Abstract
Necrotizing myometritis (NM) is a rare but life-threatening postpartum infection. Although most reported cases are attributed to Group A Streptococcus (GAS), NM can also arise as a polymicrobial process in the setting of uterine ischemia, particularly after uterine artery embolization (UAE), and this phenotype often follows a slower, more insidious course that may require different diagnostic and therapeutic strategies. We describe NM due to Enterococcus faecium, Klebsiella pneumoniae, and Eggerthella lenta after retained placenta and UAE. In contrast to the fulminant trajectory typical of GAS-associated NM, this case evolved over 48 days with intermittent symptom remission and ultimately required hysterectomy despite multiple courses of antibiotics and repeated curettage. This case highlights the need to broaden empiric antibiotics to include coverage for anaerobes and enterococci, which are frequently resistant to standard postpartum regimens, and indicates that sonographic findings such as myometrial heterogeneity, hypoperfusion, and tissue discontinuity may provide earlier diagnostic clues than clinical signs alone and should prompt early MRI evaluation. Early recognition of these atypical features is critical to avoid diagnostic delay and improve maternal outcomes.
Keywords
Introduction
Necrotizing myometritis (NM), characterized by extensive inflammation and necrosis of the uterine muscle, represents a rare yet potentially catastrophic complication of the puerperium.1,2 Despite its low incidence, NM warrants heightened clinical vigilance because it is associated with severe puerperal sepsis or toxic shock syndrome (TSS), often remains refractory to standard antimicrobial therapy and uterine evacuation, and therefore carries a significant mortality risk. 3 Historically, Group A Streptococcus (GAS) has been recognized as the principal causative pathogen, typically associated with a fulminant course marked by rapid progression to multiorgan failure and necessitating early clinical suspicion.2,4 Foundational studies, such as genome-wide screens, have begun to elucidate specific genes involved in GAS colonization and invasion of the female reproductive tract. 5 However, the broader molecular and immunological host-pathogen interactions in this context remain poorly understood, underscoring a critical gap in the current literature.
The etiological spectrum of NM extends beyond GAS. Emerging evidence indicates that polymicrobial infections, including anaerobic bacteria such as Finegoldia magna or Fusobacterium necrophorum, can precipitate NM in complex clinical contexts.6,7 Established risk factors include direct uterine trauma and retained products of conception (RPOC), which can serve as a nidus for infection.8,9 Moreover, while uterine artery embolization (UAE) is a crucial, often life-saving intervention for refractory postpartum hemorrhage (PPH), it carries an uncommon but inherent risk of uterine ischemia with subsequent necrosis. 9 This ischemic risk may be influenced by the embolic material used, and particulate agents, such as the gelatin sponge applied in the present case, can produce more extensive or distal devascularization than some alternative techniques, potentially fostering an environment favorable to anaerobic or opportunistic pathogens. 6 Underlying conditions, including subclinical uterine inflammation detectable even in asymptomatic parturients, 10 the heightened systemic proinflammatory state associated with PPH, 11 and periods of widespread viral circulation, during which transient immune dysregulation may occur,12,13 may further increase susceptibility to severe postpartum infection. It is also important to distinguish NM from noninfectious inflammatory entities such as postpartum acute myometritis, which has been linked to PPH of unknown etiology.9,14 These considerations underscore the need for heightened awareness of NM’s variable presentations and causes, particularly in complex postpartum scenarios.
Given its severity, potential for harm, and clinical complexity, we consider NM to pose substantial challenges in routine practice. Accordingly, we provide a detailed report of a case that departs from the classic GAS-associated paradigm to alert clinicians to the risks of polymicrobial NM, to delineate its distinguishing features and diagnostic implications, and to offer practical guidance for recognizing and managing similar presentations in the future.
Case presentation
Initial presentation and delivery
A 29-year-old female (G2P0A1L0), with a history of one prior elective abortion, presented at 40+3 weeks’ gestation (LMP: 2023-10-30, EDD: 2024-08-05) in spontaneous labor on August 9, 2024. After an uncomplicated vaginal delivery, the placenta failed to separate within 30 min, and approximately 300 mL of bleeding ensued. Bedside exploration revealed pronounced placental adherence with difficult separation, raising intrapartum suspicion for placenta accreta spectrum. Ultrasound-guided curettage was undertaken; however, no definitive sonographic or intraoperative features of placenta accreta spectrum were identified, and complete removal could not be achieved. Notably, antenatal ultrasounds had not suggested placenta accreta spectrum, which supported proceeding with vaginal delivery. The cumulative estimated blood loss reached 800 mL, meeting criteria for PPH. Consequently, the patient received a transfusion (2 units of packed red blood cells, 200 mL fresh frozen plasma) and underwent bilateral UAE using absorbable gelatin sponge particles. Postprocedurally, prophylactic intravenous cefoperazone-sulbactam and metronidazole were administered alongside uterotonics.
Postpartum course and initial management (first hospitalization)
On postpartum day (PPD) 1 (August 10, 2024), pelvic ultrasound revealed a large (97.8 × 57.8 mm) intrauterine hyperechoic mass consistent with RPOC (Figure 1A). Subsequently, on PPD 3 (August 12), the patient developed high fever (39.5°C), though she remained hemodynamically stable, with markedly elevated inflammatory markers (WBC: 17.18 × 109/L, Neutrophils 90.9%, CRP: 220.99 mg/L, PCT: 0.168 ng/mL). Suspecting an infected RPOC driving the sepsis, manual placental removal was successfully performed under general anesthesia. The removed tissue appeared fragmented and malodorous; cultures were obtained. Post-procedure ultrasound revealed persistent intrauterine echogenic foci (Figure 1B), interpreted as likely representing post-UAE changes (embolic material artifact or localized reaction) rather than significant residual placental tissue. Inflammatory markers remained elevated on PPD 4 (CRP 248.45 mg/L, PCT 0.376 ng/mL). Initial blood cultures flagged positive for Enterococcus spp.; subsequent speciation confirmed Enterococcus faecium, mirroring the placental tissue culture results, which grew E. faecium (4+) and Bacteroides spp. Vancomycin was added to the antibiotic regimen. The patient defervesced by PPD 6 (August 15), with improving laboratory parameters. Repeat blood cultures were negative. Having completed a course of antibiotics and demonstrating clinical stability with resolving inflammation (CRP 52.44 mg/L), she was discharged on PPD 13 (August 22).
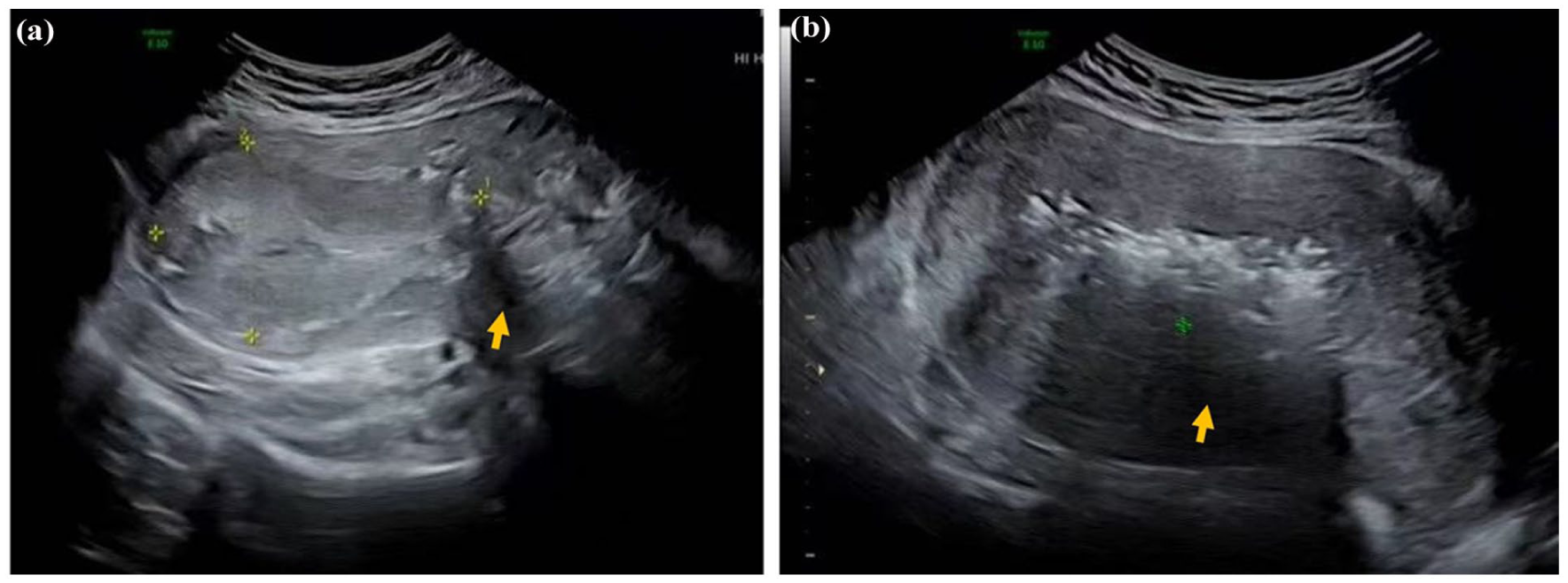
Pelvic ultrasound findings before and after initial placental removal. (a) Pelvic ultrasound on PPD 1 (August 10, 2024) showing a large hyperechoic intrauterine mass (arrow) consistent with retained placenta, with indistinct borders to the right cornual myometrium. (b) Pelvic ultrasound on PPD 3 (August 12, 2024), after manual placental removal, shows persistent intrauterine echogenic foci (arrow), with findings compatible with post–UAE changes.
Post-discharge course and readmission
The patient experienced recurrent fever beginning on PPD 27 (September 5). An outpatient ultrasound on PPD 29 (September 7) demonstrated a persistent heterogeneous intrauterine collection (58.3 × 27.1 mm). Serial serum β-hCG measurements remained negative (e.g., 0.43 mIU/mL), effectively excluding GTN; PCT was normal. During this period, alternative sources of fever were systematically evaluated and excluded—most notably respiratory infection (normal chest CT)—and no extrauterine infectious focus was identified. The patient initially declined the recommendation for readmission, opting for a period of observation instead. On PPD 32 (September 10), during this observation period, she developed a cough and was seen in consultation by our hospital’s respiratory service. Although a chest CT scan revealed no significant abnormalities, a course of oral linezolid was prescribed. This decision was not an empirical treatment for the cough but a targeted therapy based on the critical finding from her first hospitalization: the cultured Enterococcus faecium was vancomycin-resistant. Despite this intervention, her underlying uterine infection did not resolve, and she was ultimately readmitted on PPD 36 (September 14). Cervical cultures at this time yielded Parabacteroides spp. and Klebsiella pneumoniae. Laboratory tests confirmed persistent inflammation (WBC 13.39 × 109/L, CRP 86.39 mg/L). Pelvic examination revealed purulent cervical discharge and uterine tenderness, prompting readmission.
Second hospitalization: Further investigation and interventions
Intravenous linezolid and piperacillin–tazobactam were initiated, guided by available sensitivities. Although the patient’s temperature normalized by PPD 39 (September 17), purulent discharge persisted. Repeat cervical cultures on PPD 42 (September 20) grew K. pneumoniae (3+) and Eggerthella lenta (3+). Antibiotic adjustments were guided by these evolving culture results and sensitivities, reflecting the challenge of managing this complex polymicrobial infection. Ultrasound showed a persistent intrauterine/intramural lesion (39.2 × 33.6 mm). Given the refractory nature of the infection despite broad-spectrum antibiotics, a second ultrasound-guided curettage was attempted on PPD 45 (September 23). However, only a small amount (approximately 4 × 3 cm) of exceptionally tough, tenaciously adherent, necrotic-appearing tissue could be removed, a finding strongly suggesting pathology beyond simple RPOC or endometritis, raising concern for deeper myometrial involvement. The procedure was halted due to the difficulty and perceived risks. Postoperatively, metronidazole was added to broaden anaerobic coverage.
Definitive diagnosis and surgical intervention
Subsequently, on PPD 46 (September 24), the patient’s fever returned (39°C). Antibiotics were escalated to meropenem, vancomycin, and metronidazole. A pelvic ultrasound on PPD 47 (September 25) revealed clear lesion progression (now 56.8 × 35.7 mm) with extension toward the posterior serosa (Figure 2(a)). Crucially, a pelvic MRI performed the same day provided definitive evidence highly suggestive of extensive NM. Findings included uterine enlargement with a large, complex intrauterine/intramural lesion (7.0 × 4.3 × 4.6 cm) demonstrating heterogeneous signal intensity, significant peripheral restricted diffusion on DWI (consistent with abscess formation within necrotic tissue), marked posterior myometrial thinning, and focal bulging toward the serosa (Figure 2(b) and (c)). Histopathology from the recent challenging curettage confirmed extensive necrotic tissue with associated acute and chronic inflammation, but lacked definitive chorionic villi. Synthesizing the clinical picture of refractory sepsis, the failure of conservative measures (including repeated evacuation attempts), the evolving polymicrobial infection, and the compelling MRI findings, a diagnosis of NM was established. Given the MRI demonstration of extensive transmural posterior myometrial necrosis with serosal bulging, localized excision was deemed unsafe and unlikely to achieve definitive source control; accordingly, on PPD 48 (September 26), we performed a total laparoscopic hysterectomy with bilateral salpingectomy. Intraoperative inspection confirmed a severely necrotic, inflamed uterus—particularly involving the posterior wall—with full-thickness necrosis extending to the serosa, concordant with the MRI findings (Figure 3).
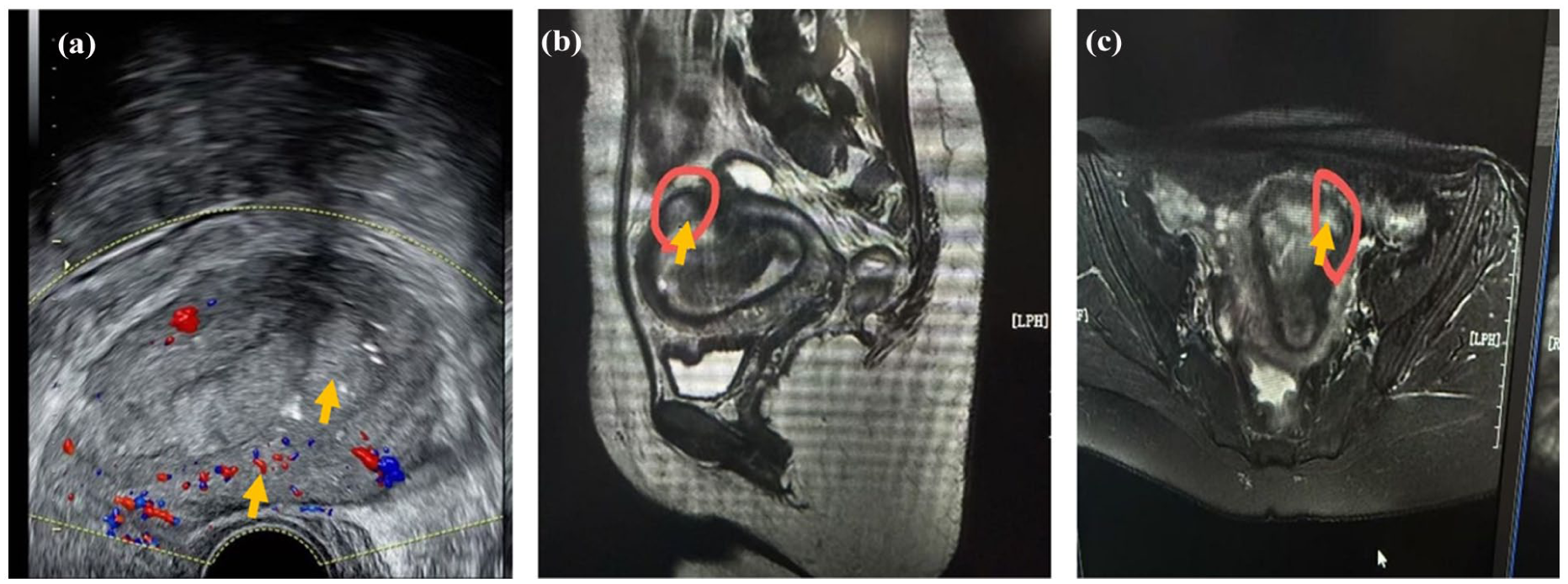
Pelvic imaging findings during second hospitalization (PPD 47, September 25, 2024). (a) Pelvic ultrasound shows a persistent, large heterogeneous region involving the uterine cavity and posterior wall, extending toward the serosa. Color Doppler ultrasound demonstrates areas of vascularity with scattered punctate signals (arrow). Note the suspected focal discontinuity in the posterior myometrium (arrow). (b) Sagittal T2-weighted MRI demonstrates uterine enlargement and a complex intrauterine/intramural lesion (region outlined in red) causing posterior myometrial thinning and bulging (arrow). (c) Axial T2-weighted MRI shows a complex lesion (region circled in red) with heterogeneous signal intensity. On diffusion-weighted imaging (not shown), the lesion demonstrates high signal consistent with restricted diffusion, suggestive of necrosis/abscess, with apparent focal thinning and a possible serosal breach of the posterior myometrium (arrow).

Gross pathology of hysterectomy specimen. (a) Posterior view of the uterus showing diffuse necrosis and discoloration of the posterior wall. (b) Sagittal section demonstrating full-thickness necrosis of the posterior myometrium with loss of normal muscle architecture. (c) Opened uterine cavity reveals a necrotic and inflamed endometrial cavity surface with no residual placental tissue identified.
Pathology and outcome
Final histopathology confirmed severe NM with extensive liquefactive necrosis and abscess formation, accompanied by severe endometritis, cervicitis, and bilateral salpingitis. The postoperative course was uneventful; the patient remained afebrile and was discharged on October 6, 2024.
Discussion
NM presents a formidable diagnostic and therapeutic challenge in the postpartum period, as illustrated by the complex case presented here. 1 This report adds to the limited literature by detailing a severe, polymicrobial case that developed after marked placental adherence with retained placenta, PPH, and UAE. This constellation of factors likely created a pathophysiologic milieu distinct from that of classic GAS-associated infection.
Compared with typical NM cases primarily caused by GAS,1,2 the present case showed a more protracted course lasting several weeks and recurrent episodes of sepsis driven by a dynamic polymicrobial flora that included Enterococcus faecium and Bacteroides species. This stands in contrast to the fulminant, early-onset (<72 hr postpartum) sepsis or TSS classically reported.8,15 The polymicrobial profile, particularly the anaerobic predominance, is more consistent with pathophysiologic models of NM secondary to postoperative tissue necrosis or ischemia, as observed after UAE.6,9 Prior reports of Finegoldia magna following UAE 6 and Fusobacterium necrophorum in suppurative myometritis with septic pelvic thrombophlebitis 7 support this inference. We therefore posit a multiple-hit pathophysiologic mechanism: initial mucosal barrier disruption during manual placental removal facilitated microbial entry; subsequent UAE performed for hemostasis likely induced uterine ischemia and further compromised tissue viability;6,9 ischemic and necrotic tissue then provided a favorable anaerobic niche for endogenous flora, and the underlying inflammatory state heightened susceptibility to infection.10,11 This cascade—mucosal injury, ischemia, anaerobic proliferation—appears to have culminated in extensive NM, confirmed by imaging and histopathology. Accordingly, we suggest that future research should explore biomarkers linked to these high-risk factors to enable earlier diagnosis.
Furthermore, this case underscores that conventional prophylactic antibiotic strategies may be inadequate for complex puerperal infections. The patient initially received cefoperazone-sulbactam and metronidazole, an empiric regimen theoretically suitable for gram-negative and anaerobic pathogens typically implicated in postpartum endometritis. However, the predominant pathogens ultimately identified included Enterococcus (intrinsically resistant to cephalosporins), Klebsiella spp. (often multidrug-resistant), and Eggerthella lenta (an anaerobe with recognized resistance), indicating insufficient initial coverage. Similar treatment failures have been reported. For instance, Tseng et al. 9 described uterine necrosis with acute suppurative myometritis after UAE in which empiric cefoxitin therapy failed and surgery was required. Likewise, Koshy et al. 7 reported persistent fever due to Fusobacterium necrophorum bacteremia in the setting of myometritis and pelvic septic thrombophlebitis, which resolved only after prolonged, targeted antimicrobial therapy. Taken together, these observations suggest that in complex or atypical postpartum infections, empiric regimens may fail to sufficiently cover critical pathogens, especially enterococci and resistant anaerobes. In high-risk scenarios such as retained placenta, UAE, or suspected tissue ischemia, broader-spectrum empiric coverage should be considered from the outset, with rapid de-escalation based on culture and susceptibility data. This strategy may help reduce the duration of ineffective therapy, minimize resistance selection, and improve maternal outcomes.
Importantly, the principal challenge in this case was not mechanistic uncertainty but the diagnostic ambiguity due to persistent, relapsing infection of unclear origin, which delayed definitive treatment. This highlights the need to refine diagnostic pathways. Although MRI ultimately proved essential for defining the extent of myometrial necrosis and directly informed the decision to perform total hysterectomy rather than localized excision, our experience and prior reports suggest that specific ultrasound findings can prompt earlier suspicion of NM. Tseng et al. 9 described “complex echogenicity within the myometrium” on initial ultrasound in a case of suppurative myometritis after UAE, later confirmed as central necrosis with abscess. In our case, Figure 2(a) revealed myometrial heterogeneity, reduced perfusion, and focal myometrial discontinuity, closely paralleling that case. Sonographic features that warrant attention include myometrial heterogeneity, diminished or irregular vascular flow, focal thinning or discontinuity of the myometrium, and hyperechoic foci with posterior shadowing suggestive of gas. While not pathognomonic, these findings, when present in the setting of persistent sepsis without intrauterine retention, should raise a strong suspicion for NM. We recognize that MRI is not always readily available, particularly in the absence of overt symptoms, and ultrasound remains the first-line modality in most institutions and should not be undervalued. In refractory postpartum sepsis, these specific sonographic findings should not be attributed solely to endometritis or RPOC but should trigger prompt pelvic MRI and early surgical consultation. A stepwise escalation in imaging and management may reduce diagnostic delays and improve maternal outcomes.
Conclusion
In summary, NM, though rare, is a life-threatening postpartum complication that warrants early recognition and decisive management. This case illustrates polymicrobial NM following retained placenta and UAE, highlighting a multifactorial pathogenesis distinct from classic GAS-related disease. Clinicians should maintain a high index of suspicion for atypical, protracted presentations, particularly after iatrogenic interventions or tissue ischemia, and empiric antibiotics may need broader coverage in such settings. While MRI is often essential for defining the extent of disease, early sonographic findings, such as myometrial heterogeneity and focal myometrial discontinuity, can serve as critical warning signs that should prompt expedited imaging and surgical consultation. Timely escalation of imaging and prompt intervention, including hysterectomy when indicated, are key to improving maternal outcomes in severe NM.
Footnotes
Acknowledgements
None.
Ethical considerations
Institutional ethics committee approval was not required for this case report according to local guidelines.
Consent to participate
Written informed consent was obtained from the patient for the publication of this case report and accompanying images.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review.
Author contributions
G.G. was the attending physician responsible for the patient’s primary clinical management and drafted the initial manuscript. L.F. conceptualized the case report, supervised the patient’s clinical course and the manuscript preparation, and provided critical revisions for intellectual content; L.F. is the corresponding author. Y.Z., J.C., and Y.F. participated in the patient’s clinical management and data acquisition. All authors contributed to manuscript revision, read, and approved the submitted version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting this case report are available within the article. Further details are available from the corresponding author upon reasonable request.
