Abstract
Natural killer cells are the most researched and employed cells for immunotherapy, which have expanded from autologous immune-enhanced therapies to genetically enhanced cell therapies such as chimeric antigen receptors natural killer cells. The present clinical case study aims to analyze and evaluate the safety and tolerability of ex-vivo expanded autologous natural killer cells via an in-house developed culture protocol. The novel, cultivation protocol enables high-yield and pure natural killer cell batches free of antibody usage, prior natural killer cell purification, or any magnetic-bead labeling, resulting in a fully human compatible cell expansion process. In this report, a total of nine patients were clinically infused with the in-house cultured natural killer cells and were monitored pre- and postinfusion for haematological parameters, liver function profile, hypersensitivity, inflammatory markers as well as pretumor markers to assess for the possibilities of natural killer cell-induced or any dose-dependent toxic/adverse reactions. The outcomes indicate that the patient infused with natural killer cells demonstrates good level of clinical safety with no signs of hepatotoxicity, hypersensitivity, inflammation, or any flares of preliminary tumor markers. The study provides supportive evidence of the clinical tolerance of autologous-derived natural killer cells cultured via the in-house protocol on patients in Malaysia and establishes a baseline for future clinical studies with a wider range of patients.
Introduction
Autologous immune enhancement therapy (AIET) is an adoptive cell therapy which involves the ex-vivo isolation, expansion, and re-infusion of patient-specific immune cells to improve their resistance in combating human disease (e.g. cancerous growths, bacterial or viral infections, anti-aging).1–3 AIET has displayed better immune priming for health-compromised patients, demonstrating strong cyto-toxicity against tumors, bacterial infections, and senescent cells that exhibit antiaging properties as well as promoting cytokine profile enhancements and monocyte activations.3–7 Natural killer (NK) cell therapy is an example of such AIET-based treatments. NK cells are part of the innate immune system that displays spontaneous cytolytic activity against biologically stressed/damaged cells such as cancerous cells, virus-infected cells, or any worn-out cells without prior sensitization, making them an ideal therapeutic agent of immunotherapy. NK cells also secrete several cytokines upon activation such as interferon-γ (IFN-γ), tumor necrosis factor-alpha, IL-2, and IL-10 that can modulate the function of other innate and adaptive immune cells. 8
A multitude of research and trials have been conducted using NK cell therapies for disease treatments which exhibit both good efficacy and safety.9,10 However, there is a lack of case studies that elaborate on the clinical safety and tolerability aspects of NK cells, centered around patient settings in the Malaysian population. The present case report focusses on analyzing and evaluating the safety aspects of ex vivo expanded autologous NK cells among the patients in Malaysia. The autologous expanded NK cells are cultured via an in-house developed and patented (MyIPO; PI2025002484), unique culture protocol. The protocol’s novelty stands by being free of animal components and free of additional steps such as antibody stimulants, antibody-coated plates, feeder cells, and any prior NK cell purification steps via magnetic bead-labeling. The aforementioned techniques are conventional methodologies used by several authors.11–17 in culturing autologous NK cells from patient blood. Our protocol stands-out by eliminating such tedious steps which in-turn both consolidates the Malaysian current Good Manufacturing Practice (cGMP) compliance of the process and reduces the overall cost which enables facilities to direct the monetary resources to incorporate GMP-grade raw materials (e.g. GMP-grade interleukins, media) instead of lower-quality grade input materials. This benefits further by avoiding the need for end-stage validation steps necessary to identify the residuals carry-overs of such components (i.e. residual antibodies, Xeno-residues, magnetic bead residues, or any feeder-cell residues), providing better control and ensure clinical safety of the cell products for human use.
This clinical study reports the examination of nine patients in Malaysia, following their infusion of the in-house developed NK cells and the monthly follow-ups conducted. The patients were examined for their hematological parameters, liver functions, and tumor markers to identify the presence of adverse effects and tolerance of the patients to NK cell infusions. The case series is presented as per the CARE (consensus-based clinical case reporting) checklist, which demonstrates real-world outcomes for patients who received NK cell infusions at ALPS Medical Centre. The study highlights and provides insight into the primary concerns, being the safety and well-being, of infusing NK cells for overall wellness and disease treatments (Supplemental Material).
Patient case description
This clinical study was registered with the National Medical Research Register (ID-25-00333-MUM). Data were extrac-ted following the ethical principles of the Declaration of Helsinki and Malaysian Good Clinical Practice.18,19 The study involved nine nonconsecutive patients who received NK cell infusions, at ALPS Medical Centre to study and establish the safety aspects of ex-vivo expanded autologous NK cells (MyImune). The clinical investigation was done in the years 2023 and 2024. The patients were selected based on specific inclusion and exclusion criteria and their data were recorded.
The patients were prescreened based on a stringent selection criterion. The patients were selected based on an age group of more than 18 years and above. The patients were subjected to a thorough examination of their medical history, physical traits and well-being, and any underlying disease conditions to ensure their appropriate vital signs and to prevent any unpredicted exacerbation of existing medical con-ditions. Patients with autoimmune conditions were strictly removed or excluded from the list due to the possibility of hyper-immune reactions immediate or postimmunotherapy interventions.20–22 The patients were also subjected to pathological tests for infectious diseases such as Human immunodeficiency viruses (HIV), Hepatitis (HEP) B/C, Cytomegalovirus (CMV), or syphilis to prevent the entry of contaminated blood samples. The patients’ hematological parameters, liver function markers, and tumor markers were analyzed as an initial data record to establish the patient/case-specific baseline values. The patients were monitored closely within a minimum of 2 weeks to 1 month time, for posttherapy analysis following NK cell doses (Figure 1).
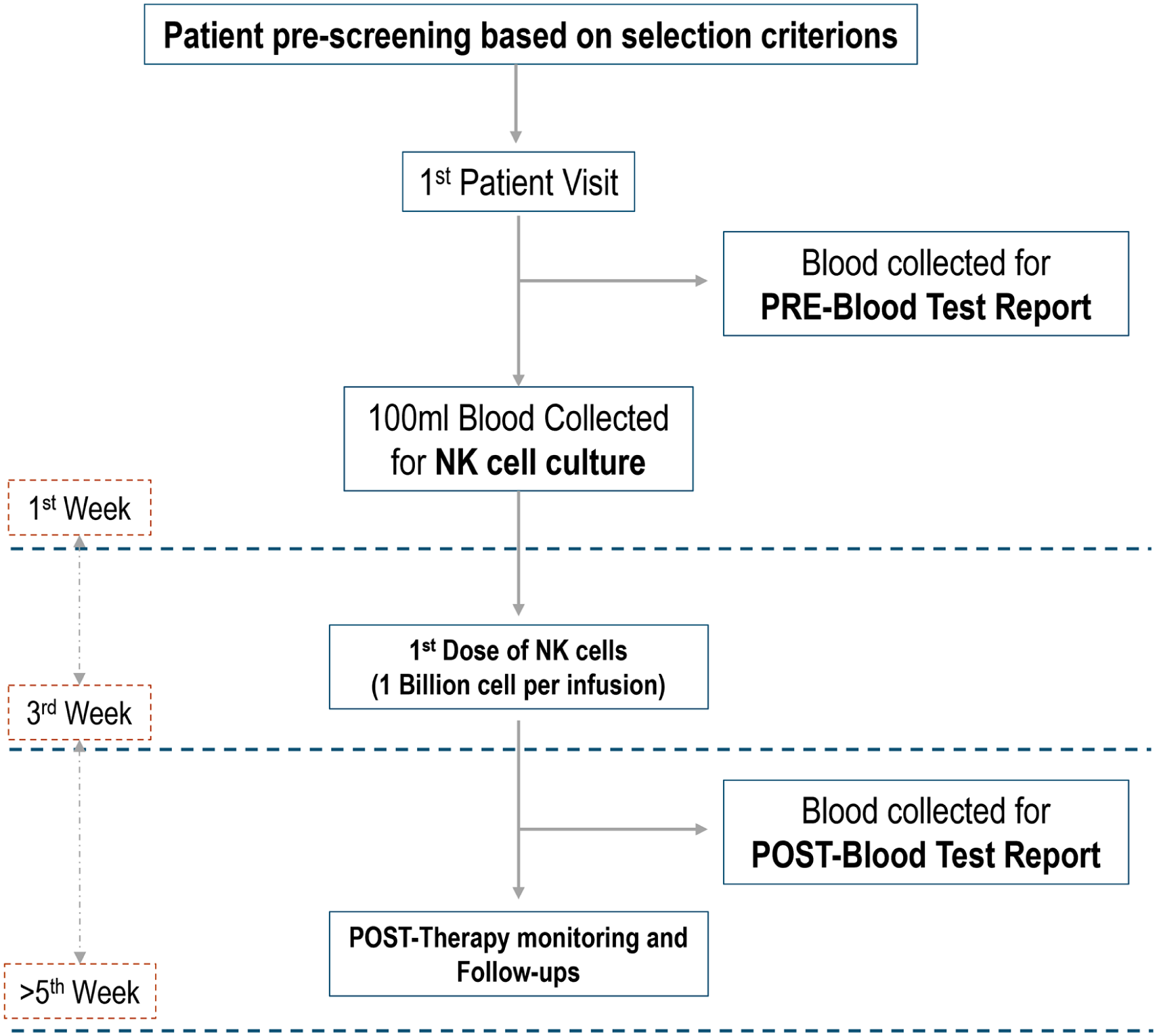
CONSORT flow diagram which details the flow from patient screening (described in detail in section “Patient case description”), until the post-monitoring and follow-ups.
The case series, conducted under institutional exemptions and according to the section 6.9, subsection D “Case reports with informed consent from patients” by the Malaysian National Institutes of Health Guidelines for Conducting Research under the Ministry of Health Malaysia, comprises nine patients/individuals where some patients are healthy individuals, while others experience health complications primarily being cancerous. The patient information has been summarized in Table 1. Following NK cell infusions, continuous monitoring is implemented to identify any forms of discomfort, stress, and even potential hyper-adverse reactions from the patients before being discharged from daycare. Patients are advised to promptly communicate any new symptoms such as fever, vomiting, insomnia, redness, or pain at the injection site, rash, or shortness of breath to the medical team at ALPS Medical Centre. After a minimum of 2 weeks to 1 month time, the patients are scheduled for postinfusion checkups, physical examinations, doctor consultations, and bioparameter analysis to ensure and study in detail the safety and tolerability aspects of the in-house developed NK cell product.
Summary of patient case pre-NK cell infusions.

DLBCL: diffuse large B cell lymphoma; NK: natural killer.
Case series
Case 1
A 55-year-old female patient diagnosed with one-side hemiplegia, which developed due to lymphocytic brain tissue infiltrations, primarily caused by diffuse large B cell lymphoma (DLBCL). The patient exhibited elevate blood pressure (BP) reading of 138/92 mmHg, pulse rate (PR) of 69/min, and an oxygen saturation SpO2 of 98%. The patient had no history of alcoholic consumption, smoking, and consumption of recreational drugs. The patient was under methotrexate–chemotherapy prior to treatment administration and tested negative for any infectious diseases. Continuous follow-up and monitoring were implemented, including physical examinations and blood analyses. The patient reported no safety concerns and no further aggravation in existing disease condition.
Case 2
A 37-year-old female patient diagnosed with nasopharyngeal cancer with no history of alcoholic consumption, smoking, or consumption of recreational drugs. The patient was in the remission phase after routine chemotherapy. Vital signs display a normal BP of 106/69 mmHg, PR of 62/min, and SpO2 of 100%. The patient raised no safety concerns post-NK therapy.
Case 3
Elderly male, 72 years old, with no diagnosed disease conditions and no history of smoking and consumption of recreational drugs, reported vital signs of normal BP of 115/87 mmHg, PR of 67/min, and SpO2 of 98%. The patient was marginally prediabetic with high blood glucose content and was tested negative for any infectious diseases. The patient exhibited no signs of safety concerns relevant to post-NK treatments.
Case 4
A 70-year-old male, with a medical history of cancer-bearing family lineage, was further diagnosed with elevated mRNA precancer markers for lung cancer. The patient had a history of taking antihypertensive medication, with slightly high BP 129/81 mmHg, high PR of 95/min, and SpO2 of 97%. The patient reported no alcoholic consumption, smoking, or consumption of unprescribed, recreational drugs. The patient was also tested negative for any infectious diseases. Subsequent follow-ups demonstrated no hepatotoxicity, no negative signs in the renal, and blood parameters with no concerns of vital sign or safety aspects postinfusions.
Case 5
A 53-year-old female patient under remission phase of lung carcinoma, with normal BP of 117/56 mmHg, PR of 76/min, and SpO2 of 96%, reported no history of alcoholic consumption, smoking, or consumption of unprescribed, recreational drugs, as well as tested negative for infectious disease. The patient reported no safety and health concerns posttreatments.
Case 6
A 72-year-old male, with prediabetic conditions and high blood cholesterol, evident from high BP reading of 178/95 mmHg, slightly high PR of 84/min, and SpO2 of 97%; reported no history of smoking and consumption of unprescribed, recreational drugs, as well as tested negative for infectious disease. Postinfusion demonstrated no safety concerns as well as no changes in blood glucose and cholesterol levels.
Case 7
A 56-year-old female with prediabetic conditions, with signs of normal BP of 102/70 mmHg, PR of 78/min, and SpO2 of 97%. Reported no history of alcohol consumption, smoking, or consumption of unprescribed, recreational drugs, as well as tested negative for infectious disease. Posttreatment indicated overall good wellness and health, with no signs of safety concerns.
Case 8
A 82-year-old male with a weak body constitution, and an overall high BP of 143/65 mmHg, normal PR of 62/min, and SpO2 of 97%, and diagnosed with stroke. Reported no history of alcohol consumption, or smoking, and tested negative for infectious diseases. Posttreatments revealed no hepatotoxicity; overall good lymphocyte counts and overall wellness. Patient similarly raised no issues of safety concerns.
Case 9
A 47-year-old female, with overall weak body constitution, PR of 63/min, and SpO2 of 97%. Reported no history of alcohol consumption, smoking, or consumption of recreational drugs, and tested negative for any infectious diseases. The patient reported no safety concerns relevant to the NK cell therapy.
Preparation and infusion of NK cells
NK cells were prepared by obtaining a minimum of 100 mL of patient peripheral blood. The peripheral blood mononuclear cells (PBMCs) we isolated using the conventional Ficoll-density gradient centrifugation method with cell washing. The isolated PBMC were then seeded at 1 million cells/mL of Roswell Park Memorial Institute 1640 culture media, supplemented with heat-inactivated plasma of 5% and cytokine stimulants combination, primarily IL-2. The in-house developed culture protocol consists of a total 18-day culture period which yields more than 90%–95% viability per production batch/patient infusion round. The harvested cells prior to release are subjected to quality control checks to ensure a minimum criterion is achieved for batch release. These quality control criteria include (1) cell morphology, (2) NK cell purity (CD56+, CD16 surface markers; CD3− surface markers), (3) yield, and (4) batch sterility which will be analyzed and confirmed for the presence/absence of bacterial contamination, mycoplasma, and endotoxins. Once quality checks are considered as accepted/passed, the cells will be harvested and packed into 0.9% saline solutions (injectable grade) and prepared for patient in-vivo infusions at ALPS Medical Center. The cells are cultivated and packed in a Malaysian cGMP-compliant cleanroom environment to ensure the safety and sterility of the cultured cell products. The patient’s bioparametric data obtained and recorded pre- and postinfusions were statistically analyzed using GraphPad prism, (Dotmatics, US).
Observations and discussions
NK cell characterization and batch measures
NK cells prior to harvesting and packing for clinical infusions are subjected to quality control tests and assays to ensure the cultivated cells are within the accepted release criteria such as visible morphology, viability, yield, purity, and sterility. The cultured NK cells via the patented culture methodology are primarily analyzed for purity through flow cytometric analysis qualified using the CD56+ and CD3− cell surface markers. NK cells with a minimum of 70% or more of the CD56+ markers and <2% CD3− markers are standardized as the accepted range for pure NK cell batches (Figure 2(b)).13,23,24 NK cells are also visually inspected during the culture process for morphological evidence of cell clumping/clustering that indicates the presence of healthy and live NK cells (Figure 2(a)). Each batch of cultured NK cells is further sampled and analyzed for sterility aspects; where the bacterial contamination (no growth), mycoplasma (qPCR method; detection limit ND <100 copies), and endotoxin levels (Limulus Amebocyte Lysate kit, ND <0.25) must be within the accepted ranges before infusions. Table 2 comprises the details and the quality control measures implemented on each batch for each patient case, from blood collection to NK cell characterization before infusions.

(a) NK cell morphology observed with cellular clustering, 40× magnifications. (b) Purity of NK cells analyzed through CD56+ and CD3− markers.
Summary of NK cell batch yield, purity, morphology, and sterility measures.

NK: natural killer.
The yield is measured as billion cells per dose.
Mycoplasma is measured using qPCR detection.
For this clinical study, the NK cells expanded ex-vivo via the culture protocol were also prior analyzed for both cytotoxic and cytokine-profile assays to ensure the in-house cultured NK cells are both active and functionable batches. However, these test cytotoxic test parameters are not inclusive in the quality control batch measures as both the timing and duration to complete the test will not coincide with the batch culture time and harvest; hence, the date given here are from pilot cultures derived via the same protocols. It is also important to understand that pilot testing of in-vitro cytotoxic NK cell activities does not correspond to the same in vivo cytotoxic effects against cancer cells; therefore, requires further studies to be conducted on this aspect since the current study only focuses on the safety and tolerability in clinical settings. The cytotoxic and cytokine parametric measures are based on the cytotoxic ability of the NK cells to kill neighboring cancer cells and the secretion of IFN-γ which directly corresponds to the active NK cell profile.8,25–27 The cytotoxic NK cell measures are conducted by prior NK cell culture via the in-house protocol which is then co-incubated with freshly cultured K562 cells (leukaemia cancer cell lines). The cells are mixed at equivalent concentrations and are co-cultured at 0, 4, 6, and 10 h of incubations. The cell populations are measured through flow cytometry at each hour using cell-death and viability markers (i.e. Annexin V and 7AAD) which indicate the presence of cell death/induced apoptosis of the K562 cells, identified through unstained control runs and corresponding cytometric population gating. The NK cell IFN-γ secretion parameters are simultaneously measured from the co-incubated culture media supernatant, using available ELISA-based detection kits and using the anti-CD107a marker via flow cytometry, specifically measured during NK cell co-incubation with the cancer cells. IFN-γ and CD107a are common immunological activity and degranulation markers measured for NK cells which define the ability to exert cytolytic properties. 28
As seen in Figure 3, the NK cells are observed to induce cell death of the model cancer cell following 4 h of incubations and onwards, where the Annexin V and 7AAD markers show a statistically significant difference from the first hour (0 h) to fourth and the sixth hour of incubations, with decreasing percentage of the K562 cell population with time (Figure 3(a)–(c)). The IFN-γ secretion and CD107a secretion are also measured parallely, which indicate functionally active or activated NK cells in terms of cytokine secretion and degranulation events of releasing granzymes and perforins for cytolytic effects. From Figure 3(a) to (c) we can see that the CD107a secretion levels are consistently higher from zero to the sixth hours of co-incubation which demonstrates an activated state of the NK cells, that is, likely due to the stimulation provided by the interleukins used during the culture procedure known to both stimulate and activate NK cell populations.

Flow cytometry analysis of NK cells and K562 cancer cell population at (a) 0 h, (b) 4 h, and (c) 6 h of co-incubations, with cell-death markers (i.e. 7AAD and Annexin V) and CD107a degranulation markers arranged from left to right. (d) IFN-γ measured at 0, 4, 6, and 10 h of co-incubations of cancer cells with nonactivated PBMCs and activated NK cells using ELISA assay (average optical density (OD) values are stated on top the columns).
Further evidence can be seen in Figure 3(d), where the co-incubation of only the freshly isolated PBMCs (non-NK cell cultures) and cultured NK cells with the model cancer cells were compared. The PBMCs before any interleukin stimulation and activations from the 0 to 10th h of co-incubation do not secrete any significant IFN-γ levels where only after NK cell expansions, the IFN-γ levels are detected during the 0 h of co-incubation and onward. This additionally corroborates the inference for interleukin-induced NK cell activations and potential antitumor/anticancerous properties.
Safety and tolerability measures
The current study reports the patient factors such as liver functions and tumor markers as well as the several focussed tolerability factors pre- and post-NK cell infusion to measure the safety and well-being of the patient in the study. Table 3 summarizes the measured factors/markers pre- and postinfusions. From the reported values, we can observe that the majority of the focussed markers and factors are unchanged, indicated by the absence of any statistical significance between the baseline values and values measured post-NK cell infusions.
Summary of hematological parameter, liver functions, and tumor markers pre- and postcell infusions.
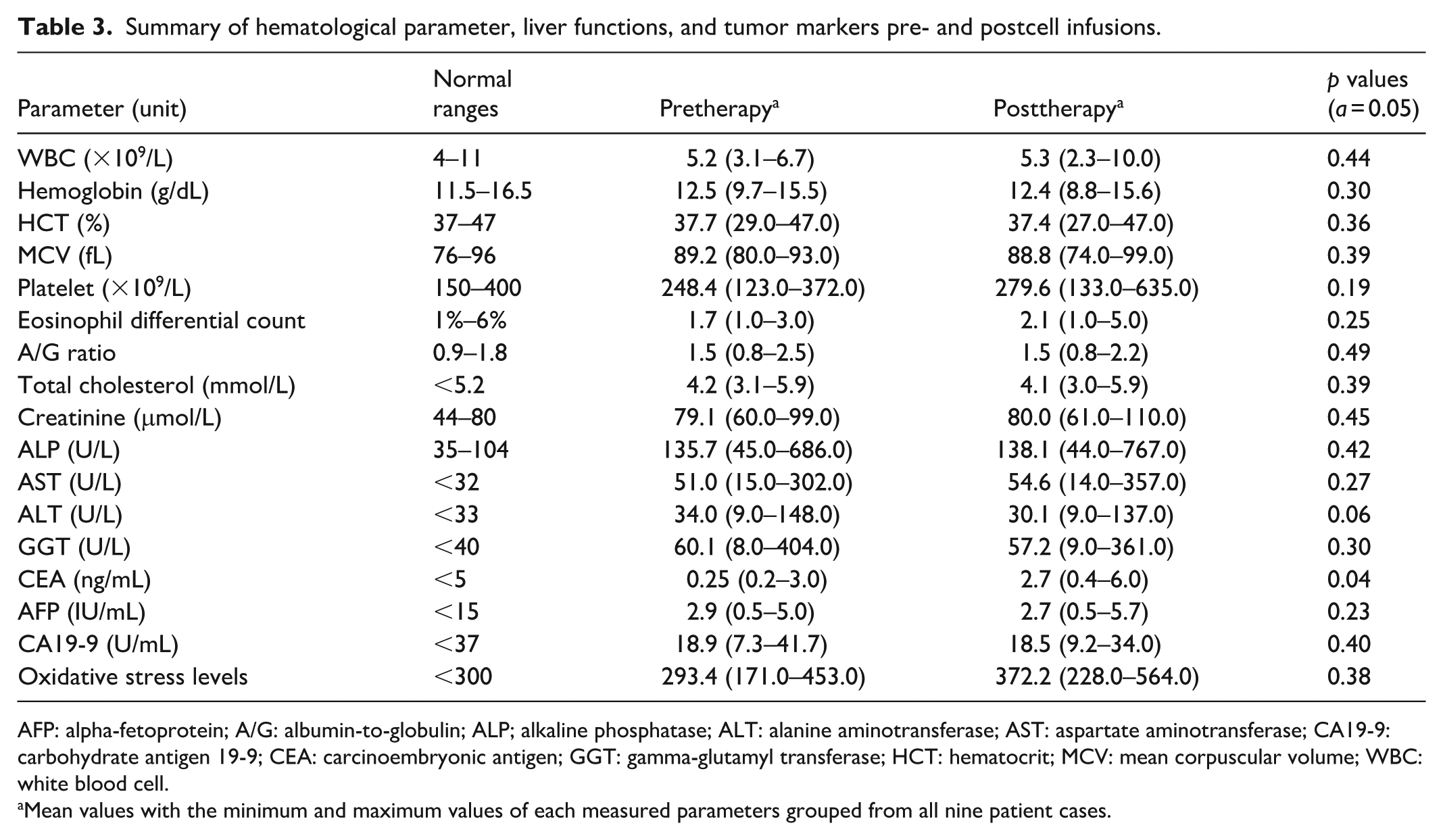
AFP: alpha-fetoprotein; A/G: albumin-to-globulin; ALP; alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; CA19-9: carbohydrate antigen 19-9; CEA: carcinoembryonic antigen; GGT: gamma-glutamyl transferase; HCT: hematocrit; MCV: mean corpuscular volume; WBC: white blood cell.
Mean values with the minimum and maximum values of each measured parameters grouped from all nine patient cases.
Upon thorough data scrutinization, it is observed that the hematological parameters that include the white cell counts, hemoglobin levels, hematocrit, mean corpuscular volume, and platelet counts remain the same for all cases. Although it can be argued that the white blood cell count should be higher post-NK cell infusions, similar findings have been reported by several authors which state that the NK cell count is unchanged postinfusions and are short-lived to within 1–4 weeks of life span.29–31 These inferences can explain the decline/unchanged measure of the white cell counts after 1 month of postanalysis. Furthermore, liver function profiles measured via liver enzymes such as alanine aminotransferase (ALT), aspartate aminotransferase, alkaline phosphatase, and gamma-glutamyl transferase 32 also demonstrated unchanged values pre- and postinfusions, indicating the absence of any liver damage. This also included physical examinations of the liver profile such as eye and skin yellowing. Previous reports have stated that altered immune conditions through immunotherapy, largely involving immune checkpoint inhibitor drugs, can lead to immunotherapy-induced hepatotoxicity. 33 However, the data collected suggest that the autologous-derived NK cell infusions did not trigger such conditions, observed within this study.
Furthermore, in studying the safety and tolerability aspects, we also focussed on the other selected markers; (1) eosinophil count, (2) serum albumin-to-globulin (A/G) ratio, and 3) oxidative stress levels. The eosinophil counts directly correspond to hypersensitive reactions, while the A/G ratio and oxidative stress levels are a preindicators of inflammatory reactions.34–36 The absence of any increase in these markers from the data analyzed indicates the patients experienced neither hypersensitive reactions nor inflammatory responses postinfusions. Pre- and postanalysis of tumor markers such as the mean alpha-fetoprotein, and carbohydrate antigen 19-9 also reveal normal range values. Interestingly, it can be observed that the carcinoembryonic antigen (CEA) markers reported a marginally significant increase post-NK cell infusion. However, the patient (case no. 9) reported elevated levels of liver ALT and marginally lower A/G ratio indicative of mild symptoms of inflammation which can also trigger a flare in CEA marker values.37–39 Since this is an isolated event and the inclusion of the data point into the set of parametric data comparison will skew the values to false significant ranges, we can assume that such increase in precancer markers while unlikely to be caused by the NK cell infusion, further investigation is warranted to fully elucidate the mechanism. The inference may also apply to any NK cell infusion-induced hepatotoxicity as the NK cells used are autologous in origins and can cause no such deleterious effects postinfusions. Nevertheless, future studies are required to understand in-details of such medical events.
Moreover, to further the safety aspects of the present NK cell clinical infusion, it can be observed that one of the patient’s cases (case no. 1) within the study scope is diagnosed and identified with blood cancer (DLBCL). As the NK cells are ex-vivo expanded through an autologous mode of therapy, the possibility of re-culturing and expanding the cancerous B cells from the extracted patient blood is highly probable. Hence, an additional cell culture harvest/release criteria was included specifically for this patient case. The patient case-specific NK cell batches were analyzed for positive DLBCL cancer-specific markers; CD20, CD79a, monocyte, undifferentiated macrophage-1, and B-cell lymphoma-6 antibodies medically confirmed through patient histopathological reports. From Figure 4 and Table 4, it can be inferred that the final cultured cell batch is free of such markers with no statistically significant difference between the control/normal values (non-leukemic samples), and the patient-batch (leukemic samples) measured values, which could be an attributed stemming from the high NK cell enrichment via the in-house culture protocol. Although CD79a B-lymphoma markers were observed to decrease further after NK cell cultures, the exact reason and mechanism underlying this significant decrease is unknown and requires further analysis. Additionally, such forms of auto-logous NK cell immunotherapies are explicitly important for cancer/tumors growths which do not benefit from surgical removal such as tongue cancer 40 and nasopharyngeal cancer 41 (as seen on our case series study, case no. 2). The physical deformation and inabilities caused by surgical interventions of such cancers far outweigh any long-duration effects of NK cell therapies, enabling patients to continue on with reduced career or life burdens.

Flow cytometry analysis of (i) CD79a and CD20 marker, (ii) BCL-6 marker, and (iii) MUM-1 markers (a) before and (b) after ex-vivo NK cell culture from blood of lymphoma patients (case no. 1).
The cell surface markers corresponding to diffuse large B cell lymphoma.

BCL-6: B-cell lymphoma-6; MUM-1: monocyte, undifferentiated macrophage-1; PBMC: peripheral blood mononuclear cell.
The tables with “superscript letters a and b” are reported as average values, while “a” are normal percentage measured from PBMC samples of nonlymphoma individuals as three replicates each.
The data observed and discussed here strongly supports the safety and well-being aspect of the autologous, clinically infused NK cell products. All nine patients reported no signs of fever, pain, and any other physiological abnormalities throughout the follow-up period.
Conclusion
In conclusion, the presented findings in this case study of the in-house developed NK cell clinically translate as safe to be used, with no signs of hypersensitive reactions, inflammation, hepatotoxicity, or any tumor marker promotions identified during the follow-up period. The NK cells also exhibit good levels of cytotoxicity toward in vitro cultured and co-incubated cancer cell models, tested based on pilot cultures instead of an inclusive test during batch release.
Although the current results support their clinical safety and tolerability, the study lacks any proven clinical efficacy toward a specific disease indication. This enables the study to be inclined toward more case series rather than a complete clinical study with a target medical condition and its symptoms pre- and posttherapy. The study also current describes an overall safety and tolerability aspects over a broader scope of medical conditions with fewer patient cases.
The future aspects of this study will focus on anticancerous properties of targeted cancer conditions to further evaluate their efficacy for cancer immunotherapy, where the study will be more oriented toward the clinical outcomes of targeted disease/medical conditions and with better case series numbers and corroborate the scientific findings.
Supplemental Material
sj-pdf-1-sco-10.1177_2050313X251376950 – Supplemental material for Purification and antibody-free ex-vivo expansion of peripheral blood-derived autologous natural killer cells: A Malaysian case series on patient safety and tolerability
Supplemental material, sj-pdf-1-sco-10.1177_2050313X251376950 for Purification and antibody-free ex-vivo expansion of peripheral blood-derived autologous natural killer cells: A Malaysian case series on patient safety and tolerability by Ramesh Thevendran, Manickam Ravichandran and Seng Kong Tham in SAGE Open Medical Case Reports
Footnotes
Ethical considerations
This case series was registered and approved by the National Medical Research Register (NMRR ID-25-00333-MUM), Ministry of Health (MOH) Malaysia. Data were extracted in accordance with the ethical principles of the Declaration of Helsinki and Malaysian Good Clinical Practice. Ethics is exempted by the Malaysian Medical Research and Ethics Committee (MREC) under section 6.9, subsection D “Case reports with informed consent from patients” by the Malaysian National Institutes of Health (NIH) Guidelines for Conducting Research under the MOH Malaysia. The CARE checklist is also attached as supplementary files.
Consent to publication
Written informed consent was obtained from all nine patients for publication of this case report.
Author contributions
R.T.: conceptualization and original drafting. M.R.: review and editing. S.K.T.: review, editing, and funding.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was sponsored by ALPS Global Holding Berhad.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
