Abstract
Cold agglutinin-induced hemolytic anemia presents diagnostic and management challenges, particularly in resource-limited settings like Pakistan. A 92-year-old male presented with altered consciousness, fever, weakness, and severe asthenia. Laboratory findings revealed severe anemia, elevated mean corpuscular volume, and deranged renal function tests. The diagnosis was confirmed via a positive Direct Coombs test and elevated cold agglutinin titers. Multidisciplinary management led to symptom resolution, including blood transfusions and immunosuppressive therapy. This case report and literature review emphasize the complexities of diagnosing and managing cold agglutinin-induced hemolytic anemia in Pakistan. By enhancing awareness and understanding of this condition, clinicians can optimize patient outcomes through timely intervention and multidisciplinary care.
Keywords
Introduction
Cold agglutinin disease (CAD) is a rare autoimmune disorder marked by IgM-mediated hemolysis, which leads to the destruction of red blood cells (RBCs) when exposed to cold temperatures. With an incidence of only one per million annually, CAD makes up about 15% of autoimmune hemolytic anemia (AIHA) cases.1,2 Recognizing its significance, both the World Health Organization (WHO) and the International Consensus Classification have acknowledged primary CAD as a distinct diagnostic entity, a decision supported by case studies from Italy published in the journal Histopathology. 3 Primary CAD is a clonal B-cell lymphoproliferative disorder (LPD) and should be clearly differentiated from cold agglutinin syndrome (CAS). While both conditions involve cold-induced hemolysis, CAS often arises as a complication of specific infections, such as Mycoplasma pneumoniae pneumonia or Epstein–Barr virus (EBV) infection, or in association with malignancies, especially aggressive lymphomas. 4 However, infectious agents, such as influenza, varicella, legionella, mycoplasma, and EBV, can also precipitate hemolysis in susceptible individuals. 5 The pathogenesis involves agglutination of RBCs and complement fixation at low temperatures (0°C–5°C) with subsequent warming of the blood (20°C–25°C) triggers complement activation, resulting in hemolysis.
Patients with CAD typically present with symptoms such as fatigue, headache, jaundice, weakness, and cardiac complications, with severity varying on the basis of the extent of hemolysis 3. Concurrent viral infections or febrile illnesses and prior surgical interventions can exacerbate these symptoms. Notably, CAD may coexist with underlying conditions such as systemic lupus erythematosus, determined lymphoma, or infections, collectively termed CAS. 6 Chronic severe anemia often necessitates blood transfusions in affected individuals. Because of the presence of cold autoantibodies, clinicians may encounter challenges in blood grouping and cross-matching. 7 Insignificant and limited data are available, signifying the lack of attention given to the disease burden, new lines of treatments, and severity of anemia, leaving a huge ground for research to optimize patient management and decrease the course of disease.
In this case report, we present the case of a 92-year-old man with CAD and illustrate the clinical manifestations, diagnostic approach, and multidisciplinary management strategies employed in addressing this challenging condition.
Case report
We present a case of a 92-year-old male presented to the emergency department with a 1-week history of altered consciousness, high-grade fever, generalized weakness, and severe asthenia. His medical history was notable for benign prostatic hyperplasia and a right hip arthroplasty 3 years prior due to a pathological fracture. The patient had been exclusively using homeopathic medications and history of multiple blood transfusion. On arrival, he exhibited hypotension (78/49 mm/Hg), a pulse of 94 beats/min, a respiratory rate of 22 breaths/min, normal blood sugar levels (110 mg/dl), and maintained 100% oxygen saturation on room air.
Following stabilization in the emergency department, the patient was admitted to the special care unit, where a Foley catheter and nasogastric tube were inserted. Physical examination revealed lethargy, conjunctival, and palmar pallor with cold, numb, and blue-tinged fingers, but no peripheral lymphadenopathy or jaundice. The abdomen was non-tender, non-distended, and lacked hepatosplenomegaly or masses. Rest of the examinations were unremarkable, including cardio-pulmonary.
Laboratory findings revealed severe anemia (hemoglobin of 2.8 g/dl) with an elevated mean corpuscular volume (MCV), deranged renal function tests, increased C-reactive protein, elevated lactate dehydrogenase (LDH), and hyperbilirubinemia. Notably, haptoglobin and procalcitonin levels were insignificantly low (Table 2). Peripheral blood smear revealed agglutinates, polychromasia, target cells, tear drop cells, and anisopoikilocytosis with rare schistocytes, but no blast cells (Figure 1). A positive urine culture for Candida albicans was identified, whereas the blood culture showed no growth. Serology for viral hepatitis A, E, B, and C and HIV were negative. An echocardiogram was also performed which indicated a low ejection fraction of 40%–45%.
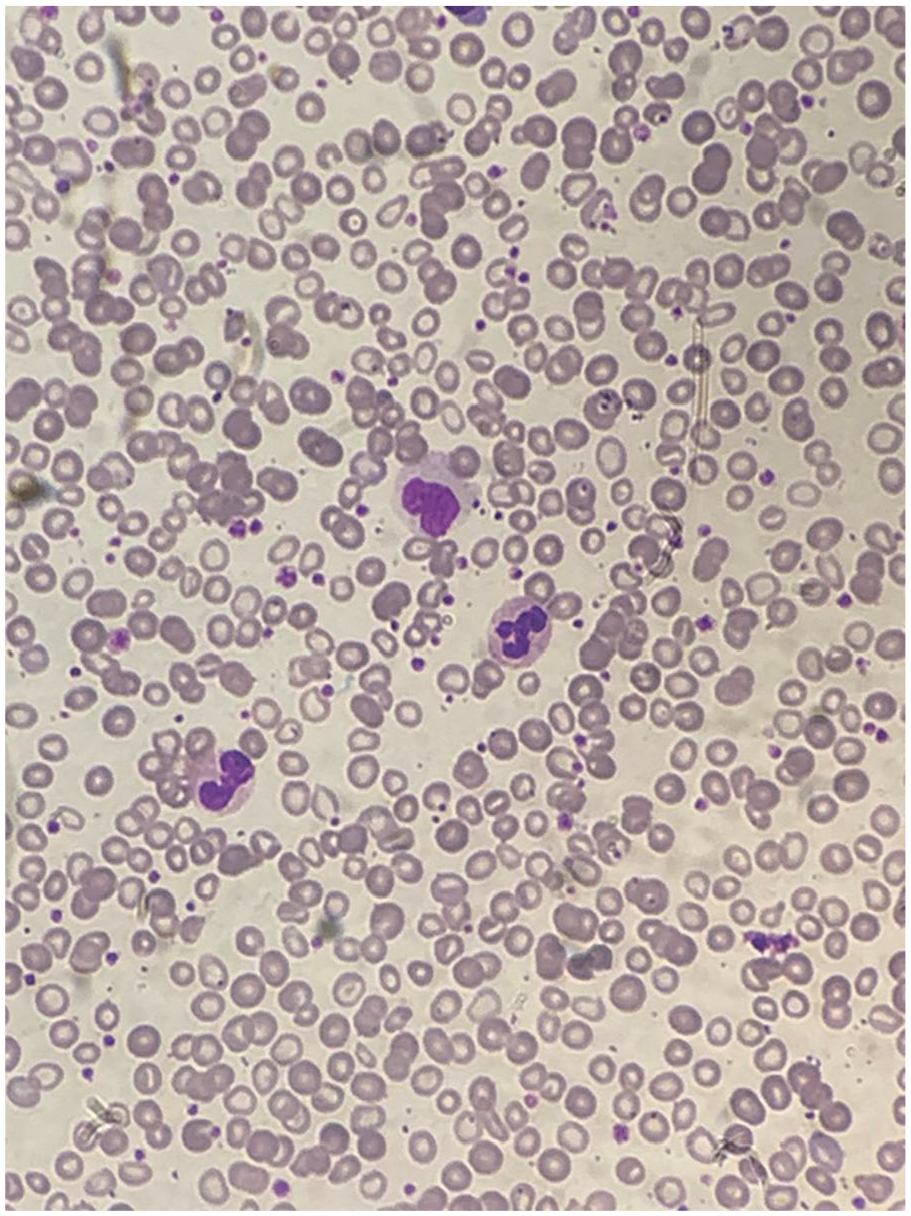
The peripheral blood smear showing polychromasia, target cells, tear drop cells, anisopoikilocytosis with red blood cells agglutinates.
A planned contrast-enhanced chest and abdomen computed tomography scan was postponed because of elevated creatinine levels. The positive (3+) direct antiglobulin test (DAT) or Direct Coombs test with cold agglutinin titers of 1:120, Monocoombs IgG (1+), and Monocoombs C3d (3+) suggested a diagnosis of cold agglutinin-induced hemolytic anemia (CAIHA) (Table 1). The patient received four units of RBCs, diuretics to maintain positive fluid balance during transfusion, intravenous antibiotic meropenam, antifungal fluconazole, nutritional supplements, and intermittent normal saline boluses. Careful measures were taken to maintain warmth, subsequently, hyperkalemia and hypernatremia were managed with kayexalate powder and free water via the nasogastric tube.
Blood parameters to rule out secondary causes.

Employing a comprehensive, multidisciplinary approach, the patient demonstrated notable progress and exhibited favorable responses to transfusions (Table 2). The administration of intravenous methylprednisolone (40 mg every 12 h for 3 days) resulted in near-complete alleviation of symptoms. Subsequent assessments revealed resolved anemia (hemoglobin of 8.9 g/dl on discharge), electrolyte imbalances, and urosepsis, with the patient exhibiting clinical alertness (Table 2). The repeated antibody titer performed after treatment as 1:64 before he was discharged home.
Analysis of blood parameters on admission and discharge.

Outcome and follow-up: Discharge instructions encompassed nursing care, oral steroid regimen, vitamin supplementation, doxycycline administration, as well as directives for maintaining warmth, daily suctioning, and engaging in physiotherapy twice daily. A 1-week follow-up confirmed the patient’s stability, orientation, and a hemoglobin level of 10.2 g/dl. Further follow-up after 4 weeks was recommended for the gradual tapering of oral steroids.
Discussion
CAIHA is an uncommon condition that tends to predominantly affect older individuals, as exemplified by the present case. An insightful retrospective study encompassing 232 patients revealed a mean age at disease onset and diagnosis was 67 and 68 years, respectively, while the average age at the time of the study was 76 years, with a male-to-female ratio of 0.56. 8 This autoimmune disorder is marked by the destruction of RBCs instigated by cold-reacting antibodies. Notably, approximately 90% of CAIHA cases involve immunoglobulin M (IgM)-mediated disease, with IgG- and IgA-mediated instances being infrequently reported. 9 Hemolysis observed in CAD is complement-mediated and can manifest either intravascularly or extravascularly. As blood circulates through the cooler skin and extremities, where temperatures can plummet to 28°C–30°C, cold agglutinins momentarily bind to the surface of erythrocytes. Subsequently, erythrocytes coated with C3b undergo sequestration by macrophages in the reticuloendothelial system, particularly by Kupffer’s cells in the liver 10 . This process culminates in the destruction of these cells, which contributes to extravascular hemolysis. 11
AIHAs are broadly categorized into warm, cold, or mixed types based on the temperature at which autoantibodies bind to surface antigens. Among these, cold AIHAs exhibit further distinctions, encompassing primary chronic CAD, paroxysmal cold hemoglobinuria, and secondary cold AIHAs associated with underlying conditions such as infection or malignancy (Table 3). 4 Despite their relative rarity, with an estimated incidence of 0.8–3 cases per 100,000 individuals per year and a prevalence of 17 cases per 100,000, AIHAs pose a considerable health burden, with an 11% mortality rate in adults. 12 CAD, a subset accounting for approximately 15%–25% of all AIHAs, holds a specific niche within this spectrum.4,6,9 Its incidence is reported to be around one per million people per year, although it is notably underreported in regions such as Pakistan.
Causes of cold autoimmune hemolytic anemias.

The clinical spectrum of CAD spans from asymptomatic cases to severe anemia. Many individuals harbor circulating cold agglutinins in their blood, often oblivious to this condition until they are exposed to cold temperatures. Clinical presentations include common symptoms such as fatigue, weakness, and pallor. The complexity of the clinical picture is amplified by cold-induced circulatory signals, such as livedo reticularis, Raynaud disease, acrocyanosis, and cutaneous necrosis, mirroring the intricacies observed in our case. 13 The consideration of primary chronic CAD is imperative in elderly patients with unexplained chronic anemia, regardless of the presence of associated cold-induced symptoms. In a study by Swiecicki et al., 9 the median age of presentation was reported as 65 years, with acrocyanosis noted in 44% of patients and other cold-induced symptoms in 39%. Additionally, specific cases exhibited hemoglobinuria, signifying intravascular hemolysis.14,15
The clinical features of patients with CAIHA present a diagnostic challenge because they often overlap with warm immune hemolytic anemia. Indirect hyperbilirubinemia, elevated LDH, reticulocytosis, absence of haptoglobin, and the presence of spherocytes in the peripheral blood film characterize the hematological profile. 16 The first two criteria are the most useful in diagnosing AIHA and are based on the DAT, commonly known as the Coombs test, and the indirect antiglobulin test (IAT), also referred to as the indirect Coombs test.12,14 The DAT demonstrates the presence of autoantibodies on the patient’s RBCs and helps define their class, thermal characteristics, and ability to activate complement. The IAT, on the other hand, identifies these autoantibodies in the serum. In more complex cases, the study of the eluate, which involves analyzing the antibodies eluted from DAT-positive RBCs, may be conducted to provide further diagnostic clarity. 12 In our case, the key to diagnosis was a positive Direct Coombs test (3+) along with elevated cold agglutinin titers, a pivotal diagnostic indicator. Ninety percent of cold agglutinins belong to the IgM immunoglobulin class, typically presenting with a titer of 1:64 or higher at 4°C. 17 In this case, the titers were positive at 1:120. These cold agglutinins can be semi-quantitated through titration based on their ability to agglutinate erythrocytes at 4°C. 14 Diagnosing CAIHA necessitates a multifaceted approach involving clinical findings, laboratory tests, and specialized immunological assessments. Importantly, the sample used for cold agglutinin testing must be maintained at 37°C–38°C post-collection until serum separation from the clot. The initial diagnostic step involves the polyspecific DAT aka direct comb test. 15 A positive result prompts further assessment with a monospecific DAT for C3d and IgG. Notably, a negative anti-C3d DAT is unusual in CAD, potentially indicating warm AIHA. 14
Secondary causes, such as infections, autoimmune diseases, and LPDs, must be systematically evaluated to rule out the underlying pathology contributing to CAD. Serum protein electrophoresis (SPEP) and serum free light chain assays are critical in this context, as they help detect monoclonal proteins, particularly IgM kappa, which are commonly associated with LPDs underlying CAD. As highlighted in the study by Sidana et al., the presence of IgM kappa monoclonal protein is a key feature in primary CAD, 18 and the absence of these diagnostic tests in our case, due to the patient’s critical condition and financial constraints, limits the ability to fully exclude these secondary causes. Additional investigations, including chest and abdominal CT scans, are warranted in cases displaying symptoms such as weight loss, hepatosplenomegaly, lymphocytosis, or cytopenias. However, in this case, the CT was deferred because of deranged creatinine levels. This underscores the importance of a comprehensive diagnostic approach tailored to each patient’s unique clinical profile, while also highlighting the challenges posed by patient condition and financial limitations in resource-limited settings.
Managing CAIHA requires a multidisciplinary strategy comprising hematologists, immunologists, transfusion medicine specialists, and support staff, including nurses and pharmacists. 19 Symptomatic anemia is addressed through RBC transfusion, with challenges in obtaining crosshatch compatible blood due to interference from cold agglutinins, potentially causing treatment delays. Patients are advised to keep warm, particularly their head, face, and extremities, and in-line blood warmers are used during transfusions to prevent clotting and occlusions. 19 Unlike warm AIHA, CAIHA does not respond to splenectomy or glucocorticoids. 20 Steroids, particularly corticosteroids, have shown efficacy, with a higher response in patients with low-titer cold agglutinins.21,22
Although no universally effective therapies exist for CAD, in this study, intravenous methylprednisolone played a pivotal role in symptom resolution because of its low antibody titer. Rituximab depletes B cells, inducing partial responses in about 50% of patients after a median of 1.5 months, with relapses typically within a year. 23 Adding bendamustine or fludarabine increases not only response rates but also serious toxic effects like severe neutropenia; however, fludarabine is avoided in most centers due to the risk of secondary malignancy. 24 Eculizumab, a C5 inhibitor, reduces transfusion needs and lowers LDH levels but has had disappointing trial results, offering a modest increase in hemoglobin levels and limited use outside of rare cases of brisk intravascular hemolysis. 25 Sutimlimab, an FDA-approved humanized monoclonal antibody targeting C1s, has shown rapid and sustained effects by preventing chronic hemolysis, significantly increasing hemoglobin levels, and improving quality of life with mild-to-moderate adverse events. 26 However, complement inhibition, including sutimlimab, does not alleviate cold-induced symptoms and requires ongoing therapy. A recent retrospective study, including 19 patients with warm and cold AIHA, reported that daratumumab monotherapy may be an effective and well-tolerated treatment option for refractory AIHA, improving both hemolytic anemia and cold-induced circulatory symptoms. 27 Future trials include the use of Bruton’s tyrosine kinase inhibitors. This case underscores the significance of early diagnosis and identification of the underlying causes of CAIHA.
Limitations
As authors, we acknowledge the challenges in managing CAD in a developing country like Pakistan. Diagnostic tests like cold agglutinin quantification and thermal amplitude testing are limited, leading to prolonged result wait times due to resource constraints. Additionally, postponed contrast-enhanced CT scans due to elevated creatinine levels and the inability to perform EBV and Mycoplasma serology tests, mainly due to their limited availability and patient financial constraints, further complicate diagnosis. Referral to tertiary care centers is hindered by logistical and financial barriers. A bone marrow biopsy was considered but not performed due to the patient’s advanced age and refusal from the attendants, who were concerned about the risks associated with the procedure, such as infection and bleeding. Additionally, the absence of SPEP and serum-free light chain assays due to the patient’s critical condition and financial constraints further limits our ability to confirm an underlying LPD. Without these crucial diagnostics and biopsy confirmation, distinguishing between CAD and secondary causes, or identifying mixed warm components (the most common cause of AIHA), becomes particularly challenging. Accessing treatments such as rituximab and complement inhibitors is challenging due to high costs, limited supply, and regulatory hurdles, exacerbated by patient age and financial constraints. These limitations emphasize the urgent need to improve healthcare infrastructure and enhance diagnostic capabilities while facilitating equitable access to essential CAD treatments in resource-limited settings like Pakistan.
Conclusion
CAIHA is a challenging condition that necessitates a thorough understanding of its clinical manifestations, diagnostic criteria, and appropriate management. The presented case exemplifies the successful application of a comprehensive treatment approach, including blood transfusions, immunosuppressive therapy, and targeted management of associated infections. Timely recognition and intervention are pivotal in optimizing outcomes for patients with CAIHA.
Footnotes
Acknowledgements
None to declare.
Authors contribution
Conception and design: Syeda Mahrukh Fatima Zaidi, Ayesha Amjad, Sajjad Ali Khan. Acquisition of data: Ayesha Amjad, Syeda Mahrukh Fatima Zaidi, Muhammad Umer Khan. Drafting of the manuscript: Ayesha Amjad, Syeda Mahrukh Fatima Zaidi. Critical revision for important intellectual content: Sajjad Ali Khan, Syeda Mahrukh Fatima Zaidi. Final approval of the study: Ayesha Amjad, Syeda Mahrukh Fatima Zaidi, Muhammad Umer Khan, Sajjad Ali Khan. All authors have read and agreed to the final version of the manuscript.
Availability of data and materials
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Research registeration
None. Research Registration is not required. This case report does not detail a new surgical technique or new equipment/technology.
