Abstract
Nature killer cell therapy has shown strong efficacy in the field of oncology in recent years and has been applied to patients with metastases with the aim of improving the prognosis of advanced gastric cancer. A 59-year-old male with gastric adenocarcinoma with pancreatic metastasis (T4N0M1) who underwent radical surgery for gastric cancer with tumor metastasis was treated with oxaliplatin and tegafur combined with cellular reinfusion in stages. Computed tomograpy scan and serum tumor markers were monitored continuously after the treatment course. After five courses of combined treatment, the patient was in disease control with no significant side effects. At the last follow-up, the alpha fetoprotein had returned to its normal value with a poor display of low-density shadows in the body of the pancreas. Pancreatic cancer is insidious in origin and has a high mortality rate. The report provides clinical evidence for cell therapy of pancreatic metastatic cancer with improved quality of life.
Introduction
Gastric cancer (GC) is a common malignant tumor with high morbidity and mortality, and its characteristics include rapid progression, high metastasis rate, and aggressiveness. The early symptoms of GC are not obvious and not easy to detect, and most patients are already in advanced stage when diagnosed, so the cure rate and survival rate are not high. Currently, the treatment of GC is still based on traditional surgery and chemoradiotherapy,1,2 but conventional chemotherapy drugs are harmful to the body, usually accompanied by various adverse reactions, which reduce the patients’ quality of life. In recent years, new treatment modalities, for example, targeted therapy and immunotherapy, have emerged and gradually been widely used in clinical practice.3–6
Immunotherapy mainly works on the immune system of patients to achieve anti-tumor effects. NK cells originate from CD34+ hematopoietic stem cells in the bone marrow, which are widely distributed in the peripheral blood, liver, spleen, and other tissues and participate in the first line of defense of the human immune system. Activated and amplified NK cells can be obtained from different sources, including NK cell lines, primary NK cells, umbilical cord blood (UCB) NK cells, and induced pluripotent stem cell-derived NK cells. 7 NK cells have multiple pathways for killing tumors. One of the pathways for NK cells to kill tumor cells is that NK cells can release perforins and granzymes, which induce apoptosis in target cells. The killing effect of NK cells is also by exerting antibody-dependent cell-mediated toxicity and expressing of tumor necrosis factor-related apoptosis-induced ligand family and Fac ligand to induce cell apoptosis. Another pathway is the secretion of cytokines and chemokines as the regulator of the immune system.8–10 The advantages of UCB-NK cells include broad UCB sources, easy collection, and high percentage of NK cell progenitor cells. UCB-NK cells are more abundant, younger, more proliferative, and more efficient at targeting cancer cells. In view of this, we used mononuclear cells extracted from cord blood in vitro to generate cord blood-derived NK cells with high cell purity, high amplification ratio, and high cytotoxicity (Figure 1). In short, MNCs (between 1 × 106 and 3 × 106 per mL) were cultured in a 175 cm2 flask with 50 mL X-VIVO 15 (Lonza) supplemented with 5% auto-plasma, 1000 U/mL IL-2 (Beijing Four Rings Bio-Pharmaceuticals Co., Ltd, China) and 1 mL laboratory self-made activating factor in a 37°, 5% CO2 incubator. The cell density was checked and adjusted by an appropriate amount of new medium every 2–3 days. For infusions, CD56+CD3−UCB-NK cells were used at the end of the culture process with >90% purity, what was typically achieved within 14 days.

Dot plots from one representative experiment depicting NK cell (CD3-CD56+) purity.
For patients with GC who cannot undergo radical surgery or have metastasis, NK cells can be used to participate in the anti-tumor immune response, which are involved in the innate immune response and have the natural anti-tumor ability to inhibit the growth and proliferation of tumor cells or kill tumor cells directly.11,12 NK cells have significant effects in inhibiting the occurrence, progression, and metastasis of GC,13,14 so it is a potential treatment with the characteristics of safe and effectiveness.
Case
This case report is reported with the written informed consent of the patient. A 59-year-old male, who complained of obstructive sensation in eating with erratic reflux for 2 months. Under gastroscope examination, a protrusive lesion extending downwards into the gastric cavity can be seen starting from 38 cm away from the incisor. Then, he underwent surgery on January 11, 2020 (Figure 2, V0). During the operation, lymph nodes with a diameter of about 1cm can be seen around the left gastric artery and vein. A tumor can be seen at the position of the gastric body, fundus, and cardia, which has not penetrated the serosa and is approximately 6.0 × 6.0 × 4.0 cm in size. Another tumor with a diameter of 3 cm invaded the tail of the pancreas, which is considered as a metastatic cancer and failed to be removed (Figure 3(a) and (b)). The postoperative pathology tests resulted in the diagnosis of middle differentiated gastric adenocarcinoma, and the mass was 5 × 4 × 1 cm in size. Immunohistochemistry analysis showed that the tumor cells were positive for CK8/18, MLHI, MSH2, mSH6, PMS2, and Ki67 labeling index was 40%.
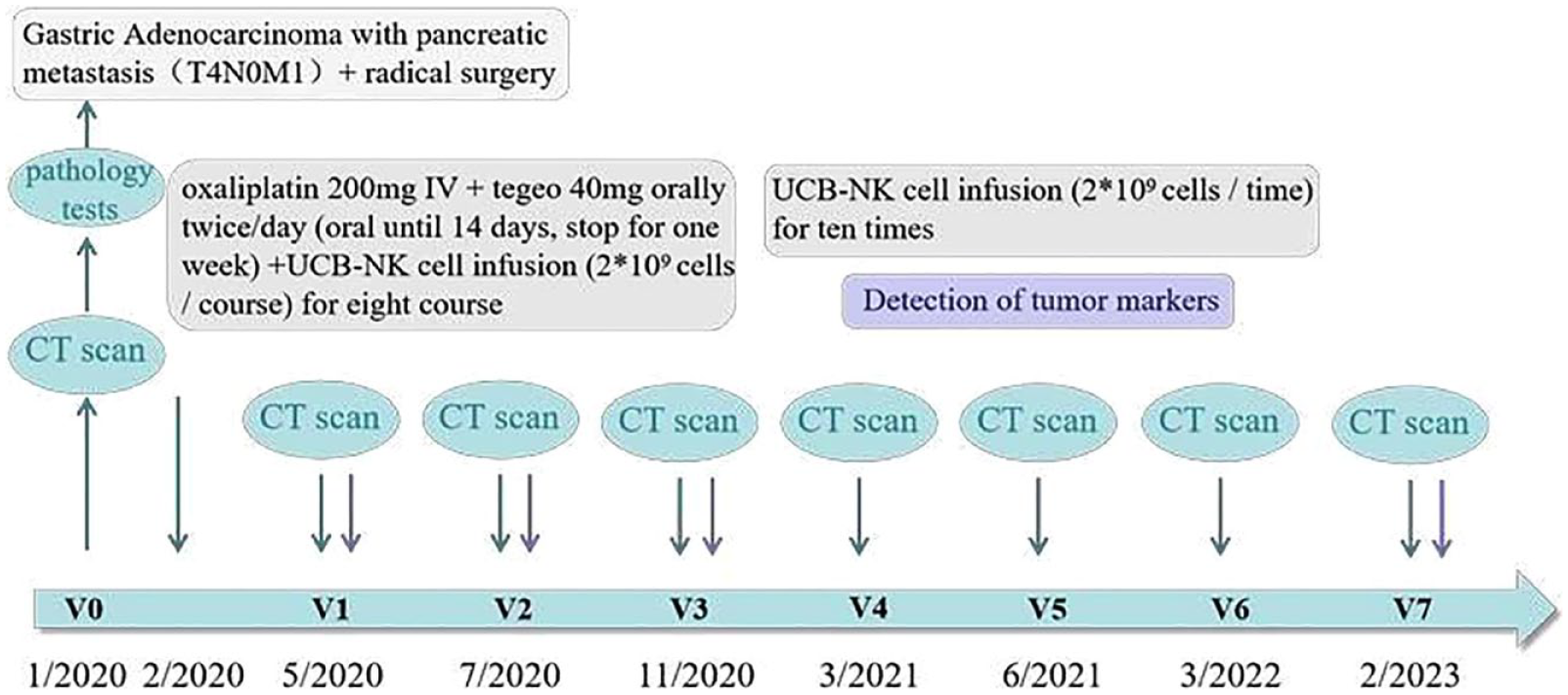
Schematic diagram of clinical history, therapy, and visits of a patient diagnosed with confirmed gastric cancer (stage IV, cT 4, cN 0, cM 1) on January 3, 2020 (V0). After four courses of combining chemotherapy with NK cell infusion (V1), computed tomography (CT) scan showed that the inflammation was slightly better than before, and the initial treatment was effective. After 2 years of combination therapy (February/2020–March/2022), no tumor progression was seen on the CT scan. Notably, at the most recent follow-up (V7), the CT scan showed that the pancreatic tail metastasis was gradually shrinking or even disappearing, and the value of tumor marker alpha fetoprotein has also dropped to the standard range.

(a) Low density shadow is visible in the body of the pancreas by computed tomography (CT) scan before surgery (January 4, 2020); (b) Low density shadow in the body of the pancreas has no significant changes compared to before by CT scan after surgery (January 17, 2020); (c) Low density shadow in the body of the pancreas has no significant changes compared to before by CT scan after three courses of treatment (November 17, 2020); (d) Low density shadow in the body of the pancreas is unclear by CT scan at the last follow-up visit (February 6, 2023).
The patient was treated with chemotherapy combined with cellular immunotherapy from February 2020 to April 2020 (Figure 2, V0–V1). The combination chemotherapy drugs chosen in this case were oxaliplatin and tegafur. Oxaliplatin is a platinum-based antitumor drug with lower adverse effects and wider drug activity. Tegafur is a pyrimidine drug, which can pharmacologically inhibit the activity of orotate phosphoribosyl transferase in the gastrointestinal mucosa and effectively prevent the phosphorylation of 5-fluorouracil, which has a significant effect on reducing the adverse effects on patients. His detailed first course of treatment included oxaliplatin 200 mg IV + tegafur 40 mg orally twice/day (oral until 14 days, stop for 1 week) + NK cell infusion (2 × 109 cells/course) (Figure 2). After the first four courses of combined treatment, the linear hyper-density shadow in the lower lobes of both lungs and density shadow of liquid in both sides of the chest cavity decreased compared to before, the increased density of fat spaces in the upper abdomen was relieved after treatment, and a small amount of fluid in the abdominal and pelvic cavities was absorbed, so the initial treatment was considered effective. The fifth course of combination therapy is on May 15, 2020 (Figure 2, V1), with the same chemotherapy regimen and NK cell infusion as the first four courses of treatment. No significant changes in computed tomography (CT) were observed at the end of the seventh course of treatment. The patient continued with the eighth course of combination therapy from July 14, 2020 (Figure 2, V2) and the treatment remained unchanged. After a period of interruption, during the patient’s regular follow-up, the patient was in a stable condition and no significant changes in CT were observed (Figure 3(c)); however, the tumor marker alpha fetoprotein (AFP) is still very high (29.63 µg/ml) (Table 1 and Figure 4), so another cycle of single tegafur (40 mg orally twice/day, oral until 14 days, stop for 1 week) treatment was given. After the end of this course of treatment, the patient stopped chemotherapy and continued with the NK cell infusion (2 × 109 cells/time, 5 times/year) from January 12, 2021 to September 23, 2022 (Figure 2, V4–V7). At his most recent check, the AFP had returned to normal value (3.84 µg/ml) with poor display of low-density shadows in the body of the pancreas (Table 1, Figure 3(d) and Figure 4).
Changes in tumor markers of patient in this case during chemotherapy combined with cellular immunotherapy.


A line chart illustrating the Table 1 biomarkers change.
Discussion
The small number of intratumor stem-like cells known as cancer stem cells (CSCs) is what causes cancer cells to grow, recurrence, and metastasize, as well as the failure of cytoreductive therapies to treat them. 15 CSCs differ from their mature progeny in numerous ways with the ability to initiate tumors and mediate recurrence following treatment. The increase in NK cell cytotoxicity is a result of up-regulating NKG2D ligands in CSCs such as MICA and MICB following chemotherapy and radiation therapy treatments. Tallerico et al. found that in colon cancer and glioblastoma, NK cells can kill CSCs that are resistant to all standard anti-cancer therapies.16–18
However, in clinical practice, surgical treatment is often the first choice. Surgical operation causes suppression of NK cells and impairment of their effector functions, which is particularly important for cancer patients who have had solid tumors removed, as surgery has been linked to the development of metastasis and cancer recurrence after surgery. On the other hand, it has been shown that the expression of NK cell activating receptors such as NKG2D and NKp30 is lower and the expression of NK inhibitory receptors such as NKG2A and CD158a is higher in GC patients with larger tumor size, lower differentiation, more advanced stage, lymph node metastasis, and deeper infiltration, suggesting that the higher the malignancy of the tumor, the weaker the activity and immune monitoring function of NK cells.19–22 Therefore, tumor patients frequently need donor NK cells, which can exercise normal immune monitoring function to prevent metastasis and recurrence.
In this case, the patient had significant pancreatic metastasis and failed to remove. Moreover, the tumor marker AFP remained high after radical gastrectomy and postoperative chemotherapy. The fact that metastasis is the most fatal characteristic of malignant tumors, causing over 90% of tumor-related mortality, and distant organ or tissue metastasis can be a sign of poor prognosis in patients with GC can be taken into account. Therefore, in the initial eight treatment courses, we adopted chemotherapy combined with NK cell therapy to more effectively and thoroughly eliminate tumor cells. Considering that NK cells have the function of preventing recurrence and metastasis, and in order to further consolidate the therapeutic effect, we have given the patient another ten courses of NK cell therapy. Fortunately, at his most recent check, the AFP had returned to normal value with poor display of low-density shadows in the body of the pancreas. The patient was observed to be in a very good condition with a long survival period and high quality of life.
Conclusion
In conclusion, our findings have interesting implications to develop therapeutic strategies for preventing recurrence and eliminating metastatic lesions after radical gastrectomy. The patient remains disease-free with a follow up of over 3 years. Moreover, combined with CT scan and the detection of tumor markers, the patient’s condition is getting better with the treatment of NK cells. Consequently, post-operative combined chemotherapy and NK cell immunotherapy are effective and safe, and we anticipate more effective treatment for these patients.
Footnotes
Acknowledgements
Thanks are due to Yuanyuan Jin for assistance with the manuscript and valuable discussion.
Author contributions
Yue Y.T. performed the data analyses and wrote the manuscript; Zi M.M. collected and analyzed the data; Feng J.N. searched and organized literature; Wang W.B. graphed the data; Ren Z.Q., Wu C.T., and Yang Z.Y. contributed to the conception of the study and helped perform the analysis with constructive discussions. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
