Abstract
Our case involves the clinical course of a healthy male infant who developed a calcified cephalohematoma despite no history of birth trauma or the use of assistive delivery instruments. Another unique feature of the patient’s presentation was the young age at which the cephalohematoma was determined to be calcified, noted at 34 days of life. The management of the patient is discussed along with a review of the epidemiology, treatment, and prognosis of cephalohematomas. It is widely understood that most cephalohematomas reabsorb within ~30 days, but it is imperative that the general pediatrician is aware of potential complications that may arise and subsequent next steps in management. The long-term neurologic effects from an untreated calcified cephalohematoma have not been well-studied; however, prompt evaluation is recommended to prevent permanent cosmetic changes and psychosocial impact due to cranial vault distortion.
Introduction
Cephalohematomas, or tumores cranii sanguineus, are relatively rare complications in the neonatal population and occur in ~0.2%–3% of live births.1–4 They are subperiosteal collections of blood under the outer surface of the skull confined within the borders of the cranial sutures (Figure 1). Risk factors for the development of a cephalohematoma most often include trauma associated with vaginal deliveries. The use of vacuum or forceps assistance can sometimes lift the scalp and pericranium off the skull bone causing shearing of the delicate blood vessels and subsequent bleeding into the surrounding space.1,5

Layers of the infant scalp with locations of common hematomas.
Other risk factors include difficult or prolonged labor, insertion of intrauterine fetal monitor wires, high birth weight (>4 kg), maternal primiparity, or family history of bleeding disorders such as Factor VIII deficiency. 5 Cephalohematomas usually occur in the parietal (88% 6 ) or occipital regions as these tend to be the most prominent presenting points of the infant’s head during vertex delivery. They appear at or shortly after birth and enlarge within the first 24–72 h of life before spontaneously reabsorbing over weeks to months. 5 They can be associated with underlying skull fractures (up to 25% 2 ), which tend to be linear. Other complications include epidural hematomas, anemia, and neonatal jaundice. 5
Several other conditions can present as soft tissue scalp swellings similar in appearance to cephalohematomas, including caput succedaneum, subgaleal hematomas, encephalocele, and tumors.5–7 Caput succedaneum refers to subcutaneous edema of the occipital region caused by the birthing process. Its rapid resolution in hours to days and lack of limitation by cranial suture lines distinguish caput succedaneum from a cephalohematoma. Subgaleal hematomas are potentially life-threatening hemorrhages marked by the accumulation of blood within the subgaleal space (Figure 1) between the epicranial aponeurosis and the skull. They are typically associated with the use of vacuum extraction devices. 8 Although rare, they can become quite large and lead to hypovolemic shock given that the subgaleal space extends over the entire scalp, providing no anatomical barriers to bleeding. 9 Neural tube defects are characterized by midline protrusions of intracranial contents through openings in the skull. Encephaloceles, particularly those in the occipital region, may mimic cephalohematomas; however, radiographic imaging reveals osseous defects and affected neural tissue contained within the area of swelling (Figure 1).
Case presentation
A term male infant was born at 39 weeks, 1-day gestational age via spontaneous vaginal delivery to a 32-year-old Gravida 1 Para 1 mother. Labor and delivery were uncomplicated; the infant head was delivered in the occiput anterior position without mechanical assistance, and Apgar scores at 1 and 5 min of life were 9 and 9. Birth weight was 3705 g, which placed the infant at the 75th percentile; length was 50.8 cm (57th percentile), and head circumference was 34.5 cm (47th percentile).
The patient was admitted to the Level 1 newborn nursery shortly after birth. Physical exam at 48 h of life was significant for an area of swelling on the posterior scalp not previously noted. Neurological exam was appropriate for gestational age. Newborn nursery course was complicated by hyperbilirubinemia in the setting of ABO incompatibility (maternal blood type O+, infant blood type B+, negative direct antiglobulin test); direct bilirubin and hematocrit levels were within normal limits. Phototherapy was initiated at 36 h of life and discontinued at 50 h of life. Infant was discharged home at 72 h of life with a phototherapy blanket given high-intermediate risk of hyperbilirubinemia, cephalohematoma noted on exam, and parental plan for exclusive breastfeeding.
The infant was seen for newborn nursery discharge follow-up in an outpatient Internal Medicine-Pediatrics clinic on day of life 4. At that time, physical exam was notable for a persistent, fluctuant cephalohematoma localized to the right superior occiput, which was stable in size from birth per maternal report. The neurologic exam was within normal limits. Laboratory studies were significant for persistent hyperbilirubinemia, which prompted inpatient hospitalization overnight for closer monitoring. At the 2-week well-child check, the patient’s neurologic exam and degree of firmness of the cephalohematoma remained unchanged.
On day of life 33, the patient’s mother contacted the clinic with concerns that the cephalohematoma, though stable in size, had become “hard to the touch.” The patient’s mother included a photo of the cephalohematoma in this communication (Figure 2). To assess concern for calcification, a head computed tomography (CT) scan was ordered on day of life 34 and confirmed calcification; a consultation to Pediatric Neurosurgery was promptly made to determine if the calcified cephalohematoma could simply be monitored or if the lesion was large enough (>7 cm) to cause a high risk of cranial disfigurement.

(a) Patient-supplied photograph. Image depicts a known cephalohematoma overlying the right parietal vertex. (b) Head CT axial view demonstrates peripheral calcification with underlying calvarium osteitic thinning indicated by the yellow arrow. (c) Head CT VRT. Demonstrates calcification of the cephalohematoma over the right parietal calvarium. (d) Head MRI 2 months postoperatively. Demonstrates approximation between the bone layers without areas of resorption as demonstrated by the yellow arrows.
The CT demonstrated peripheral calcification of a right parietal vertex scalp fluid collection consistent with the clinical finding of a non-resorbed cephalohematoma (Figure 3). The calcified wall measured up to 1 mm in greatest thickness, and the underlying calvarium demonstrated osteotic thinning. CT volume rendering technique supplied a three-dimensional reconstruction of the skull to assist with surgical planning, and this rendered image further supported the concern for calcification (Figure 2).
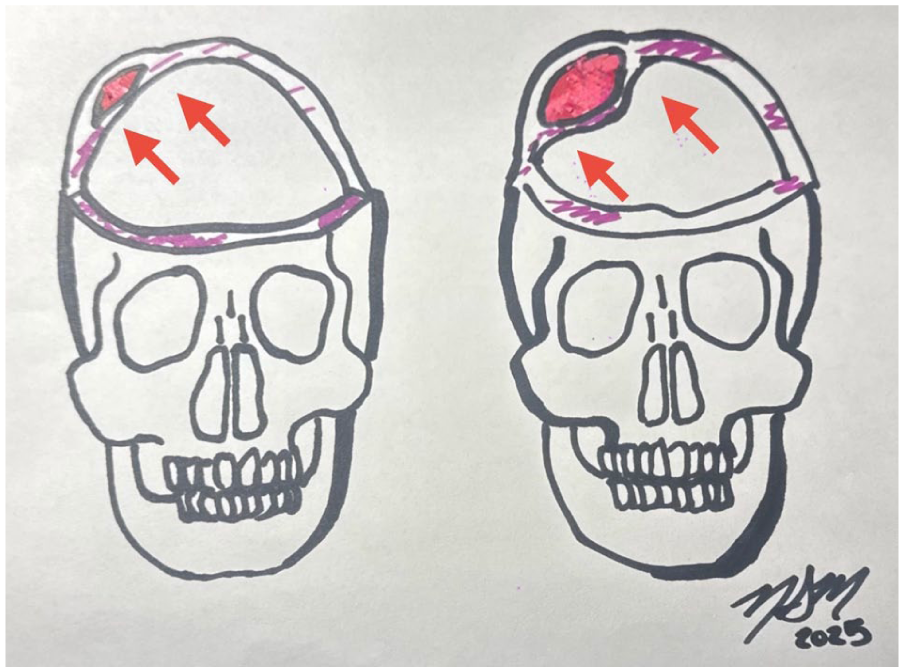
Leftward image: Type 1 calcified cephalohematoma has a non-depressed inner lamella with no encroachment into the cranial vault space (arrows). Rightward image: Type 2 has an inner lamella, that is, depressed into the cranial vault space (arrows).
The estimated volume of the patient’s cephalohematoma was 18 cm3, with a diameter of 7 cm, which was approximated via CT (Figure 4). Per the literature, the most cited criterion for what is considered a large cephalohematoma is the following: a diameter >5 cm or maximal projection >9 mm.10,11
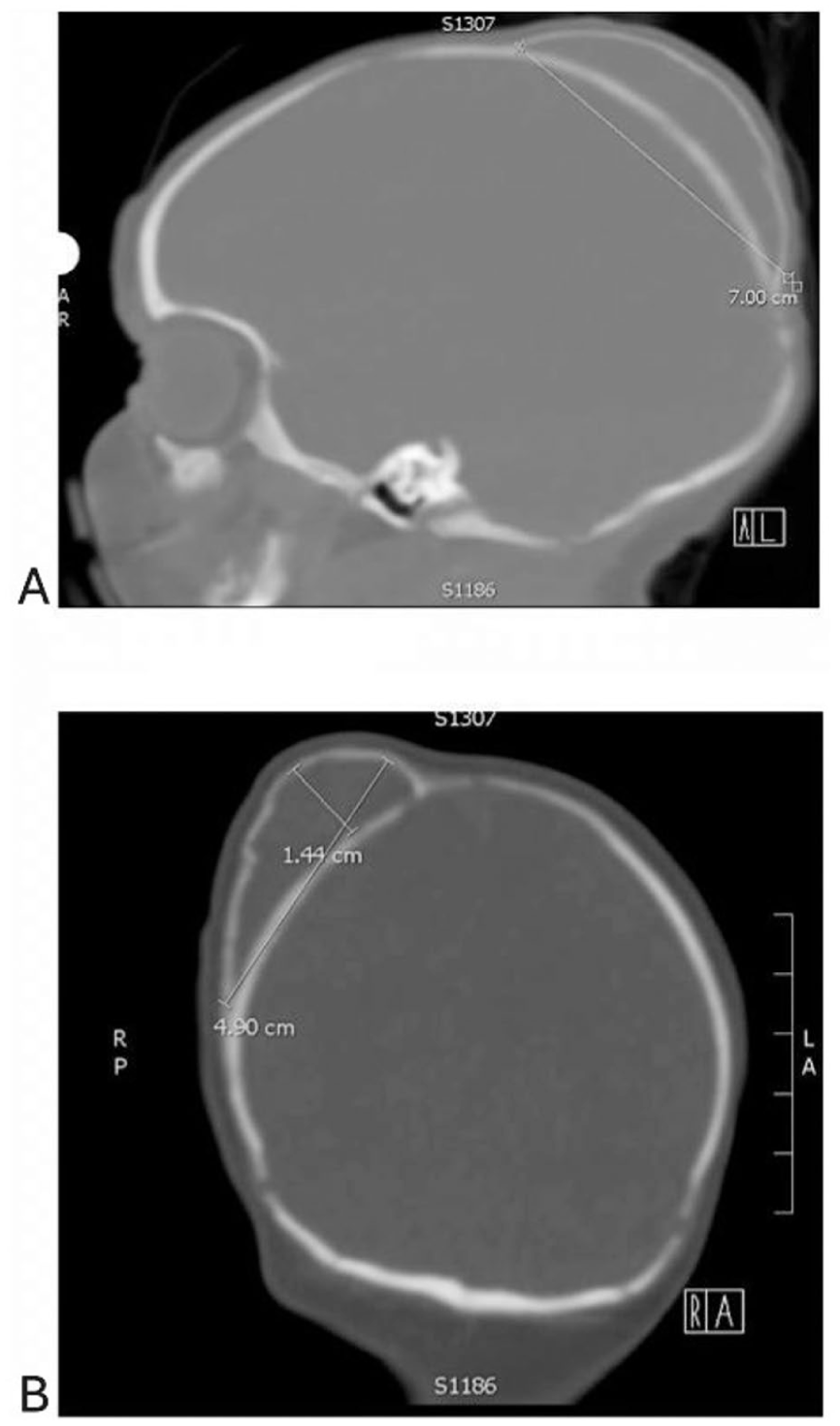
Seven-centimeter diameter of the calcified cephalohematoma in the sagittal plane (a). Remaining dimensions of the cephalohematoma in the transverse plane (b).
Of note, the patient’s neurologic physical exam remained unchanged during surgical evaluation. Due to the extent of the calvarial thinning and size of the calcified cephalohematoma, the patient was taken for surgical repair.
Surgical technique
Under general anesthesia, the patient was positioned prone, and a C-shaped incision was made over the right parietal deformity after infiltration with local anesthetic. The skin flap was dissected to the periosteum, which was elevated to expose the calcified cephalohematoma. The surface of the lesion was exposed, and a split-thickness craniotomy was fashioned using a B-1000 footplate drill. Thick, motor oil consistency fluid was suctioned from within the lesion, and the calcified dome was opened in a clamshell manner. The inner surface of the hematoma was curetted, and all loose or abnormal calcified material was removed. Areas of native skull erosion with exposed dura were carefully preserved. The outer cortical bone flap was then shaped and drilled down until it was flush with the surrounding calvarium. This reconstructed flap was replaced to restore continuity of the calvarium, ensuring that future growth would again be driven by the expanding brain. The wound was copiously irrigated with bacitracin-infused saline. Closure was performed in layers with 4-0 interrupted sutures for the galea and a 4-0 running suture for the skin. The patient was extubated uneventfully and transferred to recovery in excellent condition.
The patient had an uneventful postoperative course and was discharged home postoperative day 1. At a 2-month postoperative clinical follow-up, magnetic resonance imaging (MRI) revealed good approximation between the two bone layers, and physical exam of skull did not demonstrate findings concerning for resorption.
Discussion
Although an uncommon phenomenon, cephalohematomas can become calcified and distort the calvaria, posing three main risk categories. Calcified hematomas in the immediate newborn phase (when brain and skull growth are rapid) may cause pose restriction of brain growth from inward indentation of the calvarium. 5 A second circumstance arises when the cephalohematoma is so large that calcification develops significantly distant from the inner table. In this setting, the hematoma trapped between the two layers may liquefy under pressure, eroding the inner calvarium and risking direct compression of the dura by hematoma contents, mimicking an epidural hematoma. The third risk category involves cosmetic outcomes, including the development of contralateral severe positional plagiocephaly, as the large protrusion causes the infant’s head to rest preferentially on the opposite side during sleep. In our patient, both the second and third circumstances were present. Less common complications of cephalohematomas include infection (osteomyelitis, sepsis, or meningitis) and calcification. 5
Calcification occurs when the blood contained within the cephalohematoma fails to reabsorb within the first few weeks of life. It instead clots and begins to ossify under the lifted pericranium. This process results in a hardened, bony swelling at the area of the cephalohematoma, which can persist for months to years. If large enough, a cephalohematoma can cause significant calvarial deformity.12,13 If calcification is partial, the cephalohematoma may be managed non-surgically.1,12 However, two types of larger calcified cephalohematomas can cause significant distortion of the calvarium requiring close monitoring. Type 1 describes a smooth and non-depressed inner lamellar surface relative to the surrounding cranial vault; type 2 describes a depressed inner lamellar surface. 5 When concerns for a calcified cephalohematoma arise, CT is the imaging modality of choice to confirm the diagnosis. Findings typically include a “double skull sign,” 3 or “a uniformly homogeneous, hypodense, non-enhancing core encased by bone.”5,14
MRI may be obtained if there are concerns for seizure-like activity or other symptoms suggestive of underlying parenchymal involvement 3 ; however, when calcification is suspected and exam is not concerning for intracranial/parenchymal involvement, CT provides the most superior delineation of calvarial thinning. 15
The larger the volume of a cephalohematoma is, the higher the probability of ossification, as demonstrated in retrospective reviews.16,17 When considering the diagnosis, neurosurgery should be consulted without delay to expedite initial assessment and ensure appropriate follow-up and intervention.
Subperiosteal (cephalohematoma) collections in neonates are overwhelmingly managed conservatively, as most are small and result in minimal deformity. Surgical treatment is rarely required; however, in uncommon cases, the hematoma can exert abnormal pressure on the calvarium, leading to aberrant bone remodeling. Normally, the calvarium remodels in response to brain expansion, with the periosteum and dura driving osteogenesis. When a large hematoma is trapped beneath the periosteum, the calvarium may thin and resorb as if under brain pressure, while the periosteum misinterprets the hematoma surface as bone, resulting in calcification and poorly structured outer layers. This process can ultimately cause bone erosion, pathologic ossification, and even exposure of the dura. In our case, calcification was detected as early as 34 days of life, coinciding with severe findings such as calvarial erosion and dural exposure. The temporal association suggests that unusually early ossification may serve as a marker of a more aggressive clinical course, in which accelerated remodeling predisposes to structural compromise. Recognition of this pattern should prompt heightened vigilance and consideration of earlier neurosurgical involvement. Surgical evaluation by a pediatric neurosurgeon, along with appropriate imaging such as ultra-low dose CT or fast-brain MRI, helps identify the rare patients who require intervention.
In patients whose pathology is deemed severe enough to warrant neurosurgical repair, several cranioplasty techniques are described in the literature, including a reversed inner lamella to outer lamella technique and a cap radial craniectomy technique. 5 Our patient required an approach equivalent to a cap radial craniectomy but did not require as much of the traditional cutting features of the radial cap technique. This method increased the surface area to be approximated on the inner portion of the calcified hematoma and outer portion of the normal skull. Because the inner layer of native skull had begun to erode, the use of the calcified portion of the hematoma allowed skull coverage as well as incorporation with the pre-existing calvarium. The patient’s head was wrapped tightly enough to promote fusion between the two layers of bone. Given the few cases of calcified cephalohematomas requiring neurosurgical intervention, it is important that neurosurgical techniques and adaptations to traditional methods be described and shared such that the medical community can benefit from novel approaches to an uncommon phenomenon.
Conclusion
At 3 months of age, our patient was seen in the Internal Medicine–Pediatrics clinic and was found to be developmentally appropriate and thriving. His head was normocephalic with a well-healed surgical scar on the scalp. No evidence of complications from the calcified cephalohematoma resection were identified such as the need for perioperative blood transfusion or superficial wound infection. Figure 5 shows the chronological healing of the calvarium through 1 year of age, with no complications or significant cosmetic disfigurement observed.

Photos of the patient’s healing from the time of diagnosis (a) to 1 year of age (f).
The majority of cephalohematomas resolve spontaneously at 1 month of life. 18 Primary care clinicians may be less comfortable counseling parents and guardians on expected outcomes once calcification of the cephalohematoma occurs. If the general practitioner identifies a calcified cephalohematoma of any size, it is our recommendation that they be referred to neurosurgery for evaluation. Based on the limited number of reported cases, the prognosis is favorable for both surgically repaired calcified cephalohematomas and small lesions managed with medical observation. Due to the concern for poor cosmetic outcome, many large calcified cephalohematomas have undergone repair. The data are lacking in large comparative studies on the neurodevelopmental outcomes of early versus delayed surgical intervention. There is no robust evidence of impaired cranial growth or delayed neurodevelopment if the calcified cephalohematoma is left untreated; however, in cases where there is significant calvarial erosion and exposure of the dura, there has reportedly been the need for more complex cranioplasty to restore the normal cranial contour. 18 It is imperative that providers continue to report incidences of calcified cephalohematomas so that the understanding about the cosmetic prognosis and functional outcomes of patients may be strengthened.
Footnotes
Ethical considerations
This case report was conducted in accordance with the ethical standards of the institution and with the 1964 Helsinki declaration and its later amendments. Our institution does not require ethical approval for reporting individual cases or case series.
Consent for publication
Written and verbal informed consent was obtained from the patient’s parents for publication of this case report and any accompanying images.
Author contributions
Sophia Urban: contributed to conception and design; contributed to acquisition, analysis, or interpretation of date; drafted the manuscript; agrees to be accountable for all aspects of work ensuring integrity and accuracy. Jackson Pearce: contributed to conception and design; contributed to acquisition, analysis, or interpretation of date; drafted the manuscript; agrees to be accountable for all aspects of work ensuring integrity and accuracy. Nathalie Malcolm: contributed to conception and design; contributed to acquisition, analysis, or interpretation of date; critically revised the manuscript for important intellectual content; give final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy. Ramin Eskandari: contributed to conception and design; contributed to acquisition, analysis, or interpretation of date; critically revised the manuscript for important intellectual content; give final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Medical University of South Carolina.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
