Abstract
Giant congenital melanocytic nevus often necessitates meticulous planning and multiple treatment stages for complete surgical excision. This report presents a case of giant congenital melanocytic nevus on the back managed through serial tissue expansion and excision. Initially, two expanders were placed at the deep fascia level. Sequential outpatient expansions over 10 weeks were followed by expander removal, partial nevus excision, defect coverage with expanded skin flaps, and simultaneous placement of a new expander. The subsequent single expander expansion over 12 weeks involved a total of 600 mL of saline. After three operations spanning approximately 6 months, 54 cm × 36 cm of giant congenital melanocytic nevus skin, covering 65% of the patient’s back, was completely excised. Serial tissue expansion and excision may be an effective surgical approach for managing dorsal giant congenital melanocytic nevus, reducing the need for multiple surgeries and achieving favorable aesthetic outcomes.
Introduction
The term “giant congenital melanocytic nevus” (GCMN) describes melanocytic lesions present at birth that develop to a diameter of at least 20 cm in adulthood. 1 Surgical removal of these lesions is often preferred due to the risk of malignant transformation (melanoma), challenges in follow-up care, and the significant emotional impact of their unsightly appearance on patients and their families. 2 Tissue expanders, skin grafts, and serial excision are standard surgical procedures suggested for treating GCMN.3,4 Serial excision, a multistep process involving multiple resection surgeries, is particularly effective with low complication risks.5,6 Large grafts or tissue expanders are commonly employed for cases requiring more than three resection stages. Successful tissue expansion in plastic surgery hinges on matching the properties of donor skin, including texture, color, hair follicles, and sensitivity, with those of the recipient’s skin. However, expansion therapy may extend over three to 6 months, depending on the anatomical site requiring reconstruction.7,8 During this period, the expander may cause discomfort and aesthetic concerns, necessitating periodic injections for enlargement. 1 This report aims to present the author’s experience with a combined approach involving serial tissue expansion and excision for treating GCMN.
Case report
A woman in her early 20s presented with a large, pigmented skin lesion extending from the T2 vertebrae to the L3 region, covering the upper dorsal trunk. The lesion measured 36 cm horizontally and 54 cm vertically, with significant pigmentation visible to the naked eye, raising clinical suspicion of a GCMN (Figure 1). To confirm the diagnosis and rule out malignancy, a biopsy via fine-needle aspiration was performed. Histopathological examination confirmed GCMN without signs of malignancy. The patient, in good health, wished to have the nevus removed due to concerns about melanoma and social integration. After discussions with the surgical team, serial tissue expansion, and excision procedures were planned. All surgeries were conducted under general anesthesia with perioperative antibiotics.

A woman in her early 20s presented with a huge congenital nevus in the backside of upper trunk.
The initial procedure involved an elliptical excision within the nevus and the simultaneous placement of two tissue expanders beneath adjacent normal skin. Each expander, with a capacity of 680 mL, was positioned parallel to the nevus border. After meticulous dissection, the nevus was partially resected, and pathology confirmed a compound congenital nevus with extensive subcutaneous involvement (Figure 2). Expanders were inserted at the deep fascia level, with drains for fluid drainage, which were removed once drainage decreased below 20 mL over 24 h. The elevated flap was closed with absorbable sutures in the deep fascia and nylon skin sutures. During subsequent outpatient visits, expanders were filled twice a week to their rated capacity, with additional inflation if the skin flap exhibited adequate biomechanical capacity. Each expander was inflated to 600 mL over 10 weeks (Figure 3).

Histopathology staining of the nevus: Hematoxylin-Eosin staining ((a) and (b)) and S100 Immunohistochemistry staining ((c) and (d)). (a) Nest of melanocytes in the dermis with melanin pigments around the cells. (b) Melanocytes in the dermis with round shape and scant cytoplasm (arrow). Melanin pigments can be seen around the cells (arrowhead). (c) Nest of melanocytes in the dermis showed positive with S100 (arrow). (d) Melanocytes in the dermis showed cytoplasmic and nuclear staining with S100 marker (arrow). Scale bars represent 100 μm. Low to high magnification by optical microscopy: ((a) and (c)) 100×, ((b) and (d)) 400×.

First tissue expansion. (a) Two tissue expanders were inserted on both sides to the border of the giant nevus. ((b)–(d)) Full expansion of the first two tissue expanders.
Two weeks after the last injection, the second-stage operation was conducted to remove the expanders and excise the nevus. The excision involved partial resection from the periphery toward the center, guided by the mobilization of surrounding normal skin—from the shoulder downwards, laterally to medially, and from the bottom upwards. The excision extended full-thickness to the deep fascia, starting at the lesion’s periphery. Over half of the nevus was excised and resurfaced with the expanded flaps (Figure 4). Due to the nevus’s size, a serial tissue expansion procedure was necessary. During the second operation, one expander was implanted, and expansion commenced two weeks later with twice-weekly saline injections. Over the next 10 weeks, the tissue expander was filled with 600 mL of saline without complications. Following full expansion, the expanders were retained for two weeks before removal, and the nevus was resected. Following the third surgery, the GCMN was completely excised without any complications, such as infection or expander exposure. Furthermore, no significant restrictions on shoulder mobility were reported. Satisfactory outcomes were observed during a 1-year postoperative follow-up (Figure 5).
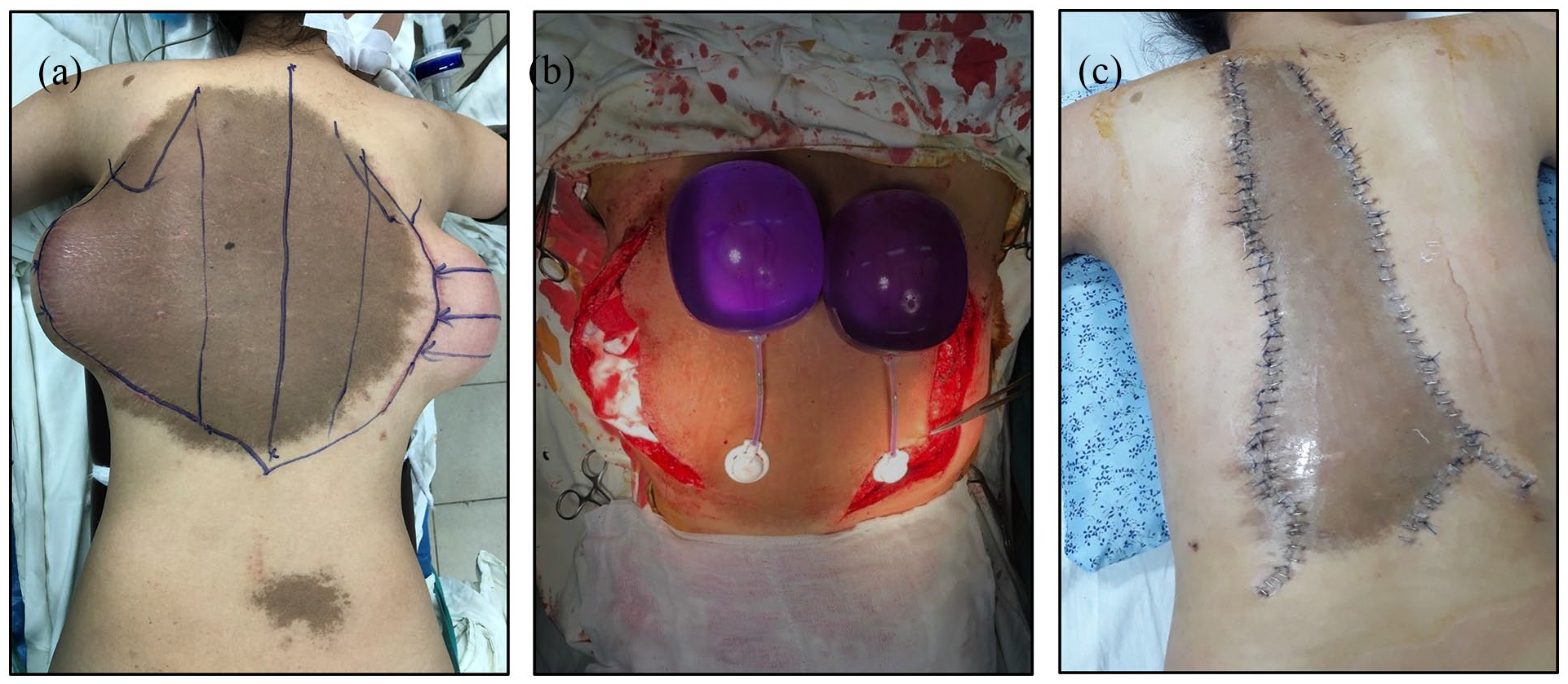
Second-stage operation two tissue expanders removal and partial resection of giant nevus. (a) Preoperative design. (b) Two tissue expanders were removed. (c) Postoperative imaging.

(a) Full expansion of the second tissue expander. (b) Postoperative of the second tissue expander removal and total resection of giant nevus. (c) Final postoperative a year follow-up.
Discussion
Congenital pigmented nevi pose an elevated risk of malignant melanoma development, with nevus size being a significant contributing factor. Complete excision remains the most effective strategy for reducing melanoma risk. 9 Large to GCMN often necessitate multiple surgical interventions such as serial excisions, tissue expansion, flaps, or grafts to achieve satisfactory closure, albeit potentially resulting in visible scarring and extensive changes. 10 However, selecting the appropriate reconstructive method can be challenging due to the limited availability of surrounding skin with suitable tension. Tissue expansion is a widely favored approach, with early expansion showing promising outcomes in various studies.7,9,11 Given the limited research on tissue expansion in clinical practice, we aimed to fill this gap by assessing different expansion techniques, including the number of expanders used, stages of operation required, and the achieved degree of inflation. Despite the scarcity of clinical data on GCMN, we successfully removed the nevus on the patient’s back, capitalizing on the skin’s excellent elasticity and its natural ability to expand under significant stretching force in young individuals. Serial expansion represents an advanced technique in tissue expansion.
Several factors favor serial expansion over single expansion, primarily to acquire sufficient expanded skin. The success of these procedures relies on tissue elasticity and mechanical properties such as creep and stress relaxation. 12 Using multiple tissue expanders often yields better outcomes than single expanders, especially for large nevi over 20 cm in diameter. Despite high inflation volumes, additional procedures are often needed to ensure adequate tissue flexibility. 13 Our clinical experience suggests that serial expansion is safer and more effective for high-risk patients, provided specific anatomical and environmental factors are considered.
Tissue expansion procedures are commonly associated with complications such as infection or expander exposure. Effective management of these complications is crucial to ensure successful outcomes. 14 In our case, we emphasized clear communication with the patient about the treatment plan and conducted frequent follow-up appointments to monitor the expansion site for early signs of complications. It is important to monitor for signs of expander exposure, such as thinning of the overlying skin or visible parts of the expander. Early signs of infection, such as redness, swelling, pain, or fever, should be promptly identified and treated. Using a tissue expander at the back can impact the shoulder joint’s range of motion, depending on factors such as the location, size, and volume of the expander. 15 Expanders placed in the mid-back areas are less likely to restrict shoulder movement directly. The tissue expanders were expanded gradually to minimize sudden increases in tension and discomfort, allowing the body to adapt more easily and maintain better shoulder mobility. If complications occurred, the treatment plan was modified accordingly to address and resolve these issues before proceeding with further expansions.
Throughout three operations over approximately six months, we successfully removed the GCMN from the patient’s back. This was made feasible by the excellent elasticity of the skin and its capacity for multiple expansions under significant stretching force, particularly in a young adult. We partially resected the nevus from its peripheral margins, aligning the excised sections to facilitate optimal skin stretching. Concurrently, we carefully positioned the expanders to maximize their effectiveness. The extent of nevus removal was predetermined by maximizing the stretching of healthy skin from the periphery to the center, ensuring sufficient tension was present at suture closure to promote the biomechanical transformation of healthy skin. 16 To maintain stable wound tension and minimize scarring, we simultaneously executed two procedures: serial tissue expansion to generate adequate expanded skin and nevus excision with suturing of healthy skin margins at the periphery to the deep fascia at the central margins. This approach stabilized wound tension and limited nevus expansion, with intracutaneous sutures employed to mitigate the risk of scar dehiscence.5,11 Despite the inherent risk of complications associated with serial tissue expansion and excision, this combined approach proved efficient, requiring only three surgeries over 6 months to completely eradicate the giant dorsal congenital melanocytic nevus. Each procedure was straightforward, and the patient experienced a smooth recovery without complications.
Conclusion
The serial expansion and excision technique effectively manages GCMN with tissue expanders. This approach is particularly suitable for lesions presenting anatomical challenges or requiring extensive expansion, this surgical approach is characterized by its simplicity, minimizing the need for multiple surgeries while achieving complete excision and yielding satisfactory aesthetic outcomes. Our study findings suggest that this technique does not heighten the risk of complications compared to single expansion methods and may offer more efficient outcomes than many partial resection surgeries, as supported by existing literature. In conclusion, the meticulous management of GCMN through serial expansion and excision demonstrates promise in delivering aesthetically pleasing surgical outcomes for patients.
Footnotes
Acknowledgements
The authors acknowledge the support/partial support of Hue University under the Core Research Program, Grant No. NCM.DHH.2022.03.
Author contributions
Conceptualization: HPL, NTT, and NTNL. Performed the operation: HPL and NTT. Pathology analysis: BSNT. Project administration: NTNL. Writing – original draft: NTT. Writing – review & editing: HPL and NTT. All authors read and approved the final paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval was waived by the authors’ institution.
Informed consent
Written informed consent was obtained from the patient for anonymized information and accompanying images to be published in this article.
