Abstract
Hypoplastic aorto-iliac syndrome is an unusual cause of claudication or limb ischemia. Hypoplasia of the vena cava and iliac veins is an even rarer entity and is usually found incidentally in patients presenting with extensive deep venous thrombosis or unexplained pulmonary embolism. Both are thought to be due to in-utero developmental defects or vascular accidents during maturation of the fetus. Each entity is so rare that only short series or case reports are found in the literature. We report an unusual case of a 64-year-old Caucasian male with no atherosclerotic risk factors who presented with a 2-month history of left hip and leg pain, followed by significant left leg swelling. Venous duplex confirmed the diagnosis of deep venous thrombosis and anticoagulation therapy and compression were initiated. Because of worsening symptoms, percutaneous venous thrombectomy was attempted with only a small amount of thrombus extracted. Venography and intravascular ultrasound revealed a hypoplastic, chronically occluded inferior vena cava and iliac veins without thrombus, venous collaterals, or an enlarged azygous system. CT angiography confirmed hypoplasia of the venous system but also revealed a severely diseased aortoiliac system with a chronically occluded left common iliac artery and reconstitution of the left common femoral artery via a large lumbar collateral vessel. Despite improvement in venous symptoms, disabling left leg claudication persisted. Aortoiliac stenting was performed but failed within 6 weeks. The patient then underwent a successful aortobi-iliac bypass with a Dacron graft for his arterial insufficiency. The aorta was noted to be thickened, fibrotic, and mildly atherosclerotic, with thrombus throughout the aorta and iliac arteries. Hypoplastic aortoiliac syndrome and hypoplasia of the vena cava and iliac veins are both rare entities. To our knowledge, this is the first published case report describing both entities in a single patient. In addition, this may be the first published report of both conditions requiring simultaneous invasive treatment.
Keywords
Background
Hypoplastic aorto-iliac syndrome (HAIS) or small aorta syndrome is an unusual cause of claudication or limb ischemia. Current literature suggests that this entity accounts for 5%–15% of all aortoiliac occlusive disease, is mostly seen in females, and typically presents in the fourth to fifth decade of life.1,2 The etiology of HAIS is thought to be a congenital ischemic incident in-utero, leading to lack of proper development of the major arterial structures of the abdomen and pelvis. 2 There also appears to be an overlap with inflammatory and fibrotic disease pathologies such as arteritis. 3 HAIS can be associated with other arterial anomalies such as subclavian, carotid, or renal artery stenoses. 2 The most common presenting symptom of HAIS is claudication.1,3,4 Symptomatic disease is best managed with arterial bypass, with few reports of successful minimally invasive treatment due to the etiology and pathophysiology of the disease process.1,4 The literature shows higher rates of re-occlusion among endovascular approaches compared to open bypass procedures and compared to endovascular therapies in non-HAIS peripheral arterial disease. 1 The underdevelopment of the vascular tree leads to extensive, diffuse thickening and fibrosis of the media, premature atherosclerosis, and eventual vessel occlusion, prohibiting long-term success with endovascular therapy.
Congenital malformations of the inferior vena cava (IVC) are an even rarer entity, prevalent in 1%–4% of the population,5,6 with hypoplasia of the vena cava and iliac veins being the rarest of said malformations, present in <1% of the general population.6,7 Venous malformations are typically found incidentally in young patients presenting with extensive deep venous thrombosis (DVT), iliac and caval thrombosis, chronic nonhealing wounds of the lower extremities, or unexplained pulmonary embolism (PE).6–9 Congenital venous malformations are often associated with other developmental anomalies, such as renal aplasia and polysplenia.5,9 Typically, symptomatic disease is treated through suction thrombectomy and prolonged or life-long anticoagulation.5,7,8 Surgical repair is typically reserved for cases of severe symptoms or complications that do not respond to medical management, and for symptomatic agenesis of the IVC.10–12 It is likely that the etiology of this disease process is also an in-utero vascular accident, preventing the proper development of the IVC and iliac veins; however, the exact mechanism is not well understood.10,13 A high index of suspicion is necessary in young patients with atypical presentations of DVT, with further imaging investigation warranted to identify possible venous anomalies.5,6,13
Several case reports and small case series have been published describing each entity independently.7,14,15 Herein, we report a rare case of a male patient who presented with simultaneously symptomatic HAIS and hypoplastic IVC and iliac veins, requiring an iliofemoral venous thrombectomy and anticoagulation for an acute, severely symptomatic left DVT, followed shortly thereafter with aortoiliac bypass for treatment of short distance disabling claudication.
Case description
A 64-year-old Caucasian male with no previous significant past medical history was initially referred to vascular surgery for evaluation of an extensive left femoropopliteal DVT causing severe acute thigh pain and persistent swelling despite proper anticoagulation and compression therapy. Upon questioning of his symptoms, he revealed significant left hip and buttock pain for 4–6 weeks preceding the onset of swelling and thigh pain. This pain was initially thought to be arthritic or musculoskeletal in nature, but plain radiographs did not show significant hip arthritis. Once the diagnosis of DVT was established by duplex scan, the initial symptoms were thought to be venous claudication caused by a possible iliofemoral DVT. Prior to his visit, he had a full medical evaluation. There was no hypercoagulable state found on extensive hematological workup and his labs were otherwise unremarkable. There was no dyslipidemia, diabetes, hypertension, or history of smoking. There was no family history of atherosclerosis or coronary artery disease. The patient had a normal 2D echo. Physical exam showed a moderately swollen thigh and calf, with a normal arterial exam. He was on Apixaban for the DVT. He was compliant with thigh-high 20–30 lb. graded compression stockings.
Due to his persistent symptoms of pain, swelling, and inability to ambulate despite standard maximal medical treatment, he underwent a left leg venogram from a left popliteal approach that revealed acute and subacute thrombosis of his left femoral and popliteal venous system (Figure 1). A percutaneous thrombectomy of the iliofemoral system with a ClotTriever Device (Inari Medical/Skryker) was performed, but the procedure yielded minimal clot extraction from the iliac veins and IVC. Completion venography showed chronic thrombus in the common femoral vein and narrowing of the left iliac system with significant underfilling of the vena cava. An intravascular ultrasound (IVUS; Volcano System; Philips Medical) evaluation was then performed, showing chronic narrowing and scarring of the infrarenal vena cava and entire left iliac venous system to the level of the common femoral vein with thickened walls and almost no lumen. No residual thrombus was identified on IVUS (IVUS images are not available for publication). With findings consistent with hypoplasia of the venous system, the patient was treated with continued anticoagulation and compression.

Operative venogram left femoral-popliteal system. (Left) Pre thrombectomy with thrombus. (Right) Post thrombectomy.
Further evaluation with CT angiography confirmed the diagnosis of hypoplasia of the venous system. Surprisingly, it also revealed a severely diseased, undersized, and atherosclerotic infrarenal aorta and iliac arteries with complete occlusion of the left common iliac artery (CIA). A large lumbar collateral reconstituted the left common femoral artery. The right iliac arterial system was extensively diseased but not occluded (Figure 2). Given his persistent left hip, buttocks, and thigh pain, despite improvement in his venous symptoms, it became clear that he was suffering from arterial claudication in the setting of severe aorto-iliac disease. It was clear from his imaging that he had both HAIS and hypoplasia of his IVC and iliac veins. With that diagnosis in mind, we offered him definitive surgical repair with aortoiliac or aorto-bifemoral bypass; however, he initially refused open surgical intervention. Therefore, at the patient’s request for minimally invasive therapy, we placed two kissing stents (Gore VBX) extending from the distal aorta into the bilateral CIA with good restitution of flow (Figure 3). As expected, this endovascular intervention failed after 6 weeks, despite continued anticoagulation. Follow-up angiography revealed early re-occlusion of both iliac stents (Figure 4) with preservation of lumbar collaterals. A suction thrombectomy of the stents was attempted, but was unsuccessful.

CTA abdomen and pelvis demonstrating extensive atherosclerotic changes of distal aorta and bilateral iliac arteries, left worse than right, with left common iliac artery occlusion. CTA: CT angiography.
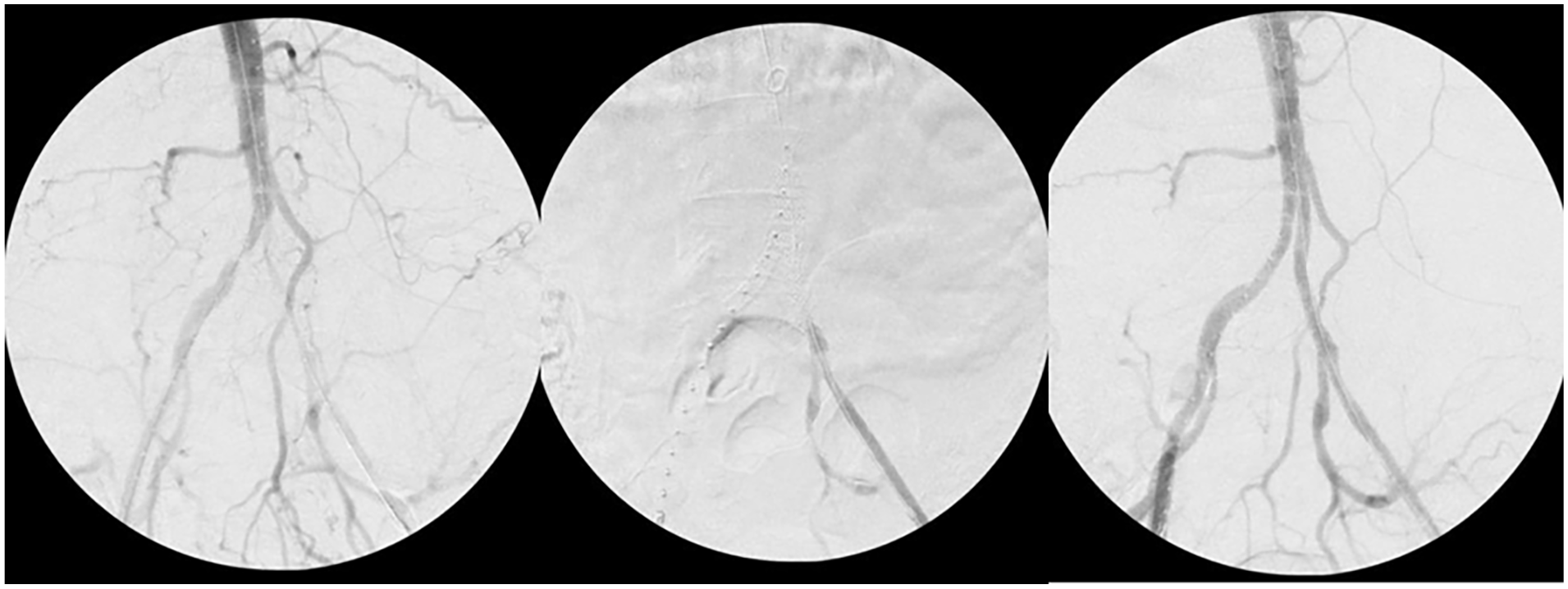
Intra-operative fluoroscopy from diagnostic angiography and bilateral kissing stent placement. (Left) Diagnostic angiography showing hypoplastic bilateral iliac arteries, left worse than right, and showing large collateral. (Middle) Kissing iliac stent placement. (Right) Post stent placement with improved flow.

OR fluoroscopy of diagnostic angiogram and attempt at suction thrombectomy of kissing stents. (Left) Diagnostic angiography. (Right) Post thrombectomy with poor restitution of flow.
After failed endovascular treatment, the patient agreed to open surgical bypass, and underwent an uncomplicated aortobi-iliac bypass with a 16 × 8 mm Gelsoft Dacron Graft (Terumo). Operative findings at the time of bypass confirmed a thickened and fibrotic media of the aorta, atherosclerotic degeneration with areas of thrombosis, and undersized CIA with compressed and occluded common iliac stents. The aorta at the level of renal arteries was mildly atherosclerotic as well. The external iliac arteries bilaterally were completely free of disease. The fibrosis and thickening of the CIA mimicked that of an arteritis.
His postoperative course was unremarkable. At 1-year follow-up, he remains claudication free with a patent bypass. His left limb swelling is now minimal with compliance with compression therapy. He will be treated with lifelong low-dose anticoagulation for his venous disease to prevent future venous thromboembolic events. Endovascular reconstruction of his vena cava and iliac veins was not indicated as his venous symptoms were eventually controlled with compression therapy and anticoagulation.
Discussion
The development of the IVC is a complex process from an embryological perspective. The IVC is a composite structure that develops from three pairs of embryologic veins (posterior cardinal, subcardinal, and supracardinal). During embryological maturation, these veins merge, regress, and coalesce, forming in most people one right-sided IVC. However, many variations may persist at full term, leading to a variety of known venous congenital anomalies. The most common are duplication of the IVC, a left-sided IVC, atresia (aplasia) of the IVC, and hypoplasia of the IVC.10,13
The incidence of congenital venous anomalies ranges from 0.5% to as high as 3% in most series, and abnormalities found incidentally on CT scanning account for more than half of these reported cases. The incidence of finding a venous anomaly as the cause for as clinical event is much higher than its incidence in the general population.5–9 Hypoplasia and aplasia of the IVC are the rarest of all congenital venous anomalies, with a reported incidence of 0.005%–1% in the general population.6,7
Hypoplasia is rarely diagnosed early in life as it is usually asymptomatic. Due to the incredibly low incidence and the slow progression of disease, hypoplasia typically presents in the fourth or fifth decades, and most commonly as a DVT or PE.6,7 Interestingly, of all the congenital venous anomalies of the IVC, hypoplasia is by far the most common one associated with a venous thromboembolic event. 6 It often goes undiagnosed until there is a clinical event, such as leg pain or swelling, an unprovoked iliofemoral venous thrombosis, or PE, as in this patient.
Hypoplasia of the IVC and iliac veins is best treated with long-term anticoagulation to prevent further thromboembolic events. If necessary, invasive treatment such as thrombolysis or thrombectomy can be performed for acute DVT or PE when indicated by clinical severity, such as in this case. Reconstruction of the venous system with stenting is rarely necessary or recommended.10–12 Without severe bilateral edema and ulcers, reconstruction was not indicated in this patient.
In contrast, agenesis or atresia can be easily diagnosed with associated findings of a large azygos or hemi-azygos system on CT or venography, and usually presents early in life in the first or second decades with leg swelling, large varicose veins in childhood, abdominal varicosities, and venous ulcers.10,11 These patients are usually very symptomatic at an early age. Treatment of atresia has surprisingly excellent results with endovascular reconstruction of the cava and iliac venous system, and anticoagulation.10–12
HAIS typically occurs in women and presents in the fourth or fifth decade of life.1,2 HAIS is thought to be due to a vascular accident in utero or a developmental anomaly, but there are reports in the literature suggesting an inflammatory component as well, similar to arteritis.2,3 Commonly, there is significant early atherosclerotic degeneration of the hypoplastic segment, leading to occlusion and ischemic symptoms. These patients frequently have no atherosclerotic risk factors and no family history of early atherosclerotic disease, though some may have a significant smoking history. The exact etiology of the inflammatory and degenerative components of the disease are not fully understood. Although almost all case reports and case series describe this as occurring predominately in females, it is possible for it to occur in males as well. The typical presentation is of hip, thigh, and buttocks claudication due exclusively to aortoiliac disease in the absence of infra-inguinal disease.
While endovascular therapies continue to gain traction, aortoiliac or aortofemoral bypass remains the best option for repair of HAIS, as the disease is not well suited for long-term success with endovascular therapy. Surgical bypass/reconstruction should yield excellent lifetime patency in the absence of infra-inguinal disease in a nonsmoker.
This patient’s initial symptoms of claudication due to iliac artery disease were not initially recognized as such due to his normal arterial examination upon presentation, and were further masked by his extensive and symptomatic iliofemoral and femoropopliteal DVT. The large collateral that reconstituted his left common femoral artery was so robust that he had palpable pulses in his left leg. Since his initial presentation was for the DVT, our assumption was that his hip pain was venous claudication due to an iliofemoral venous thrombosis. As there was no previous history of claudication, nor any atherosclerotic risk factors, we did not expect to find aortoiliac occlusive disease on evaluation. With both arterial disease and venous disease presenting at the same time, with equal severity and acuity, this underscores the importance of a complete diagnostic evaluation prior to planning definitive treatment.
Conclusion
Although both HAIS and hypoplasia of the IVC are rare, both are well described independently in small case series and case reports. We chose to present this case for publication as, to our knowledge, HAIS and hypoplastic vena cava have not been previously reported in the same patient, nor have these two entities been described as presenting simultaneously.
In isolation, HAIS and hypoplastic cava are extremely rare entities. Patients who present with arterial or venous disease later in life with no previous contributing history, and without risk factors for either entity, should be evaluated extensively for vascular anomalies. A high index of suspicion must be present for these disease entities for the proper diagnosis to be made, and for appropriate planning of definitive therapy. This patient’s presentation reinforces the need to include these entities in the differential diagnosis for patients with atypical presentations of claudication and DVT, as well as the need to evaluate for further vascular and developmental anomalies in the setting of either entity.
While endovascular therapies continue to gain traction as definitive repair for HAIS, surgical bypass remains the gold standard for treatment, particularly in a relatively young patient population, given the higher rates of re-occlusion for endovascular repair. Endovascular reconstruction for hypoplasia of the vena cava and iliac veins is rarely necessary, as clinical severity at time of presentation usually does not warrant reconstruction of the IVC. Anticoagulation remains the treatment of choice.
Footnotes
Acknowledgements
We would like to thank Monmouth Medical Center and the Division of Vascular Surgery for their support in this research. Special thanks to the patient and their family for their encouragement throughout the study.
Ethical considerations
There was no ethical board or committee approval required for this case report. Our institution does not require ethical approval for reporting individual cases or case series.
Consent for publication
The patient provided written informed consent for publication of this case report and any accompanying images.
Author contributions
Conceptualization: Devon Kelley and Randy Shafritz. Data curation: Devon Kelley and Randy Shafritz. Supervision: Randy Shafritz. Writing – original draft: Devon Kelley and Randy Shafritz. Writing – review and editing: Devon Kelley and Randy Shafritz.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
