Abstract
This case report details a rare presentation of acute coronary syndrome in a young adult male with no significant medical history. A 24-year-old male presented with acute chest pain, and his initial ECG revealed the rare de Winter pattern, an underrecognized ST-elevation myocardial infarction (STEMI) equivalent. The de Winter T-wave morphology evolved into anterior STEMI, emphasizing the critical need for prompt recognition and intervention. Coronary angiography demonstrated mild coronary artery ectasia (CAE) with slow blood flow in the left anterior descending (LAD) and left circumflex arteries, alongside intramuscular bridging. Advanced imaging via optical coherence tomography identified ruptured plaque and mild spontaneous dissection as the underlying pathology. Initial treatment involved dual antiplatelet therapy, beta-blockers, statins, and low-molecular-weight heparin, leading to clinical stabilization and normalization of cardiac function. This case underscores the diagnostic challenges associated with CAE, particularly in young patients, and highlights the utility of advanced imaging modalities in personalizing treatment strategies. CAE, affecting 1%–5% of patients undergoing coronary angiography, is characterized by aneurysmal coronary dilation. It poses unique therapeutic challenges due to its association with turbulence, thrombus formation, and vulnerability to ischemic events. The findings revealed that even mild CAE, when coupled with structural abnormalities such as ruptured plaque, can result in significant ischemic complications. The case demonstrates the importance of multidisciplinary care and individualized management, combining medical therapy with advanced diagnostic tools. It also emphasizes the need for familiarity with the de Winter ECG pattern to facilitate timely reperfusion therapy and mitigate adverse outcomes. Further research into CAE pathophysiology, including genetic, inflammatory, and hemodynamic aspects, is essential to enhance risk stratification and optimize treatment approaches for this complex condition.
Keywords
Introduction
Coronary artery ectasia (CAE) is a rare coronary disorder characterized by diffuse aneurysmal dilation of coronary arteries, affecting around 1%–5% of patients undergoing coronary angiography (CAG).1,2 While often considered a variant of coronary atherosclerosis, CAE has complex and multifactorial causes. This case highlights a rare yet significant association between ST-elevation myocardial infarction (STEMI) and CAE.
Case report
A 24-year-old male with no prior medical or surgical history presented to the emergency room with sudden onset of central chest pain radiating to the left shoulder, lasting for 1 h, and associated with vomiting. He reported a similar episode 4 months earlier that was relieved by antacids. His family history was significant for ischemic heart disease, as his father had died of cardiac arrest. The patient was a heavy smoker and consumed alcohol occasionally.
Upon arrival at the hospital, the patient was hemodynamically stable with a blood pressure of 160/100 mmHg. Physical examination revealed no significant abnormalities. A chest X-ray demonstrated increased bronchovascular markings. The initial high-sensitivity cardiac troponin-I level was normal at 14.3 ng/L (normal range < 40 ng/L).
The admission ECG showed a regular sinus rhythm with features indicative of de Winter’s T-waves combined with a feature of an anterior STEMI. Key findings included upsloping ST-segment depression with tall, symmetrical, and peaked T-waves (de Winter’s T-waves) in leads V3–V6, preceded by ST-segment depression ⩾1 mm at the J point, with the ascending limb of the T-wave starting below the isoelectric baseline. In addition, there was ST-segment elevation ⩾1 mm in the septal leads (V1–V2) and high lateral leads (I and aVL). Reciprocal changes included ST-segment elevation (⩾1 mm) in lead aVR and ST-segment depression in the inferior leads (II, III, and aVF). These findings are consistent with an acute occlusion of the left anterior descending (LAD) coronary artery proximal to the first diagonal branch (Figure 1).
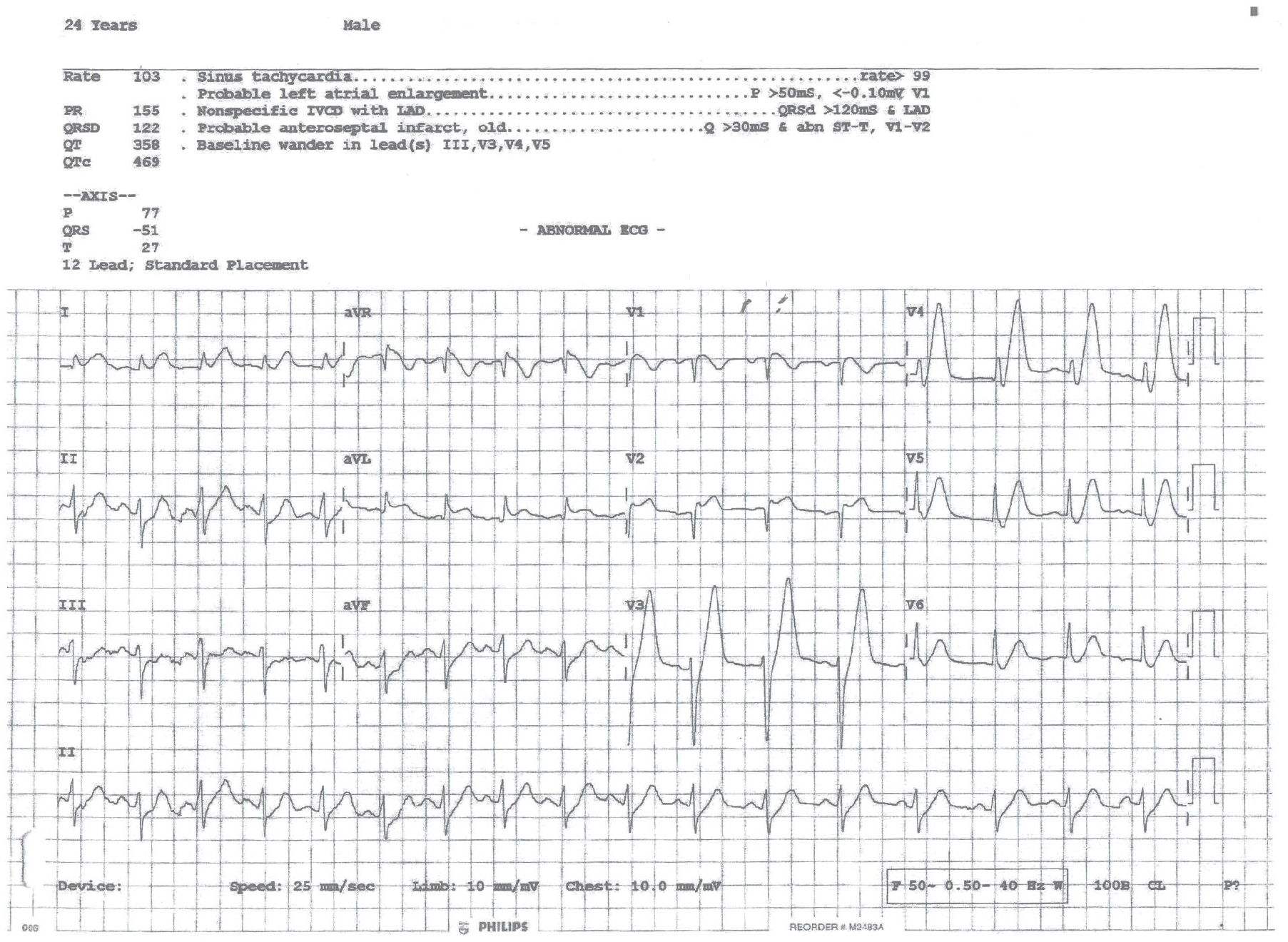
The admission ECG showing a regular sinus rhythm with features indicative of de Winter’s T-waves combined with feature of an anterior STEMI. There is upsloping ST-segment depression with peaked de Winter’s T-waves in leads V3–V6, preceded by ST-segment depression ⩾1 mm at the J point, with ST-segment elevation leads V1–V2 and high lateral leads (I and aVL), and reciprocal ST-segment elevation in aVR and ST-segment depression in the inferior leads.
The patient was treated with oxygen, 300 mg of aspirin, 300 mg of clopidogrel, and 0.5 mg of sublingual glyceryl trinitrate before being transferred to the cardiac care unit (CCU) for close monitoring. Serial cardiac enzyme tests revealed a significant rise in troponin-I levels, increasing to 797.9 ng/L 4 h after hospital admission and peaking at 955 ng/L at the 10-h mark. Transthoracic echocardiography showed mild left ventricular systolic dysfunction with an ejection fraction of 50% and mild hypokinesia of the anterior wall.
CAG via the right radial artery (Figure 2, and Supplemental Videos 1–6) revealed a mild coronary ectasia in the LAD artery with slow blood flow and evidence of intramuscular bridging in the mid-segment (Figure 2, Panels a, b; and Supplemental Videos 3–6). Mild ectasia with the slow flow was also observed in the left circumflex artery (Figure 2, Panels a–c; and Supplemental Videos 3–6) while the right coronary artery showed a mild slow blood flow (Figure 2, Panel d; and Supplemental Videos 1 and 2). Optical coherence tomography (OCT) revealed a ruptured plaque and mild spontaneous dissection, neither of which impeded coronary flow.

CAG via the right radial artery (Figure 2) revealing a mild coronary ectasia in the proximal left anterior descending artery with slow blood flow and evidence of intramuscular bridging in the mid-segment (Panels a, b), mild ectasia with slow flow in the left circumflex artery (Panels a–c) while the right coronary artery showed a mild slow blood flow (Panel d).
The patient was treated with 100 mg aspirin, 75 mg clopidogrel, 2.5 mg bisoprolol, 40 mg atorvastatin, and 60 mg subcutaneous enoxaparin. After 24 h, a follow-up ECG showed sinus rhythm with T-wave inversions in leads I, aVL, and chest leads (V2–V6), consistent with the evolving ECG pattern in STEMI after reperfusion (Figure 3). Troponin-I level decreased to 132 ng/L. A repeated echocardiogram revealed normal chamber dimensions, an ejection fraction of 64%, and grade-1 diastolic dysfunction. The patient was stabilized and discharged three days later on a regimen of aspirin, clopidogrel, atorvastatin, and bisoprolol. The patient was reviewed in the Cardiology clinic 1 month later and was found to be stable, with no further episodes of chest pain. He demonstrated strict adherence to medical treatment and lifestyle modifications, including complete smoking cessation.

Follow-up ECG done after 24 h, showing a sinus rhythm with deep T-wave inversions in leads I, aVL, and chest leads (V2–V6), consistent with the evolving ECG pattern in STEMI after reperfusion.
Discussion
This report presents a rare case of a young patient with mild CAE who developed STEMI, a condition typically observed in older individuals with conventional risk factors. The admission ECG notably demonstrated the rare de Winter pattern, observed in approximately 2% of acute LAD coronary artery occlusions and often underrecognized by clinicians. 3 Verouden et al. reinforced this finding in a subsequent case series, 4 identifying the de Winter ECG pattern in 35 out of 1890 patients (2%) undergoing PCI for LAD occlusion. Patients with this pattern were younger, predominantly male (as seen in our patient), and exhibited a higher incidence of hypercholesterolemia compared to those with a classic STEMI presentation. It is critical for cardiologists and emergency physicians to recognize the distinctive de Winter ECG pattern to ensure prompt triage for immediate reperfusion therapy.3,4 Lack of familiarity with this high-risk ECG presentation can lead to delays in initiating appropriate treatment—such as failure to activate the catheterization lab—resulting in adverse effects on patient morbidity and mortality. 4 The de Winter ECG pattern is now recognized as a “STEMI-equivalent” in many acute coronary syndrome guidelines, warranting urgent reperfusion therapy.4–6
This case highlights the diagnostic challenges, clinical presentation, and management considerations for such atypical cases, noting the patient’s young age, mild ectasia, and spontaneous dissection as the underlying mechanism.
CAE is defined as coronary artery dilation exceeding 1.5 times the diameter of adjacent normal segments. 2 Its prevalence among patients undergoing coronary angiography ranges from 1.2% to 5%. 7 The right coronary artery is most frequently affected, followed by the LAD and left circumflex arteries.8,9 In this case, CAE was mild, with the proximal LAD measuring 4.0–4.5 mm, and slow flow was observed on angiography. Despite the lack of pronoun-ced ectasia, the OCT findings of ruptured plaque and spontaneous dissection confirmed a mechanism capable of triggering significant ischemic events. This underscores that even mild ectasia exists on a spectrum and may result in severe complications.
CAE has a multifactorial etiology, including genetic predispositions, inflammation, endothelial dysfunction, and hemodynamic alterations. 10 Ectatic segments disrupt laminar blood flow, promoting turbulence, thrombus formation, and plaque rupture, while altered vessel geometry and shear stress further contribute to endothelial damage and accelerated atherosclerosis. These abnormalities, coupled with impaired vasomotor tone, increase vulnerability to ischemic events during periods of heightened oxygen demand, such as physical exertion or emotional stress. 11
OCT was pivotal in diagnosing spontaneous dissection, which differentiated this case from vasospasm 12 (unfortunately, the release of OCT images was prohibited as per the hospital’s policy). Unlike vasospasm, which resolves with nitrates or calcium channel blockers and lacks structural changes, OCT identified ruptured plaque and dissection, confirming a structural etiology. Clinically, the patient’s presentation of sudden chest pain, ECG changes, and troponin elevation aligned with spontaneous dissection rather than vasospasm, where ischemia usually resolves without structural damage. Advances in imaging, including OCT and intravascular ultrasound (IVUS), enhance understanding of CAE morphology and pathophysiology. 13 Noninvasive imaging techniques such as CT coronary angiography provide detailed visualization of lumen size and morphology and are useful for monitoring disease progression. 14 While follow-up imaging was not performed in this case, incorporating CT angiography in future evaluations is recommended to assess changes in coronary anatomy and ectasia progression.
Managing STEMI in patients with CAE poses unique challenges. Treatment options include manual aspiration thrombectomy, intracoronary glycoprotein IIb/IIIa inhibitors, thrombolysis, and mesh-covered stent implantation. 15 However, these interventions are complicated by high thrombus burden, difficulty in stent sizing, and risks such as the no-reflow phenomenon and distal embolization.16,17 When percutaneous coronary intervention is not feasible, surgical options like bypass grafting or aneurysmal thrombectomy may be necessary.13,15
In this case, medical therapy with antiplatelets, beta-blockers, and statins successfully stabilized the patient, emphasizing the importance of individualized management strategies. Emerging biomarkers for vascular inflammation and endothelial dysfunction may further improve risk stratification and guide targeted therapies in CAE patients.
This case highlights the need for advanced imaging, multidisciplinary management, and personalized treatment approaches to address the complexities of CAE. Continued research into the genetic, inflammatory, and hemodynamic factors of CAE is essential for improving patient outcomes.
Conclusions
This case underscores the diagnostic and management challenges of CAE in young patients with de Winter ECG pattern morphing into anterior STEMI, offering insights into its partially understood mechanisms. Multidisciplinary care and personalized treatment are crucial for better outcomes. Further research into the genetic, inflammatory, and hemodynamic aspects of CAE is essential for improved risk stratification and management.
Supplemental Material
sj-docx-1-sco-10.1177_2050313X251331733 – Supplemental material for A case report of complex acute coronary syndrome presentation: Plaque rupture and mild coronary artery ectasia presenting as de Winter T-waves morphing into anterior ST-elevation myocardial infarction in a young adult male
Supplemental material, sj-docx-1-sco-10.1177_2050313X251331733 for A case report of complex acute coronary syndrome presentation: Plaque rupture and mild coronary artery ectasia presenting as de Winter T-waves morphing into anterior ST-elevation myocardial infarction in a young adult male by Momena A. Daas, Maryam A. Almasaabi, Eman M. Abdrabou, Mohammed Elmahal, Abdul-Majeed O. Mahdi, Eva A. Tello, Ousama Mahdi, Abdulrahman E. Alayyaf, Ahmad J. Aladwani and Mahmoud M. Ramadan in SAGE Open Medical Case Reports
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
