Abstract
Uterine arteriovenous fistula is a vascular disease with a short circuit between the uterine arteries and veins. It is rare in clinical work with unclear incidence. Therefore, we aimed to summarize the disease characteristics of uterine arteriovenous fistula and provide a basis for clinical diagnosis and treatment. Herein, we present a case of cesarean scar pregnancy with uterine arteriovenous fistula in a 35-year-old woman, treated successfully with uterine artery embolization with preservation of her uterus. A preliminary diagnosis of cesarean scar pregnancy was made based on the patient’s history and MRI. Bilateral uterine arteriography was performed to diagnose a cesarean scar pregnancy combined with uterine arteriovenous fistula bleeding. Hysteroscopic electroresection was performed for the uterine scar. Massive uterine bleeding occurred during the operation. There was still active bleeding after medication and intrauterine compression balloon hemostasis. Urgent bilateral uterine artery embolization was performed. After the operation, leprorelin 3.75 mg was given by intramuscular injection three times, followed by oral contraceptive drospirenone ethinylestradiol tablets, one tablet a day, to prevent recurrence of uterine arteriovenous fistula. After outpatient follow-up every month, the patient was discharged from the hospital for 3 months. Menstruation returned to normal after drug withdrawal, and no abnormal vaginal bleeding symptoms appeared. Uterine arteriovenous fistula should be considered in the clinical treatment of patients with abnormal vaginal bleeding who cannot be diagnosed clearly. Uterine arteriography is the definitive method, and after interventional treatment, drugs can be used to prevent recurrence and close follow-up.
Keywords
Introduction
Uterine arteriovenous fistula (UAVF) is a vascular disease with a short circuit between uterine arteries and veins. It is a special form of uterine arteriovenous malformations, which is rare in clinical work with unclear incidence.1,2 In recent years, with the gradual increase in cesarean, the number of cesarean scar pregnancy (CSP) patients has also increased. UAVF and CSP can rupture blood vessels to a certain extent, leading to massive uterine bleeding and endangering life.3,4 At present, uterine artery embolization (UAE) is the most commonly used vascular interventional technique for treating CSP and UAVF. It is characterized by safety and a significant hemostatic effect. It can block the main blood supply to the uterus, causing local ischemia and hypoxia to accelerate the necrosis of villi, and achieve rapid and effective hemostasis. Prophylactic embolization can reduce the risk of massive hemorrhage during and after the operation, and also shorten the time for the resumption of menstruation after the operation.5,6 This paper reports the diagnosis and treatment of a patient with CSP combined with UAVF, and reviews the relevant literature.
Case report
A 35-year-old female patient was admitted to the hospital on July 16, 2024 due to “abnormal vaginal bleeding for 3 days after medical abortion.” Last menstruation (Figure 1): on May 16, 2024, the patient self-tested positive for human chorionic gonadotropin (HCG) in urine for more than 30 days after menopause. On July 2, the color Doppler ultrasound of the other hospital indicated that intrauterine pregnancy. On July 8, medical abortion (Mifepristone + misoprostol) was carried out in the other hospital. On July 13, vaginal bleeding occurred, and on July 16, a vaginal color Doppler ultrasound in our hospital indicated that the embryo had stopped developing. History of two cesarean section operations in 2011 and 2017.

The timeline of the diagnosis and treatment course of the patient in the present case.
Gynecological examination: a little dark red bloody liquid in the vagina, cervix smooth, uterine enlargement, no active bleeding. Auxiliary examination: Red blood cell count was 3.76 × 1012/L, hemoglobin 118 g/L, serum β-HCG 118,584.0 IU/L. Pelvic color Doppler ultrasound indicated that intrauterine examination and echo of the pregnancy sac with a size of about 50 × 27 × 40 mm, the cesarean section scar echo could be seen on the anterior wall of the lower uterine body, and the lower margin of the pregnancy sac reached the scar. The pelvic MRI plain scan showed that the uterus volume increased, the uterine cavity showed a cystoid pregnancy sac signal shadow, the lower margin protruding into the uterine scar in the lower anterior wall of the uterus, local endometrial line partition, and the thickness of the local scar was about 3.3 mm (Figure 2).

Pelvic MRI plain scan, the gestational sac is located in the lower segment of the anterior wall of the uterus at the scar site, where the muscle layer becomes thinner and discontinuous. (a) Coronal images, (b) sagittal images, (c) cross-sectional images.
After admission, combined with medical history and auxiliary examination, the diagnosis was as follows: (1) CSP; (2) after medical abortion. The patient’s pregnancy sac was located in the uterine scar, classified as type I CSP, suitable for hysteroscopic surgery. On July 18, 2024, Mifepristone 50 mg was administered orally every 12 h. On July 20, methotrexate 75 mg was injected into the anterior lip of the uterus for aborticidal chemotherapy. Hysteroscopic treatment was performed after embryo activity decreased. On July 22, under general anesthesia, “hysteroscopic electroresection for uterine scar” was performed. The hysteroscope showed that the lower portion of the uterus could see pregnancy-like tissue, about 5 cm in size, accompanied by blood clots and loose fibrous tissue. Ovarian forceps were used to extract part of the pregnancy, and the pathology was retained. Under hysteroscopic vision, the scar villi were subjected to multiple circular electric incisions, resulting in heavy bleeding and poor uterine contraction. Hysteroscopy was withdrawn, and the uterus was massaged. At the same time, active bleeding was still observed after rapid uterine aspiration after the application of oxytocin, carbetocin, carboprost tromethamine, and pituitrin, and the emergency arterial blood gas showed hemoglobin 85 g/L. The active bleeding flow was slowed down by intrauterine compression balloon placement. The total bleeding was about 1000 mL, and the emergency application was made for suspending erythrocyte 4 U and plasma 200 mL to correct the bleeding.
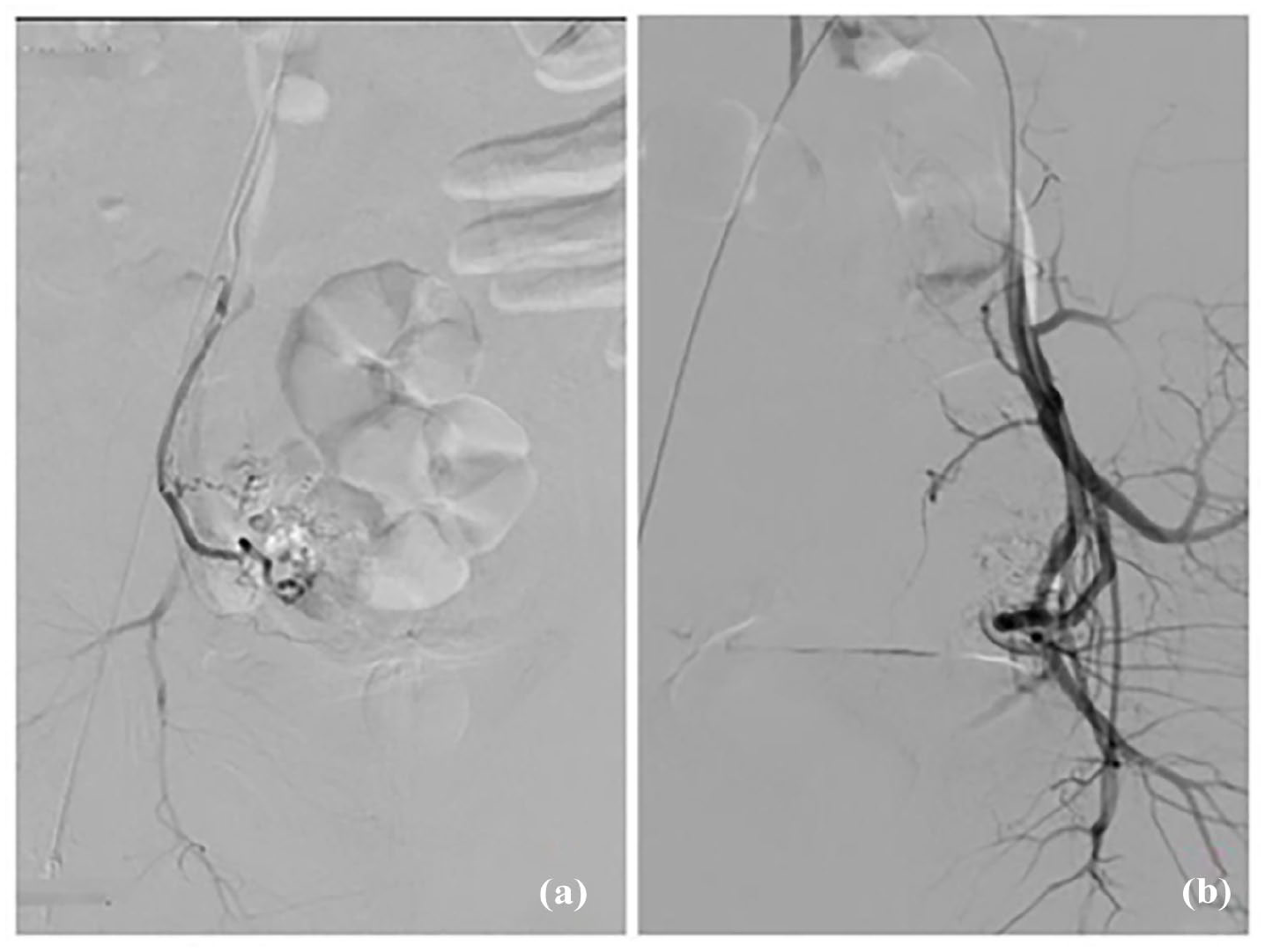
“Bilateral uterine arteriography + embolization” was performed urgently. During the operation, it was found that the bilateral uterine arteries were tortuous and thickened, and malformed vascular masses were visible at the distal end, and the contrast agent was foggy extravasation (Figure 3), which was diagnosed as CSP combined with UAVF bleeding. After the catheter was in place, 150–350 µm gelatin sponge particles with an appropriate amount of contrast agent were mixed and slowly embolized under the monitoring of digital subtraction angiography (DSA). The total dosage of gelatin sponge particles is about 100 mg. After embolization, the contrast agent was stagnant in the proximal section of the bilateral uterine arteries, and the malformed vascular masses in the distal end disappeared (Figure 4). No active bleeding was found after embolization, and the patient was returned to the ward for fluid rehydration and blood volume supplementation. On July 23, the first day after embolization, the uterine drainage tube extracted 10 mL of light bloody fluid, the intrauterine compression balloon was removed, uterine contraction was good, no obvious vaginal bleeding, hemoglobin 92 g/L. On the second day after embolization, there was no obvious drainage fluid in the drainage tube, no vaginal bleeding, and no other abnormalities, and the hemoglobin was 99 g/L. Postoperative pathology showed that the villi and decidual tissues were disintegrated by microscopy (Figure 5). After the operation, leuprorelin acetate 3.75 mg was given intramuscular injection, once every 4 weeks, a total of three injections, and then oral contraceptive drospirenone ethinylestradiol tablet, once a day, to prevent the recurrence of UAVF. On November 2, 2024, the patient was re-examined 3 months after discharge, and her menstruation returned to normal after stopping the drug, and no abnormal vaginal bleeding symptoms appeared.

Preoperative bilateral uterine arteriography, the bilateral uterine arteries are thickened and tortuous. The veins appear early and there are large draining veins. At the lesion site, disordered and malformed vascular masses can be observed. (a) Right uterine artery, (b) left uterine artery.
Postoperative bilateral uterine arteriography, the main trunks of the bilateral uterine arteries are clearly visible, and the malformed vascular masses have disappeared. (a) Right uterine artery, (b) left uterine artery.

Postoperative pathology.
Discussion
UAVF is classified as congenital or acquired. The former is very rare and stems from developmental disorders of vascular structure during the embryonic period, often accumulating multiple vascular communicating branches and other nearby organs. 7 The latter is often caused by uterine trauma such as vaginal delivery, cesarean section, induced abortion, pregnancy-related diseases such as gestational trophoblastic disease, placental implantation, infection, or exposure to drugs such as diethylstilbestrol, resulting in uterine injury or invasion of the uterine blood vessel wall, forming arteriovenous communication branches, and eventually forming UAVF.8–10 This patient was a woman of childbearing age who had undergone cesarean section twice. This hospital admission was mainly due to abnormal vaginal bleeding for 3 days after a medical abortion. In clinical practice, when women of childbearing age have a history of uterine operations and intermittent massive vaginal bleeding occurs after uterine operations, the possibility of UAVF should be considered after excluding diseases such as uterine fibroids, endometrial lesions, and dysfunctional uterine bleeding. 11 In addition, the history of uterine operations is closely related to acquired UAVF, and may be related to the number of uterine operations and the degree of injury, 12 suggesting that doctors should avoid damage to the uterine wall as much as possible when performing uterine operations to achieve medical purposes and avoid the formation of UAVF from the etiology.
Patients with UAVF have no specific clinical manifestations, and the typical clinical manifestations related to pregnancy are massive vaginal bleeding that suddenly stops 24 h after delivery. However, due to large individual differences, some patients may have repeated bleeding, a small amount of bleeding, or no obvious symptoms. In severe cases, hemorrhagic shock and disseminated intravascular coagulation can result in life-threatening outcomes. The cause of bleeding may be arteriovenous blood communication within the abnormal vascular structure, with large and rapid blood flow. Once the sac wall of the arteriovenous fistula is ruptured, fatal massive bleeding may be caused.13–15 This patient presented with a small amount of vaginal bleeding after medical abortion and hemorrhagic shock after hysteroscopic electroresection of pregnancy with uterine scar. Some studies have pointed out that the differences in UAVF bleeding signs between individuals may be related to the slow development of UAVF and the existence of a certain incubation period. 16 Moreover, some scholars have pointed out that this difference may be related to changes in hormone levels during pregnancy or the location and diameter of UAVF. 1 However, the correlation between the two is not clear and needs to be confirmed by further studies. In summary, UAVF patients are mainly characterized by uninduced vaginal bleeding, but the bleeding time and bleeding characteristics are different. Some patients may be accompanied by symptoms such as dull pain in the lower abdomen due to the lesion site, and the clinical manifestations are not typical, so the diagnosis should be confirmed by pregnancy and childbirth history, uterine operation history, β-HCG, Computed Tomography (CT), MRI, and other auxiliary examinations.
Ultrasound examination has become the preferred method for UAVF because of its advantages of safety, convenience, and economy. In the diagnosis of UAVF, color Doppler ultrasound is characterized by “low resistance, high speed blood flow, and colorful Mosaic blood flow signal.” However, due to the rarity of UAVF and the similarity between ultrasound imaging and rich blood flow signal in the myomyia shown by residual pregnancy tissue and trophoblastic diseases, it is easy to misdiagnosis and uncertainty in diagnosis.17,18 In this case, there was no rich blood flow signal of UAVF in the preoperative pelvic color ultrasound, which may be related to residual interference of pregnancy tissue. This suggests that when patients with CSP complicated with hemorrhage in clinical work, differential diagnosis should be made based on the bleeding characteristics and high-risk factors such as the history of uterine operation, and the possibility of complicating UAVF should be considered, and relevant examinations should be improved to confirm the diagnosis. DSA, as the gold standard for the diagnosis of UAVF, can quickly diagnose the lesions and bleeding sites of UAVF, and UAE can be performed to accurately block blood flow in the bleeding sites and play a rapid hemostatic role. However, since this angiography is an invasive procedure, it is usually not the first choice for patient diagnosis. 19 In this patient, the preoperative pelvic color ultrasound and MRI plain scan did not indicate UAVF, and the emergency DSA was performed to diagnose UAVF clearly, suggesting that in the diagnosis process of UAVF, for patients with high-risk factors such as multiple pregnancy history and history of uterine operation, enhanced MRI scan can be used to further confirm the diagnosis and avoid missed diagnosis. 9
The treatment of UAVF should be formulated according to the patient’s age, vaginal bleeding volume, severity of urgency, lesion site size, fertility requirements, and other comprehensive conditions, including conservative treatment, drug treatment, and surgical treatment.20–22 For those with mild symptoms, conservative treatment may be attempted, including intrauterine compression balloon, intravenous use of pituitrin, oral contraceptives, and intramuscular injections of gonadotropin-releasing hormone analog (GnRH-a). Intrauterine compression balloon and intravenous administration of pituitrin are mainly used to control acute bleeding.23,24 Oral contraceptives and intramuscular GnRH-a can reduce estrogen levels and promote lesion atrophy, and are often used to prevent UAVF recurrence after symptom relief. 25 This patient received three injections of GnRH-a after UAE, followed by oral contraceptives. Menstruation returned to normal 3 months after surgery, and the disease did not recur. With the improvement of interventional embolization technology, UAE has become the preferred treatment for UAVF patients who need to preserve fertility function, with a success rate of up to 96%, and has the advantages of less damage, fewer complications, and faster recovery. 26 For patients with CSP with UAVF, UAE can identify the location of the lesion and the bleeding situation, and then accurately block the bleeding site, so as to preserve the patient’s fertility function without affecting the endocrine function, which has high applicability and safety. 27 Effective embolization of the bleeding artery or the superior vessels of the bleeding artery is the key to rapid hemostasis. In this case, gelatin sponge granules were used as the main embolization material, which was successful once, and no blood was sent out after surgery, and the uterine function of the patient was preserved. Some studies reported that vaginal bleeding occurred again after gelatin sponge granule embolization, and then polyvinyl alcohol (PVA) granule was selected for embolization. 28 With the development of medical technology, the choice of embolization materials is gradually increasing, but unfortunately, there is no large sample clinical control study at this stage to show which embolic agent is better and the recurrence rate is lower. Therefore, the selection of clinical embolic agents should be considered in combination with the patient’s condition and clinical technology.
Conclusion
The clinical manifestations of CSP patients with UAVF bleeding often lack characteristics, and the bleeding characteristics are different. Attention should be paid to the medical history, especially the fertility history, uterine operation history, and surgical history. During ultrasound examination, attention was paid to the results of color Doppler ultrasound flow imaging and blood flow spectrum. When clinically suspected of UAVF, individual treatment measures should be selected according to the patient’s symptoms, vital signs, and fertility requirements. When conservative treatment fails or symptoms are severe, an MRI enhancement scan, or even uterine arteriography should be performed in time for further diagnosis and treatment. UAE should be followed by relapse prevention drugs such as GnRH-a and oral contraceptives, close follow-up for recurrence, recurrence patients can be repeated UAE or focal resection, and other treatments. In clinical practice, we should enhance the understanding of UAVF, improve the diagnosis and treatment ability of UAVF, and reduce the pain of patients.
Footnotes
Ethical Considerations
Ethical approval to report this case was obtained from the medical ethics committee of the Third People’s Hospital of Hefei (approval number: 2025LLWL013).
Consent for Publication
Written informed consent has been obtained from the patient for their anonymized information to be published in the article.
Author Contributions
Conceptualization: Lei Qi. Data curation: Hongzhi Sun. Resources: Lei Qi, Peng Ji. Formal analysis: Hongzhi Sun, Tingting Wu. Supervision: Maoneng Hu, Peng Ji. Funding acquisition: Maoneng Hu, Peng Ji. Investigation, Methodology: Lei Qi, Hongzhi Sun. Project administration: Lei Qi. Writing – original draft: Lei Qi. Writing – review and editing: Peng Ji.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by 2024 Annual Research Project on the Inheritance and Innovation of Traditional Chinese Medicine in Anhui Province (2024CCCX235) and The Seventh Cycle Key Specialty Construction Project of Hefei City (Hewei Medical Secret (2023) No. 72). The funder had no role in the study design, data collection, data interpretation, writing of the report, decision to publish, or preparation of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
