Abstract
Clot-in-transit is associated with severe pulmonary embolism and higher mortality than acute pulmonary embolism without clot-in-transit. The optimal treatment of clot-in-transit is not established. Multiple treatment options have been described, including anticoagulation alone, systemic thrombolysis, surgical embolectomy and endovascular catheter-based therapies. Clot-in-transit can embolize to the pulmonary circulation in a matter of seconds and be immediately fatal. We describe two cases of clot-in-transit which embolized quickly upon Intensivist’s evaluation and were associated with serious consequences. Management decisions for clot-in-transit should be emergent and based on multidisciplinary discussion of the pulmonary embolism response team.
Introduction
Clot-in-transit (CIT) is a rare but serious manifestation of venous thromboembolism where the thrombus is temporarily lodged in the right heart before entering the pulmonary vasculature. 1 The thrombus could be located in the right atrium, right ventricle (RV), superior vena cava, inferior vena cava, patent foramen ovale (PFO) or septal defect.2–6 CITs are highly mobile serpiginous masses which are free floating or have thin attachments, and can readily embolize into the pulmonary circulation with disastrous consequences. We describe two cases of CIT incidentally diagnosed by bedside point-of-care ultrasonography (POCUS) performed by the Intensivist, and both CITs dislodged quickly into the pulmonary vasculature with serious manifestations.
Case presentation
Case 1
An 89-year-old female with a past medical history of compression deformity of L1 vertebra presented with weakness, dry cough and shortness of breath for 3 weeks. She had difficulty walking due to back pain and was mainly bedbound. She also complained of bilateral leg swelling. She denied any chest pain, palpitations, fever or any other systemic complaints. She was vaccinated against Covid-19 and received a booster dose. She did not have any history of venous thromboembolism or cancer and denied any history of recent prolonged travel.
On arrival to the emergency department (ED), she was found to be hypoxic to SpO2 of 88% on room air, which improved to 94%–96% with 2–3 L/min nasal cannula 02. She was afebrile with initial blood pressure (BP) of 93/51 mm Hg and sinus tachycardia of 120/min. Rest of the physical examination was unremarkable except bilateral pitting pedal edema.
Electrocardiogram (EKG) on admission showed sinus tachycardia and new complete right bundle branch block. Lungs were clear on chest X-ray. Laboratory results were significant for white blood cell count of 15.1 K/μL, hemoglobin 14.8 g/dL, platelet 275 K/μL, INR 1.5, normal partial thromboplastin time (PTT), blood urea nitrogen (BUN) 33 mg/dL, creatinine 1.54 mg/dL (baseline 0.8), troponin 0.12 ng/mL, brain natriuretic peptide of 655 pg/mL and negative Covid-19 result. On venous blood gas analysis, pH was 7.36, PaCO2 35 and lactic acid 6.2. Heparin infusion was started in the ED given strong clinical suspicion for acute pulmonary embolism (APE) and the patient was awaiting computed tomography (CT) angiogram to diagnose APE. Critical care medicine (CCM) service was consulted for intensive care unit (ICU) monitoring. On CCM review, she was alert and oriented, with heart rate (HR) of 112/min and BP of 128/78 mm Hg, and was not in respiratory distress. POCUS revealed extensive deep venous thrombosis (DVT) extending from the left common femoral vein to the left popliteal vein and DVT involving the right popliteal vein. There were multiple, large, mobile and serpiginous CITs in the right atrium, also prolapsing into the RV across the tricuspid valve (Supplemental case 1, video 1). Two minutes later, while still performing POCUS, the whole clot burden moved across the right atrium into the pulmonary vasculature and POCUS did not reveal any clot in the right atrium or RV (Supplemental case 1, video 2). Immediately following that, the patient became progressively more dyspneic with respiratory rate (RR) in the 40s and HR in 140s. She became altered and went into pulseless electrical activity (PEA) cardiac arrest. Cardiopulmonary resuscitation (CPR) was initiated, and the patient was intubated during the cardiac arrest. Alteplase 100 mg was emergently given intravenously during the CPR for massive APE. Repeat multiple POCUS during CPR showed severely dilated, hypokinetic RV (Supplemental case 1, video 3). Return of spontaneous circulation (ROSC) was achieved 5 times with epinephrine pushes but, eventually, ROSC could not be obtained. She was pronounced dead 4 h after presentation to the ED. The cause of death was acute massive pulmonary embolism (PE).
Case 2
An 84-year-old female with a past medical history of hypertension, hyperlipidemia and multiple PEs was admitted with 2 days of history of sharp pleuritic chest pain and shortness of breath. Patient denied any fever, dizziness, leg swelling, recent immobilization or prolonged travel. Patient was switched from apixaban to Coumadin which she stopped taking for 3 months prior to admission due to elevated INR problems. Patient was afebrile and initial vitals on ED arrival were HR 93/min, BP 137/88 mm Hg, RR 17/min and SpO2 of 97% on room air. Patient was Covid-19 negative and was vaccinated against Covid-19. Initial labs on admission were unremarkable except for creatine phosphokinase (CPK) of 266 U/L, troponin I of 0.67 ng/mL and B-type natriuretic peptide (BNP) of 537 pg/mL. EKG showed normal sinus rhythm and inverted T waves in inferolateral leads. Due to the high suspicion of APE, the patient underwent CT with PE protocol which showed acute saddle PE, right heart strain and serpiginous filling defect in the right atrium. POCUS showed severe RV dilatation and CIT prolapsing across the tricuspid valve (Supplemental case 2, video 1). Lower extremity Doppler showed extensive acute DVT in the left common femoral extending down to the popliteal, gastrocnemius and peroneal veins. Patient was started on heparin drip. Interventional radiology was consulted, and the patient was admitted to the ICU for closer monitoring. Upon arrival to the ICU, the patient started coughing, became tachypneic to high 30s and desaturated to 85% on room air. She was placed on 6 L/min nasal cannula oxygen followed by need for high-flow nasal cannula oxygen. Repeat emergent POCUS at bedside did not show right atrial CIT but worsening RV dilatation, McConnell’s sign and severe tricuspid regurgitation (Supplemental case 2, video 2). The clot dislodged to the pulmonary circulation. Endovascular catheter-based therapy was put on hold and the patient was closely watched in the ICU. Her respiratory status slowly stabilized, and BP remained stable without the need for vasopressors. Patient was slowly weaned down on oxygen support and discharged from the ICU in stable condition after 4 days.
Discussion
Our case report shows a series of two cases of highly mobile CIT which embolized to the pulmonary circulation within an hour of evaluation by the critical care team. Case 1 was associated with cardiac arrest and dismal outcome and case 2 was associated with worsening respiratory status. The different outcome between the two cases was likely due to the poor cardiopulmonary reserve of the patient in the first case.
CITs or type A right heart thrombi (RHT) are defined as the highly mobile serpiginous masses embolized from deep veins, temporarily lodged in the right heart and associated with concomitant APE. 1 They are usually free floating or have a thin point of attachment. The exact prevalence is estimated to be around 2%–5% of all PE cases, although, it may be as high as 18% in patients with massive PE.1,7 CIT is associated with more severe PE and higher mortality compared to PE alone.1,7–9 The reported mortality rates of CIT range from 19% to 38%, and if left untreated, as high as 80%–100%.7,10 Garvey and colleagues 8 also found in their PE response team registry that patients with CIT had more heart failure, presence of central venous catheter and hypotension than patients with PE without CIT.
With increasing utilization of the POCUS and transthoracic echocardiography in PE patients, the incidental detection of CIT is increasing.
The treatment of CIT presents a therapeutic dilemma for physicians as the optimal therapy is not well established and no societal recommendations or clear consensus exists till date. Multiple treatment options have been described, including anticoagulation alone, systemic thrombolysis, surgical embolectomy and endovascular catheter-based therapies.1,7,11–17 Table 1 lists these treatment modalities with relative advantages, disadvantages and associated references. All of them can also be used to treat APE (except AngioVac endovascular system). The use of these modalities depends upon patient characteristics (comorbidities and hemodynamic status), clot characteristics (location of CIT, acute versus chronic clot, presence of PFO and clot lodged in PFO) and institutional factors (resource availability and local expertise to perform the procedure). Therefore, management of CIT cannot be based on “one size fits all” approach and requires emergent activation of multidisciplinary pulmonary embolism response teams (PERTs) involving emergency physician, Intensivist, hospitalist, cardiology, cardiothoracic surgery and interventional radiology, to make appropriate individualized management decisions based on above factors. 18 Factors determining the timing of clot migration into the pulmonary vasculature have not been studied well, although highly mobile, free-floating clots, those prolapsing across the tricuspid or pulmonic valve, or associated with large clot burden, are likely to embolize soon. Since it is difficult to predict when clot can dislodge and move into the pulmonary circulation, patients with CITs should be closely watched in the monitored setting like intensive care unit or until more definitive interventions like endovascular catheter-based therapies or surgical embolectomy is performed. Systemic thrombolysis should be available at bedside (unless contraindicated) and be administered readily if patients show any evidence of clinical deterioration due to clot dislodgement into pulmonary circulation (vital signs, respiratory or mental status). Intensivists should readily perform bedside POCUS if the clinical status changes to evaluate for clot migration.
Treatment modalities for CIT management.
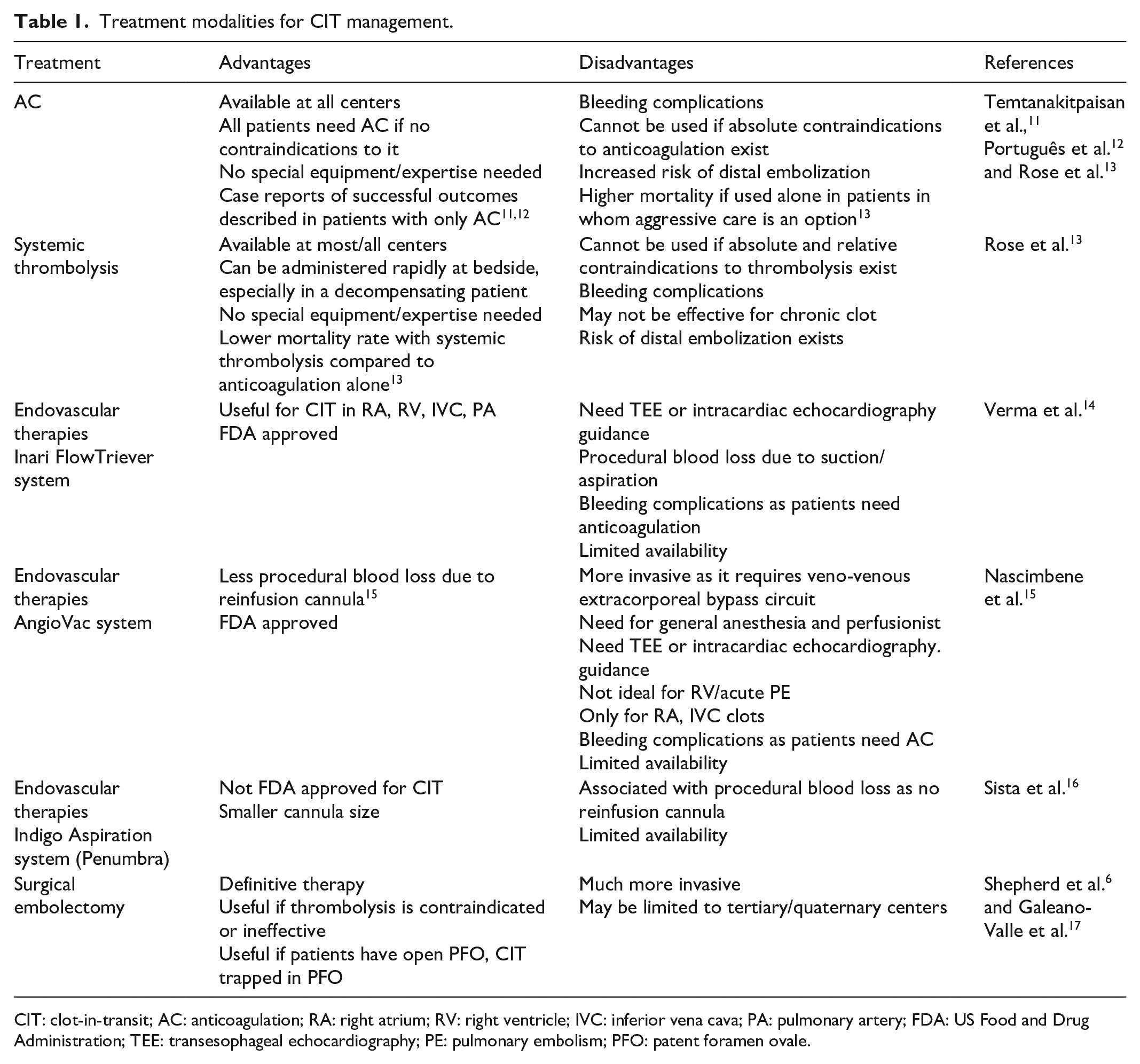
CIT: clot-in-transit; AC: anticoagulation; RA: right atrium; RV: right ventricle; IVC: inferior vena cava; PA: pulmonary artery; FDA: US Food and Drug Administration; TEE: transesophageal echocardiography; PE: pulmonary embolism; PFO: patent foramen ovale.
Conclusion
CIT, a ticking time bomb, is a serious life-threatening manifestation of acute venous thromboembolism. Our case series report emphasizes that highly mobile CITs can embolize in the matter of seconds and be immediately fatal. Management requires emergent multidisciplinary involvement and quick decision-making by the PERT, bedside availability of systemic thrombolysis and immediate POCUS examination to monitor the CIT if any change in clinical status.
Footnotes
Author contributions
R.I. and F.A. wrote the initial draft of the manuscript; A.P. contributed by collecting/processing images, table, editing the manuscript; S.K. contributed by editing the manuscript, images, table and providing overall supervision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual anonymized case report or series.
Informed consent
Written informed consent was obtained from a legally authorized representative for anonymized patient information to be published in this article (Case 1) as the patient was deceased and written informed consent was obtained from the patient for their anonymized information to be published in this article (Case 2).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
