Abstract
Although disseminated cryptococcosis can occur occasionally, it is most commonly seen in immunodeficient patients. In 2005, a 43-year-old man was diagnosed with polycythemia vera. Following in 2018, he experienced an unknown-cause fever and headache. To establish the source of the symptoms, a magnetic resonance imaging scan of the brain was performed, which indicated meningeal and gyral-leptomeningeal thickening and several localized T2 hyperintense lesions measuring up to 10 × 14 mm in diameter. Cryptococcus neoformans was then cultivated from cerebrospinal fluid. Serum IgM antibodies against West Nile Virus were positive. After 8 weeks of treatment with amphotericin B and fluconazole, the overall condition improved, and the cerebrospinal fluid control culture became negative. The symptoms returned shortly after discontinuing antifungal therapy, necessitating the reintroduction of fluconazole. Currently, the patient is stable and responding positively to ruxolitinib. Here, it is demonstrated how a patient with polycythemia vera due to immunological weakness might develop disseminated cryptococcosis of the brain after West Nile virus infection.
Introduction
Cryptococcosis is an opportunistic fungal infection that typically manifests as a pulmonary infection or meningoencephalitis. It is caused by Cryptococcus neoformans in 95% of patients and Cryptococcus gatti in 4%–5%. 1 Cryptococcus can be found in the excretions of birds, most commonly pigeons, in soil, juices, and even milk. Infection occurs through inhalation of spores in respiratory passages, where they remain silent and asymptomatic, but after the patient’s immune system is compromised, they may hematogenously disseminate in almost any organ, usually the brain, leading to death in nearly one-third (28.8%) of the patients.2,3 During the last few decades, the incidence of disseminated cryptococcosis has increased, particularly in HIV-infected patients and especially in resource-limited countries. 4 However, since the introduction of more effective antiviral therapy for HIV restoration of immune function and early diagnosis, the incidence of HIV-associated cryptococcosis has decreased significantly, but it has increased in other immune deficient patients, such as those with organ or bone marrow transplantation, particularly those performed for lymphoproliferative diseases, and in patients on long-term immunosuppression.3,5,6 Cryptococcal meningoencephalitis can be fatal or curable, depending on the patient’s previous general condition, level of immunodeficiency, multiplicity, and size of lesions within the brain, virulence, burden, and molecular specificity of Cryptococcus, as well as timely, appropriate, and long-term treatment.7,8 The purpose of this study is to show that disseminated cryptococcosis brain alterations in polycythemia vera patients can be treated successfully in HIV-negative patients.
The case report
During a routine checkup in 2005, a 43-year-old man was diagnosed with polycythemia vera with the JAK2 mutation. Serum erythropoietin (Epo) level was low, which corresponded to the diagnosis of polycythemia vera. The patient rejected treatment because he had no symptoms. A year later, he reported abdominal pain and an investigation indicated portal and splenic vein thrombosis. He was started on 1500–2000 mg/dl hydroxycarbamide, acetylsalicylic acid, and oral anticoagulant medication, with regular INR checks and phlebotomies. A medical examination in 2011 found that the thrombotic veins had not been recanalized, the spleen was 230 mm, the accessory spleen was 50 mm, and grade I/II varicosities were present in the distal esophagus and proximal stomach.
The patient was taken to the hospital in October 2018 with a fever and headache. Standard tests were performed, which revealed that the repeated blood culture, urine culture, and the patient’s HIV status were negative. IgM antibodies to the West Nile virus (WNV) were found in the serum, and C. neoformans was found in the cerebrospinal fluid culture. It is known that disseminated cryptococcosis can occur in immunocompromised patients. 2 We suspected this because in that period there was an epidemic of WNV infection in Europe and Serbia, which can be asymptomatic.
A brain magnetic resonance imaging (MRI) revealed that seven heterogeneous and inhomogeneous hyperintense T2 lesions up to 10 × 14 mm in diameter are observed in the head of the caudate nucleus bilaterally more pronounced on the right (Figure 1(a)), in the genu of the internal capsule, and the globus pallidus on the left (Figure 1(a)) without diffusion restriction (Figure 1(d)–(g)) and post-contrast enhancement of the signal intensity (Figure 1 (h) and (i)). Similar but less oval, inhomogeneous lesions can be seen along the body of the left lateral ventricle parietal (Figure 1(b)), in the center of the semioval on the right, as well as frontally and parietal on the left in the center of the semioval (Figure 1(c)). The described lesions correspond to dilated perivascular spaces in the sense of gelatinous pseudocysts, which are typical for C. neoformans but are not seen in WNV infection. In Cryptococcus infection, lesions can also occur in the basal nuclei and changes are more homogeneous and diffuse. Thus, in the case of cryptococcal infection, the presence of pseudocysts has been reported as a specific finding on MRI. The post-contrast FLAIR sequence shows dural augmentation of signal intensity occipitoparietal more pronounced left, as well as the gyral-leptomeningeal frontoparietal bilateral more prominent right (Figure 2). Dural and leptomeningeal enhancement of signal intensity can characterize both C. neoformans and WNV infections and cannot be denied neuroradiologically because they occur in both entities.
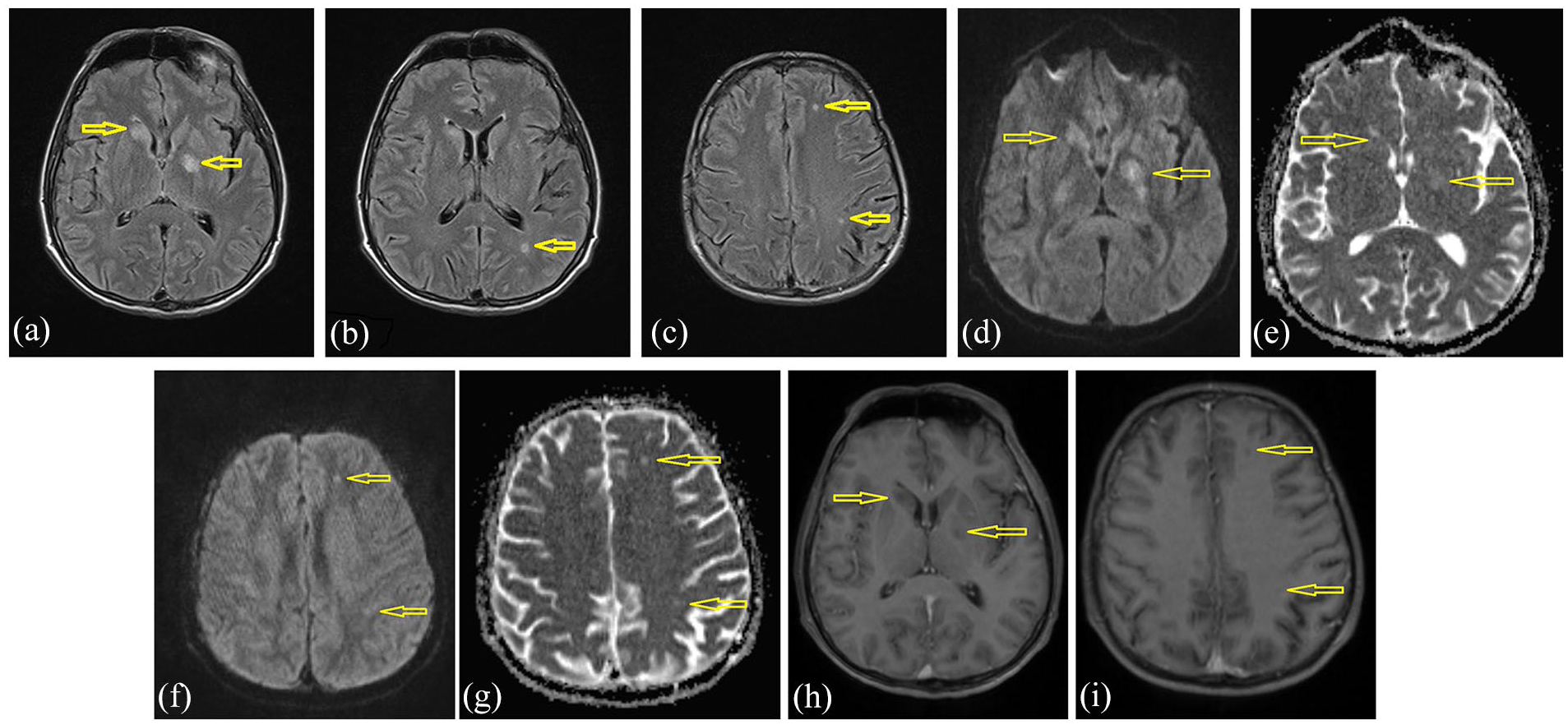
T2 hyperintense lesions are seen in the head of the nucleus caudatus on the right, globus pallidus on the left (a), peritrigonal on the left (b), and frontoparietal subcortical on the left (c), without diffusion restriction ((d)–(g)) and enhancement of the signal intensity ((h) and (i)) (yellow arrows).

Axial postcontrast T1W scan.
Antifungal therapy with liposomal amphotericin B (5 mg/kg/day i.v.) and fluconazole (initial dose 2 × 400 mg p.o. first day, followed with 1 × 400 mg p.o.) was started. After 3 months of the treatment, the fever and headache decreased, and the cerebrospinal fluid culture remained negative after 2 weeks of incubation. Antifungal therapy was stopped, but the patient rapidly acquired a fever of 39°C, increased leukocyte count to 46 × 109/l (Table 1), and became bradypsychic and bradykinetic. On January 23, 2019, an abdomen computed tomography scan was performed. The findings revealed a 206 mm liver, mildly dilated intrahepatic bile ducts, a 277 × 222 × 105 mm spleen, a cavernous portal vein, a thrombosed splenic vein, and an auxiliary spleen of prior size. The oral anticoagulant, hydroxycarbamide, and fluconazole medication were continued for an additional 9 months. After the blood test results reverted to normal, hydroxycarbamide was ruled out. The patient developed cardiac insufficiency with pro-BNP levels of 12.000 ng/l. CD4+ cells were 150/mm3 (Table 1) at the time, and CD8 lymphocytes were 243/mm3. Even without hydroxycarbamide, hemoglobin was 100 g/l after 3 months of hospital therapy, white blood cells were 16 × 109/l, and platelets were 374 × 109/l, while bradypsychia and bradykinesia gradually disappeared. By the end of March 2019, polycythemia had worsened, and the liver and spleen were significantly enlarged. Because bone marrow cytology and biopsy revealed that polycythemia vera had progressed to post-polycythemic myelofibrosis, ruxolitinib (2 × 20 mg/day) was introduced on September 12th, 2019, along with prophylactic treatment with fluconazole 200 mg/d for the next 3 months. The liver and spleen were greatly reduced in size. Unfortunately, after 21 months of ruxolitinib treatment, he was diagnosed with epiglottic planocellular carcinoma, which required a laryngectomy and radiotherapy on August 16, 2019. The patient is currently stable on ruxolitinib, acetylsalicylic acid, and an oral anticoagulant therapy.
Complete blood findings, biochemical findings of serum, and cerebrospinal fluid in patients with cerebral cryptococcosis.
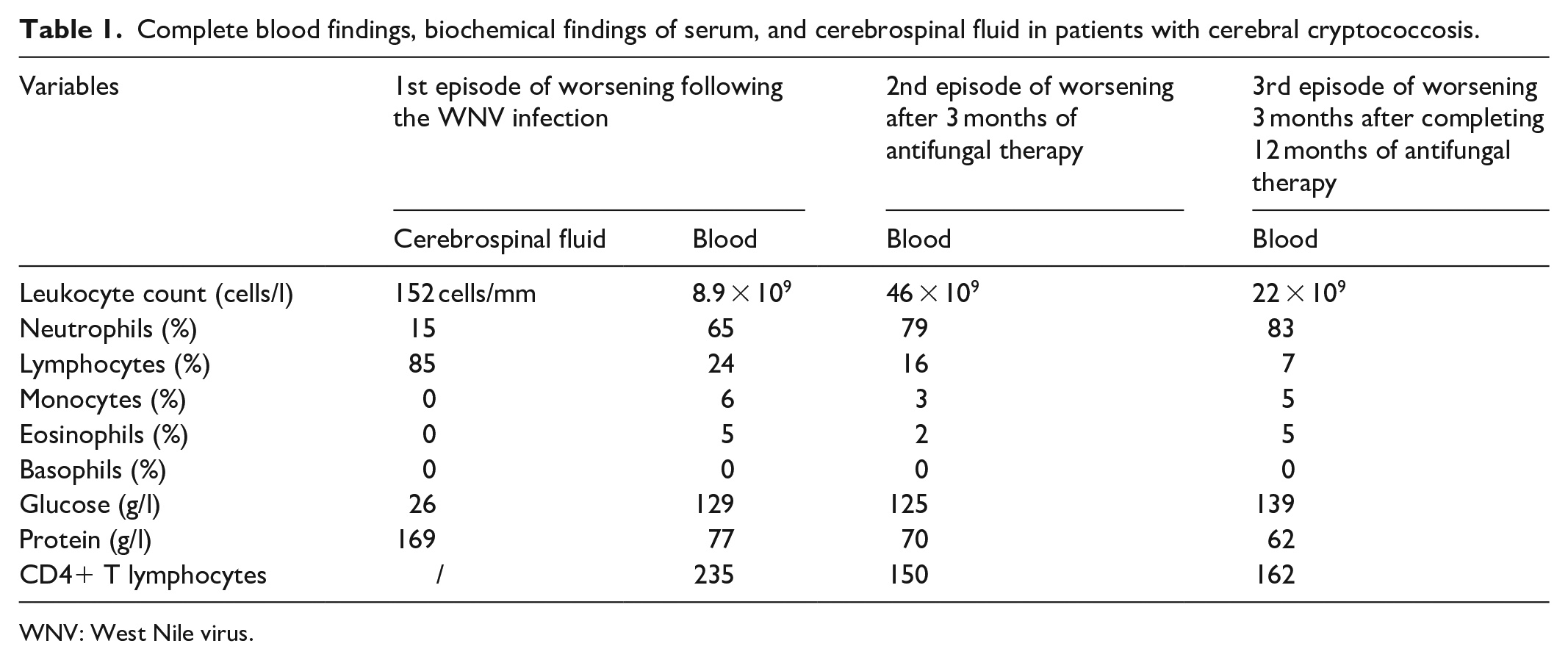
WNV: West Nile virus.
Discussion
Polycythemia vera is a Philadelphia-negative myeloproliferative disease that is frequently complicated by vein or arterial thrombosis, especially in the elderly and patients with JAK2 mutation. Surprisingly, thromboses are frequently found in unusual locations, such as the splanchnic area. They typically occur either before the diagnosis of polycythemia vera was established or in patients during the first period of treatment prior to normalization of the blood picture. 9 For this case, it was interesting to consider that from June to October 2018, mosquito-borne WNV infection was epidemic in 15 European countries, affecting 1670 patients. 6 Because approximately 80% of infected people have no symptoms and thus go undiagnosed and unregistered, the true number of infected people must be much higher. 10 WNV infection in this case was diagnosed by the presence of IgM antibodies against the virus in this patient. Antibodies are known to appear between 3 and 8 days after infection and can be confirmed within 30–90 days. 3 Temperature and headache appeared in our patient by the end of October 2018, which on brain MRI was strongly suggestive of brain cryptococcosis. Given that cellular immunity, including CD4+ and CD8+ lymphocytes, plays a critical role in protecting the body and brain from infection,11,12 we suspect that the patient’s immunity was reduced by previous asymptomatic and undiagnosed WNV infection, allowing cryptococcus to spread to the brain. In addition, in almost all hematological diseases, there is a decrease in cellular immunity, as previously shown. 13 Based on all these contributes, Cryptococcus most likely easily enters the blood–brain barrier, causing meningitis, followed by meningoencephalitis, encephalitis, and ventriculitis, resulting in high temperature, headache, bradykinesia, and bradypsyhia, as seen in the presented patient, a condition that can progress to lethargy, uncontrolled muscle cramps, memory loss, coma, and death. The brain lesions are typically multiple and small in size, but they can occasionally be tumor-like masses measuring more than 30 mm in diameter, known as cryptococcomas. 14 Confirmation of disseminated cryptococcosis is easily diagnosed using the following diagnostic procedures: MRI, cryptococcal antigen in serum, cerebrospinal fluid cultures, and yeast staining in cerebrospinal fluid. Patients with polycythemia vera are extremely unlikely to develop disseminated cryptococcosis. So far, just three such patients have been reported. After receiving immunosuppressive therapy with corticosteroids, tacrolimus, and azathioprine after liver transplantation performed for Budd-Chiari syndrome as a complication of polycythemia Vera, one patient developed disseminated skin cryptococcosis. Fluconazole was used successfully to treat the patient. 15 During ruxolitinib treatment, the second patient with polycythemia vera and JAK2 mutation developed histoplasmosis and disseminated cryptococcosis of the brain. 16 As ruxolitinib is used more frequently in patients with myelofibrosis, the number of reports of cryptococcal cases and other opportunistic infections in those patients is increasing.17,18 The third case of disseminated cryptococcosis of the brain was described in a patient with polycythemia vera and an unexplained monoclonal gammopathy. 5 To the best of our knowledge, our patient is the rare case of disseminated cryptococcosis of the brain in polycythemia vera patients, but the first who developed cryptococcosis following WNV infection, most likely due to impaired cell-mediated immunity. This case indicates that in a patient with hematological malignancies and neurological symptoms, one should always think about the possibility of cryptococcosis infection in everyday clinical work. Although this is very rare, one should always think about the possibility of infection, especially with any immunocompromised patients or conditions that can lead to transient immunodeficiency.
Conclusions
If a polycythemia vera patient develops a headache, fever, and neurological symptoms, disseminated brain cryptococcosis must be considered and diagnostic procedures must be performed. Because the disease can have a severe course and a poor outcome, therapy should be tailored to the individual patient for as long as necessary. Larger brain lesions, known as “cryptococcomas” may require surgical removal.
Footnotes
Author contributions
N.Č. Conceptualization; Investigation, Writing – original draft; J.L. Data curation; D.K. Formal analysis radiology; A.B. Conceptualization, Writing – original draft; V.J. Supervision, Writing – review & editing, Corresponding.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was partially financed by the Ministry of Science of the Republic of Serbia.
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. The investigation was approved by the local Ethical Committee.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
