Abstract
Available data regarding iatrogenic vascular trauma in the pediatric population mainly comprise case reports and series, with management protocols not firmly established. This study aims to report seven pediatric cases of iatrogenic vascular trauma in the extremities, presenting as arteriovenous fistula and pseudoaneurysm, along with their surgical management. We further reviewed the literature and discussed other possible management approaches for arteriovenous fistula and pseudoaneurysm in this specific population. We reviewed the cases of seven pediatric patients (six male and one female) with iatrogenic vascular trauma presenting as arteriovenous fistula and pseudoaneurysm in their extremities. Four patients (57%) were admitted to the intensive care unit due to their severe medical condition before developing these complications. Arterial injury during venous access accounted for five cases (71%). Two patients developed arteriovenous fistula after cardiac catheterization through the common femoral artery for their underlying tetralogy of Fallot. All patients underwent surgical repair. While arteriovenous fistula and pseudoaneurysm are infrequent, their occurrence is on the rise, primarily due to the more frequent use of venous access and catheterization procedures. Surgical approaches include ligation and lateral arteriorrhaphy/venorrhaphy, or resection with arterial end-to-end anastomosis. Despite the growing preference for endovascular treatment in the adult population, such as stent placement or coil embolization, pediatric patients require meticulous attention to the possible risk of stent migration, chronic ischemia, and radiation exposure. Based on our experience, surgical management has generally provided a satisfactory treatment response and recovery. However, this needs to be further evaluated in larger, prospective studies.
Keywords
Introduction
In the pediatric population, the most frequent cause of vascular trauma is iatrogenic, accounting for up to 50% of the cases. These injuries usually occur in procedures such as peripheral catheter insertion, sampling for blood gas and acid-base determinations, hemodynamic monitoring, and diagnostic and therapeutic endovascular approaches like cardiac catheterization. Since these procedures require arterial puncture, they carry a potential risk of complications, including arteriovenous fistulas (AVFs) and pseudoaneurysms (PSAs). AVF and PSA may develop months after iatrogenic trauma, and mimic other vascular and cutaneous pathologies. 1 These factors, along with the presence of other serious comorbidities, can collectively contribute to delays in the diagnosis and management of these complications. Given the rarity of AVF and PSA in the pediatric population, available data mainly comprise case reports and series, with the largest number of AVF cases documented as five. 2 Consequently, with management protocols not firmly established in children, diagnosis, and treatment primarily rely on extrapolations of the data obtained from adult patients.
Endovascular intervention and surgical approach are the two main treatment options for chronic cases of AVF and PSA without spontaneous resolution. Despite the growing preference for endovascular treatment in the adult population, such as stent placement or coil embolization, pediatric patients require meticulous attention to the possible risk of stent migration, chronic ischemia, and radiation exposure. On the other hand, surgical treatment is further technically challenging due to the smaller size of vessels, arterial spasms, and anatomic variations in pediatric patients. 3
This study aims to report seven pediatric cases of iatrogenic vascular trauma presented as AVF and PSA and their management at a university-based tertiary referral hospital. Additionally, it aims to scrutinize the alternatives of the surgical approach by reviewing the current literature.
Patients and methods
After obtaining approval from the hospital's Institutional Review Board, written informed consent was obtained from the parents or guardians of all consecutive pediatric patients who presented with AVF or PSA following iatrogenic vascular trauma. Retrospectively, office charts from 2018 to 2024 were reviewed for this series of cases. Follow-up data were collected through a systematic review of clinic records, and, in selected cases, supplemented by telephone interviews with the attending pediatricians. Patients underwent regular physical examinations by both a vascular surgeon and a pediatrician at intervals of 2 to 3 months. This article is in line with SCARE guideline. 4
Presentation of cases
Seven pediatric patients (six male and one female) were diagnosed as having AVF or PSA (Table 1) in their extremities. The ages of these patients ranged from 1 month to 7 years (mean: 2.8 years), with weights varying from 4.5 kg to 22 kg (mean: 13 kg). Initial symptoms and signs included the detection of a mass and bulging (Figure 1), thrill, bruit, edema, and varicosity (Figure 2). One patient exhibited limb overgrowth on the same side as AVF (Figure 3).
Mechanism of injury, underlying disease, and presentation of the patients.
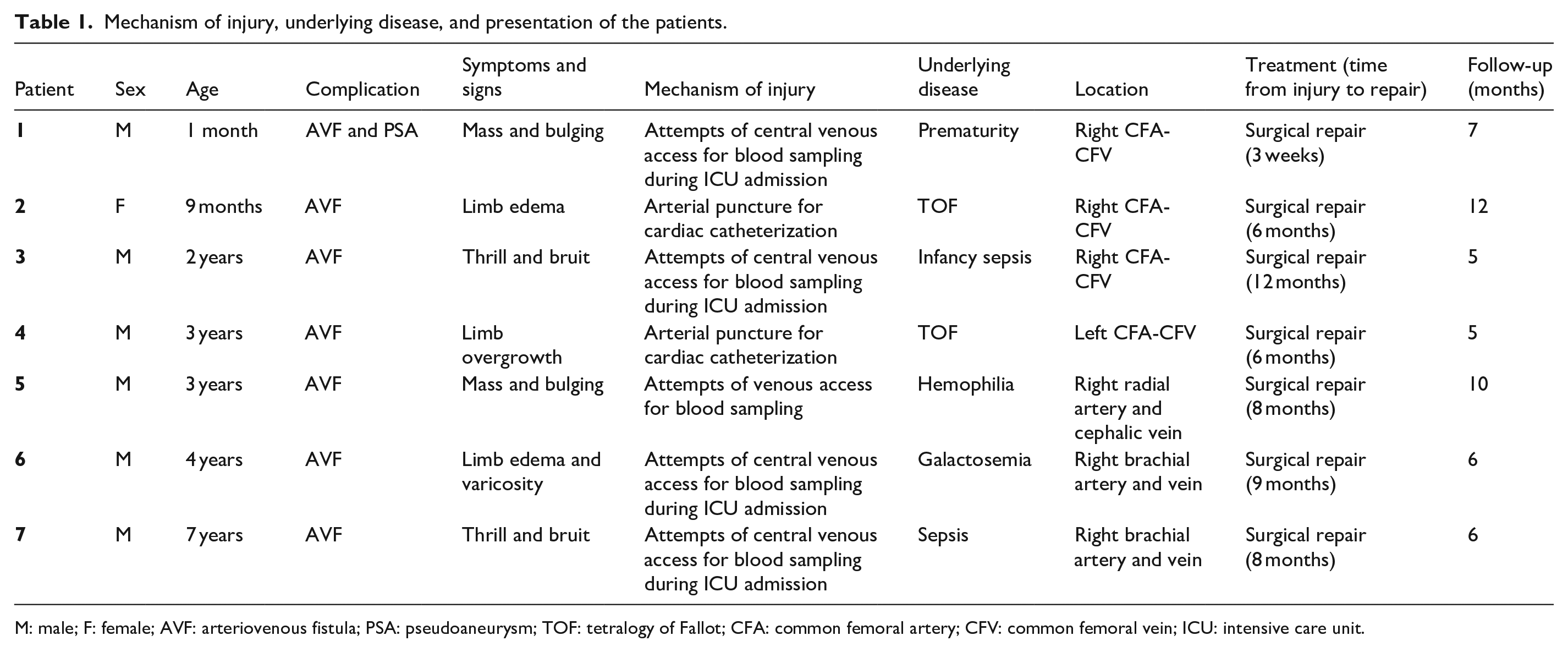
M: male; F: female; AVF: arteriovenous fistula; PSA: pseudoaneurysm; TOF: tetralogy of Fallot; CFA: common femoral artery; CFV: common femoral vein; ICU: intensive care unit.

Preoperative image of a radial-cephalic AVF presented as mass and bulging in a 3-year-old patient.

Right brachial artery-brachial vein AVF shown with arrows in a 4-year-old patient. (a) Preoperative varicosity and dilation of superficial veins in the upper extremity. (b) CT angiography showing AVF (posterior view).
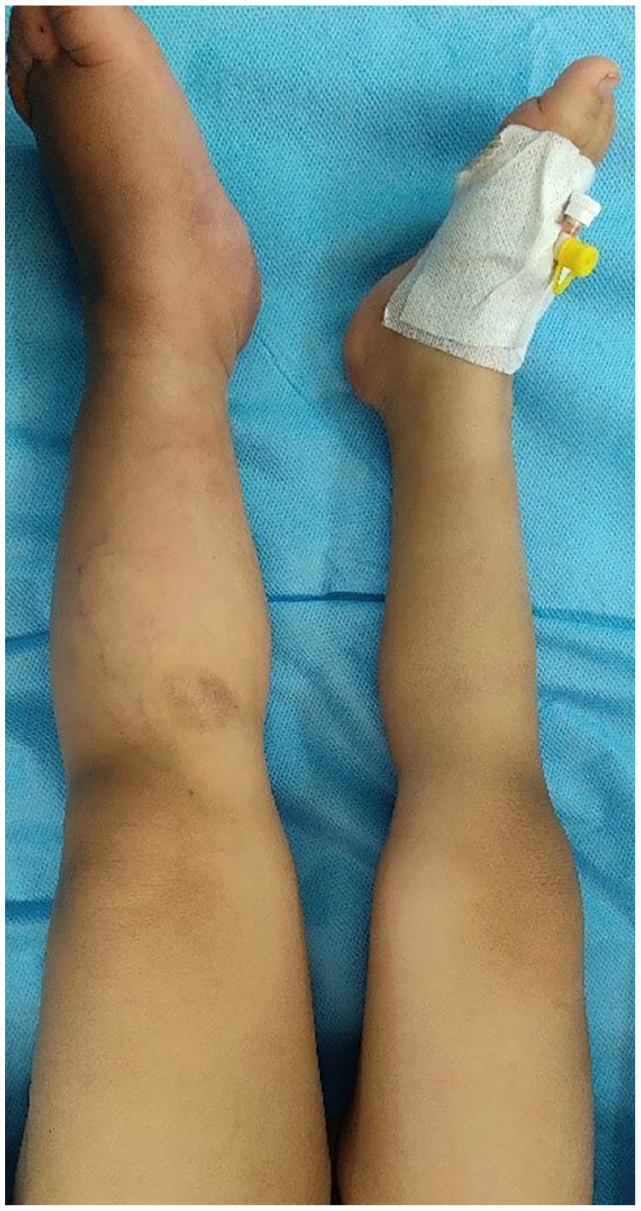
Limb length discrepancy in a 3-year-old patient due to left CFA-CFV AVF.
The most frequent site of complication was the common femoral artery and vein (57%). Four patients (57%) were admitted to the ICU due to their severe medical condition before developing AVF or PSA. Arterial injury during venous access accounted for five of our cases (71%). Two patients developed AVF after cardiac catheterization through the common femoral artery for their underlying tetralogy of Fallot. All patients underwent surgical repair (resection with arterial end-to-end anastomosis) performed by a vascular surgeon (Figure 4). The time from injury to surgical repair varied widely among patients, with a median of 8 months (range: 3 weeks to 12 months). Postoperative follow-up (median duration of 6 months, range 5–12 months) was uneventful, with no complications detected on physical examination. Although Color Doppler ultrasonography was planned for any abnormal clinical findings, no such findings were observed during the follow-up period, and imaging was therefore not necessary.
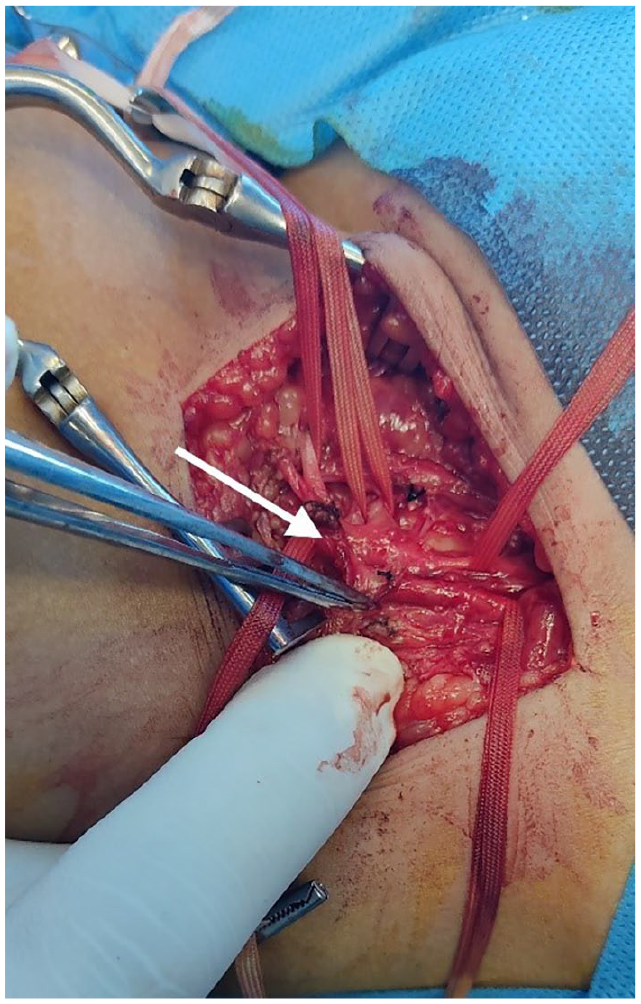
Intraoperative image of surgical treatment of right CFA-CFV AVF shown with arrow.
Discussion
Iatrogenic causes account for almost half of pediatric vascular injuries. The incidence of these injuries declines with age, with neonates being at the highest risk. 5 Iatrogenic vascular trauma is the leading cause of AVFs and PSAs in pediatric cases. To the best of our knowledge, this article is the largest series reporting on AVFs and PSAs in the pediatric population. While these complications are infrequent, their occurrence is on the rise, primarily due to the more frequent use of venous access and catheterization procedures. In our series, these procedures accounted for 71% and 29% of complications, respectively. Almost half of our cases had been admitted to the ICU before developing AVF and PSA, underscoring the severe underlying medical conditions that can overshadow these complications and contribute to delays in diagnosis and management.
Among pediatric patients, AVFs occur at a rate of approximately 0.3%, and the overall incidence of PSA is below 0.01%.2,6 Our experience similarly reflects a much higher occurrence of AVFs than PSA, with seven AVFs and just one PSA in our cohort of patients. These rates are expected to be higher if an ultrasound (US) test is actively performed after catheterization. Generally, complications occur more frequently in pediatric patients and management success rates are lower compared to adults. 7 The incidence of these complications rises with various factors like the prolonged duration of catheter use, the higher number of lumens in the catheter, the increased catheter-to-vein diameter ratio, repeated attempts, younger age, lower weight of the patient, and hemodynamic compromise.8–11 Additionally, cardiac catheterization, particularly for therapeutic purposes, can cause these complications at the access site due to the use of larger caliber catheters.12,13
AVFs form following the initial puncture of an artery or vein, which results in the formation of a hematoma. Adhesion between the artery and vein occurs during the healing process and the fibrosis of the hematoma. Further punctures can then lead to the formation of a fistula. AVFs can present asymptomatically when small or intramural, or with symptoms and signs such as limb edema (two of our patients), claudication, pain from nerve compression, dilated superficial veins and varicosity (one patient), palpable thrill, or detectable bruit (two of our patients). Even high-output cardiac failure has been reported in rare cases. 2 Doppler US is the preferred choice for imaging evaluation, providing insights into shunt volume and, if tolerated by young patients, serving as a means of treatment through US-guided compression.
Asymptomatic AVFs can be managed with careful observation as the first choice, given that up to one-third of them may spontaneously seal within a year. Treatment is indicated in symptomatic AVFs, progressive enlargement observed in US surveillance, and unresolved asymptomatic AVFs after 6 to 12 months. The primary treatment for these AVFs in the pediatric population is surgical, including fistula ligation and lateral arteriorrhaphy/venorrhaphy, or resection with arterial end-to-end anastomosis. 5
Closing an AVF can redirect blood flow into smaller, hidden AVFs nearby. Thus, as demonstrated by Papes et al., 2 intraoperative exploration of vessels for thrill and Doppler US examination is warranted for detecting these AVFs. If symptoms of varicose veins persist after surgical treatment, patients may be offered phlebectomy and/or compression therapy.
Endovascular options for AVF treatment, such as covered stent placement and embolization with coils or N-butyl-cyanoacrylate, are not well-studied in young patients. Stent placement necessitates long-term anticoagulation. Additionally, in children with growing limbs, the stent diameter may not adequately maintain proper blood flow, potentially resulting in limb length discrepancy due to chronic ischemia or thrombosis. 14 Moreover, when AVFs are positioned in proximity to joints like the common femoral artery (as in almost half of our patient cohort), movement can result in stent deformation and breakdown. Likewise, AVF embolization with glue or coil in pediatric patients can lead to large vessel thrombosis due to glue leakage or coil displacement. Furthermore, in pediatric patients with minimal soft tissue coverage, stents or coils may remain palpable, increasing the risk of skin breakdown due to pressure necrosis. As proposed by Pagès et al., 15 infants, especially given their rapid limb growth, are not optimal candidates for endovascular treatment due to the risks of stent migration and high radiation exposure associated with these procedures.
Another potential complication of iatrogenic trauma is PSA, which we encountered in one of our cases. PSA occurs when there is a defect in the arterial wall, allowing extra-luminal blood flow, which over time can lead to thrombus formation within the PSA cavity. Subsequently, a fibrin wall develops, contained by the surrounding adventitial and perivascular tissues. Iatrogenic vascular trauma during diagnostic and interventional procedures accounts for up to 80% of PSA incidences. 16
The presentation of PSA can range from asymptomatic to the manifestation of a pulsatile mass (patient number one in Table 1) accompanied by pain and compression of nearby structures. Despite various imaging modalities available for diagnosing PSAs, they are frequently identified by the characteristic “yin-yang” sign and “to-and-fro” flow pattern on color Doppler US, resulting from turbulent blood flow during systole and diastole in their communicating channel. 17
The management of PSAs is contingent on their size and duration (acute or chronic), ranging from observation and compression to percutaneous US-guided thrombin injection, endovascular approaches, and open surgical methods. However, in the pediatric population, the management of PSAs is not well established. Acute cases (<4 weeks) can be managed with a conservative approach including observation or compression with US or bandage, if tolerated. Small PSAs, measuring below 1 cm, have been observed to spontaneously resolve. 5 For larger PSAs, compression with US guidance and occasional thrombin injection offers a noninvasive treatment option, with success rates reaching up to 88%. 18 Attention should be given to the early diagnosis and, especially, follow-up of patients treated conservatively, as there is a potential risk of tissue loss due to chronic ischemia. 19
For chronic cases of PSA (>4 weeks), endovascular techniques or surgical approaches may be considered. In cases where a PSA has a wide-based neck or when nonoperative management has failed, surgical exploration of the vessel may be warranted. Approaches may include primary repair, resection, and subsequent primary end-to-end anastomosis, as well as the use of an interposition vein graft or patch angioplasty. 5
As Aspalter et al. 19 reported, preoperative arteriography is usually not required. Additionally, they suggested that longitudinal arteriotomy and topical use of papaverine can relieve arterial spasms. In cases where the arterial defect is too extensive to permit a direct end-to-end anastomosis, the consideration of interposing an autologous vein graft, such as the basilic vein, accessory saphenous vein, or distal greater saphenous vein, is justified. Then a permeability test is performed to validate the anastomosis. This approach eliminates the risk of narrowing the artery through the suture line or encountering a stretched suture that might fail or thrombose immediately.15,19
Surgical treatment may improve arterial patency and reduce the risk of ischemia-related complications, such as tissue loss and potential limb length discrepancy. However, surgical treatment in the pediatric population carries inherent risks and unique challenges, primarily due to the small size of vessels and their tendency to spasm during manipulation. Additionally, limited vessel length and fragility can complicate dissection and anastomosis. Achieving durable repairs in growing children is technically demanding, as the need for tension-free suturing must be balanced against ongoing somatic growth. 20
Conclusion
The rarity of AVFs and PSAs in the pediatric population makes it challenging to conduct high-level research, such as clinical trials, to compare different treatment options, including surgical and endovascular approaches. Based on our experience, surgical management has generally provided a satisfactory treatment response and recovery. However, this needs to be further evaluated in larger, prospective studies.
Footnotes
Ethical considerations
Ethical approval for this study was waived by IRB TUMS because our institution does not require ethical approval for reporting individual cases or case series. Not applicable due to the retrospective design of the study.
Consent to participate
Written informed consent was obtained from the patient's parents/legal guardian for participation in this study.
Consent for publication
Written informed consent was obtained from the patient's parents/legal guardian for publication and any accompanying images.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are not openly accessible due to sensitivity concerns but can be obtained from the corresponding author upon reasonable request.
Trial registration
Not applicable.
