTEs are present. A simple urinalysis can help guide the direction in workup for either disorder, and appropriate diagnosis can significantly change management. Most importantly, we conclude that further trials need to investigate the benefits of VTE prophylaxis in nephrotic syndrome patients, monoclonal gammopathy patients, and more so in patients with a combination of both.
Conclusion
We present a rare case of a patient with significant VTE in a patient with both MGUS and MCD. We demonstrate how nephrotic syndrome and plasma cell dyscrasias should be considered in the differential for patients who present with VTE. In addition, this case increases awareness of the significant risk and danger of VTE in patients with plasma cell dyscrasias, nephrotic syndrome, and a combination of both of these diagnoses. Benefits of prophylactic anticoagulation in these patients is still controversial.
Footnotes
Acknowledgements
This research was supported (in whole or part) by HCA Healthcare and/or an HCA Healthcare affiliated entity. The views expressed in this presentation represent those of the author and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.
Author contribution
Thanuja Neerukonda: Conceptualization, Writing—original draft. Alexandra Witt: Supervision, Writing—review & editing. Arsen Tan: Visualization. Bilal Farooqi: Visualization, Writing—review & editing. Yasna Chaudhary: Resources, Validation. Christina Kovacs: Writing—review & editing. All authors have read and approved the manuscript.
Data availability statement
No data were used to support this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
