Abstract
Totally implantable venous access ports are widely used for long-term intravenous therapy in oncology patients. However, catheter malpositioning may result in catheter-related right atrial thrombosis, a rare but potentially life-threatening complication. We report three breast cancer patients who developed catheter-related right atrial thrombosis due to deeply located catheter tips within the mid-right atrium. All patients were successfully treated with systemic thrombolysis combined with anticoagulation, resulting in complete thrombus resolution. These cases underscore the preventable nature of catheter-related right atrial thrombosis through accurate intraoperative catheter tip localization using intracavitary electrocardiography or echocardiography and highlight the limitations of relying solely on postprocedural chest radiographs. Early recognition, precise localization, and individualized treatment are essential, but prevention remains the cornerstone of safe totally implantable venous access port utilization.
Keywords
Introduction
Central venous catheters (CVCs), including peripherally inserted central catheters, non-tunneled and tunneled CVCs, and totally implantable venous access ports (TIVAPs), are widely employed in oncology patients to ensure reliable vascular access for chemotherapy, parenteral nutrition, and blood sampling. However, central venous catheterization carries risks, among which catheter-related thrombosis remains a significant concern, particularly with long-term catheter use.1,2
Catheter-related right atrial thrombosis (CRAT) is an underrecognized yet potentially fatal complication, particularly in oncology and dialysis patients. Retrospective studies report mortality rates of ~20%, with overall death rates ranging from 18% to 21%.3,4 The incidence varies widely due to differences in detection methods and patient populations, estimated between 2% and 20% by routine imaging, and up to 30% in autopsy series. 5 These data underscore the urgency of improving early detection and standardizing management strategies for CRAT. Importantly, the present report aims to contribute to this understanding by describing three breast cancer cases of iatrogenic CRAT and discussing diagnostic and therapeutic challenges.
Multiple factors contribute to the development of CRAT, but malposition of the catheter tip—especially deep placement within the right atrium—has emerged as a principal risk factor.6,7 According to current guidelines and expert consensus, optimal catheter tip positioning at the cavoatrial junction (CAJ) is critical to minimize thrombogenic risk. 8 Intraoperative tools such as intracavitary electrocardiography (IC-ECG), fluoroscopy, or echocardiographic guidance are strongly recommended to ensure accurate placement.9–11 In contrast, relying solely on postprocedural chest radiographs has been shown to be inaccurate and insufficient for safe localization. 12
Although anticoagulation remains the mainstay of CRAT management, particularly in asymptomatic cases, 5 the role of thrombolytic therapy is debated and typically reserved for symptomatic or hemodynamically unstable cases. 13 Device repositioning or removal is indicated when tip malposition or thrombus adherence is confirmed. In this study, we report three breast cancer cases of iatrogenic CRAT resulting from malpositioned TIVAP tips. Through case analysis and literature review, we aim to illustrate the diagnostic and therapeutic challenges of CRAT, emphasize its preventable nature, and advocate for adherence to evidence-based catheter placement protocols.
Treatment protocol
The thrombus management strategy combined systemic thrombolysis and anticoagulation. Thrombolysis was performed using urokinase at a daily dose of 300,000–500,000 IU administered via a micro-infusion pump. Anticoagulation consisted of low-molecular-weight heparin (dalteparin) 5000 IU given subcutaneously twice daily. Treatment was continued for 7–9 days, depending on the thrombus size and patient response. The primary goals were thrombus resolution, restoration of catheter function, and prevention of recurrence. Coagulation parameters, including activated partial thromboplastin time and fibrinogen levels, were monitored throughout treatment to guide dosing and assess safety.
Case presentation
Case 1
A 27-year-old woman (Stage IIB, T2N1M0, ER+/PR+/HER2−) underwent TIVAP implantation via ultrasound-guided puncture of the right internal jugular vein under local anesthesia. During the procedure, IC-ECG was used for tip localization; however, P-wave changes were not pronounced. Consequently, intraoperative chest radiography was performed to estimate catheter tip position, which was confirmed at the level of the ninth posterior rib, corresponding to the mid-right atrium. Seventy days postimplantation, transthoracic echocardiography performed incidentally during pre-chemotherapy cardiac assessment revealed a 3.4 × 2.5 cm thrombus adherent to the catheter tip. The patient remained asymptomatic. Comprehensive laboratory evaluation, including coagulation studies, D-dimer levels, and thrombophilia screening, revealed no abnormalities. Combined systemic thrombolysis and anticoagulation achieved complete thrombus resolution within 9 days without bleeding complications. To prevent recurrence, the TIVAP was removed, and chemotherapy was continued via an alternative central access. The patient remained free of thrombus recurrence during follow-up (Figures 1–3).

Chest X-rays showed the catheter tip is located at the level of the ninth posterior rib.

Transthoracic echocardiogram confirmed a large immobile mass in the right atrium, the size of mass was 3.4 × 2.5 cm.

Transthoracic echocardiogram confirmed thrombus resolution in the right atrium after the ninth day of thrombolytic therapy.
Case 2
A 36-year-old woman (Stage IIIC, T2N3M0, ER+/PR+/HER2−) underwent TIVAP placement via ultrasound-guided right internal jugular vein cannulation for systemic therapy. The catheter tip was initially confirmed by IC-ECG and postoperative chest radiography to be positioned at the level of the eighth posterior rib, corresponding to the lower right atrium. Forty-five days after implantation, transthoracic echocardiography performed during pre-chemotherapy cardiac function assessment incidentally detected a 1.4 × 1.3 cm thrombus adherent to the catheter tip, accompanied by catheter dysfunction. The patient remained asymptomatic throughout this period. Laboratory evaluation, including coagulation parameters, D-dimer measurement, and thrombophilia screening, revealed no significant abnormalities. A combined approach of systemic anticoagulation and thrombolytic therapy was initiated, resulting in complete resolution of the thrombus within 7 days. Under fluoroscopic guidance and contrast visualization, the catheter tip was repositioned to the CAJ to prevent recurrence, and chemotherapy proceeded without further complications (Figures 4–7). No thrombus recurrence was observed during follow-up.

Chest X-rays showed the catheter tip is located at the level of the eighth posterior rib.

Transthoracic echocardiogram detected a right atrial thrombus measuring 1.4 × 1.3 cm.

Transthoracic echocardiogram confirmed thrombus resolution in the right atrium after the seventh day of thrombolytic therapy.

The chest X-ray shows the adjusted catheter tip is located at the seventh posterior rib.
Case 3
A 44-year-old woman (Stage IIB, ER+/PR+/HER2+ FISH−) had previously undergone TIVAP insertion via ultrasound-guided right internal jugular vein access for adjuvant chemotherapy. The catheter tip was verified by IC-ECG and confirmed on immediate postoperative chest radiograph to be situated at the level of the eighth posterior rib, corresponding to the CAJ. Eighty-four days after placement, a pre-chemotherapy cardiac evaluation with transthoracic echocardiography unexpectedly identified a 1.6 × 1.3 cm thrombus attached to the catheter tip, along with impaired catheter performance. The patient exhibited no clinical symptoms. Comprehensive laboratory tests, including coagulation profiles, D-dimer levels, and screening for hypercoagulable states, were unremarkable. A treatment regimen combining systemic anticoagulation and thrombolysis achieved complete thrombus resolution within 9 days. Under fluoroscopic imaging guidance with real-time contrast injection, the catheter tip was carefully repositioned to the CAJ to ensure optimal placement and minimize the risk of recurrent thrombus formation, and the patient subsequently completed the planned chemotherapy course without further adverse events (Figures 8–11). The thrombus did not recur during the follow-up period.

Chest X-rays showed the catheter tip is located at the level of the eighth posterior rib.
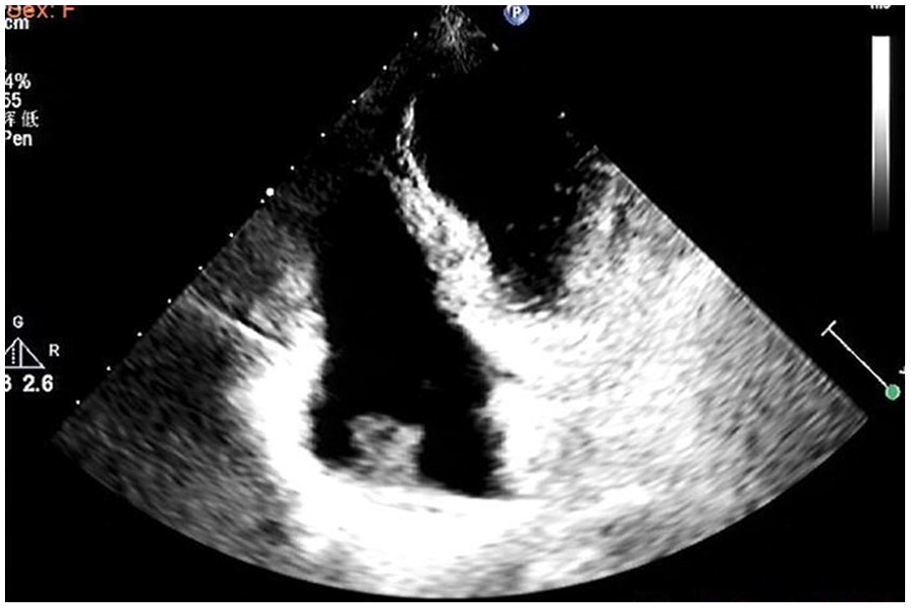
Transthoracic echocardiogram detected a right atrial thrombus measuring 1.6 × 1.3 cm.

Transthoracic echocardiogram confirmed thrombus resolution in the right atrium after the ninth day of thrombolytic therapy.
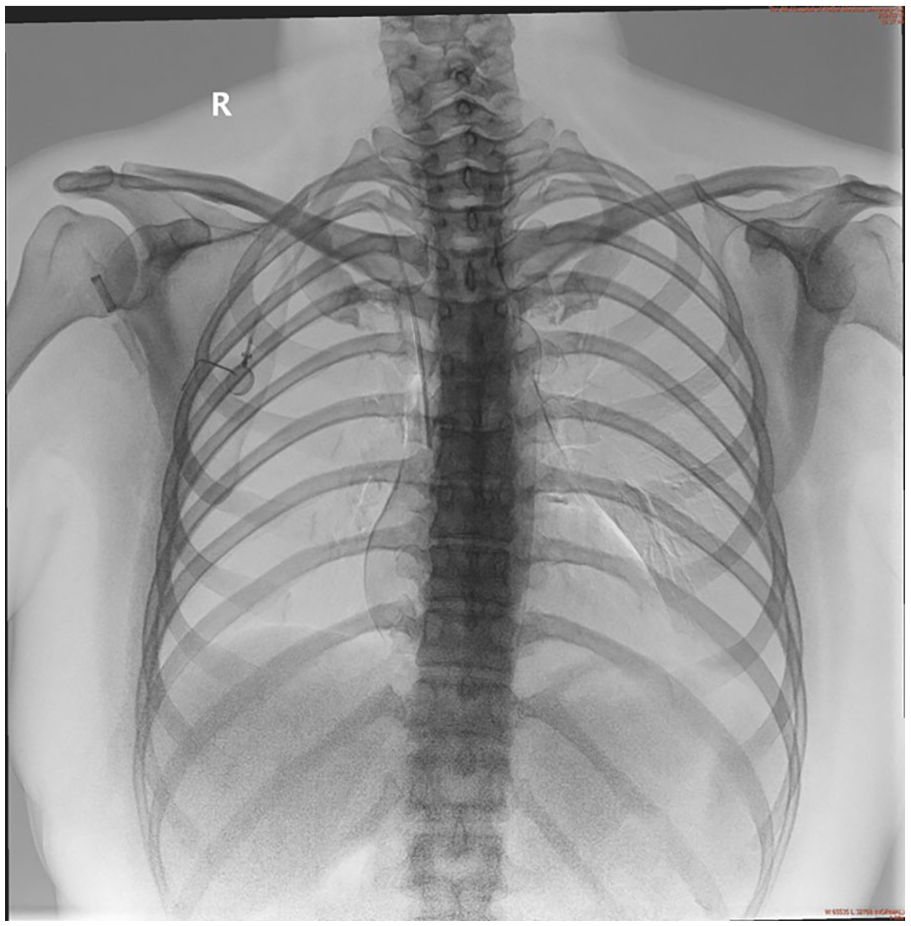
The chest X-ray shows the adjusted catheter tip is located at the upper margin of the eighth posterior rib.
Discussion
CRAT is a potentially preventable but underrecognized complication of central venous access, particularly in oncology patients requiring TIVAPs. Although its pathogenesis is multifactorial—encompassing malignancy-associated hypercoagulability, endothelial injury, and disturbed flow dynamics—the most critical and modifiable factor is catheter tip malposition.14–16
In all three cases, the catheter tips were located abnormally deep within the mid-right atrium, a position strongly associated with endothelial irritation and thrombus formation.17,18 While IC-ECG guidance was employed intraoperatively, the absence of pronounced P-wave changes limited its reliability. Consequently, intraoperative chest radiography was used; however, reliance on radiographs alone, particularly rib landmarks, is now considered inadequate for safe localization.6,7
According to the Infusion Nurses Society and European Society for Medical Oncology, optimal catheter tip placement at the CAJ significantly reduces thrombotic and mechanical complications.8,19 IC-ECG is particularly cost-effective, safe, and accurate, and should be regarded as the standard of care.20,21 Our findings reinforce this recommendation and highlight that CRAT often results from avoidable technical shortcomings.
In line with international guidelines,8,19,22,23 anticoagulation remains the cornerstone of treatment for asymptomatic CRAT, while thrombolysis is generally reserved for large (>2 cm), mobile, or symptomatic thrombi. Our decision to initiate thrombolysis was based on specific clinical considerations: one case involved a thrombus exceeding 2 cm, while the other two exhibited catheter dysfunction likely due to thrombus adherence. In addition, all patients required uninterrupted central venous access for chemotherapy. Although this approach deviates from conservative recommendations, we considered thrombolysis necessary to restore patency and avoid treatment delays. Notably, prior studies reported a thrombolysis success rate of ~64%, 3 whereas all patients in our series achieved complete resolution without bleeding complications. This suggests that carefully selected patients may benefit from early combined therapy. Furthermore, our findings reinforce that accurate intraoperative tip localization, rather than reliance on chest radiographs alone, is critical for prevention—a point not consistently emphasized in earlier reports.
Catheter management is equally crucial. Functional, uninfected catheters can often be preserved with close monitoring and anticoagulation. 24 However, in cases of confirmed malposition or thrombus adherence, repositioning or removal is warranted. In our cohort, one patient required port removal (Case 1), while two underwent tip adjustment and continued chemotherapy, demonstrating the feasibility of image-guided catheter correction.
Study limitations and future directions
Although this case series provides valuable insights, it is limited by the small sample size, the absence of a control group, and the short follow-up duration. Without a comparator arm, it is not possible to draw definitive conclusions regarding the efficacy of thrombolysis versus anticoagulation alone. Additionally, long-term outcomes, recurrence rates, and treatment-related complications, particularly bleeding, could not be comprehensively evaluated. Future research should prioritize prospective, multicenter randomized controlled trials to compare monotherapy with combination therapy, especially in asymptomatic patients. Furthermore, advances in catheter materials and real-time tip tracking technologies may help reduce iatrogenic complications such as CRAT. Despite these limitations, this case series contributes practical insights to inform management strategies and highlights areas for future investigation.
Conclusion
This report highlights the iatrogenic nature of CRAT due to malpositioned TIVAP tips. Although systemic thrombolysis combined with anticoagulation was effective in all patients, this approach remains controversial. Accurate intraoperative tip localization and adherence to evidence-based guidelines are essential to improve patient safety in oncology vascular access.
Footnotes
Acknowledgements
We thank all patients and their families for their cooperation.
Ethical Considerations
The case series was conducted in accordance with the Declaration of Helsinki. Ethics approval was waived for retrospective, anonymized case data.
Consent to Participate
Written informed consent was obtained from all three patients for the publication of this case series, including use of clinical data and images. In cases where a patient was deceased or unable to provide consent, written informed consent was obtained from the legally authorized representative. This complies with COPE guidelines.
Author Contributions
Yanshou Zhang: study conception, case analysis, manuscript drafting. Fangyi Sun: cardiologic evaluation, treatment monitoring, critical revision. Chao Yang, Weina Wang, Shuo Zhang: data collection and imaging support. Xiaolong Li: literature review. Yunjiang Liu: senior supervision, final manuscript approval. All authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by the Key Research Program of Medical Science in Hebei Province (No. 20160666).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
