Abstract
As clinical experience with surgical techniques and immunosuppression in vascularized composite allotransplantation recipients has accumulated, vascularized composite allotransplantation for hand and face have become standard of care in some countries for select patients who have experienced catastrophic tissue loss. Experience to date suggests that clinical vascularized composite allotransplantation grafts undergo the same processes of allograft rejection as solid organ grafts. Nonetheless, there are some distinct differences, especially with respect to the immunologic influence of the skin and how the graft is affected by environmental and traumatic insults. Understanding the mechanisms around these similarities and differences has the potential to not only improve vascularized composite allotransplantation outcomes but also outcomes for all types of transplants and to contribute to our understanding of how complex systems of immunity and function work together. A distinct disadvantage in the study of upper extremity vascularized composite allotransplantation recipients is the low number of clinical transplants performed each year. As upper extremity transplantation is a quality of life rather than a lifesaving transplant, these numbers are not likely to increase significantly until the risks of systemic immunosuppression can be reduced. As such, experimental models of vascularized composite allotransplantation are essential to test hypotheses regarding unique characteristics of graft rejection and acceptance of vascularized composite allotransplantation allografts. Rat hind limb vascularized composite allotransplantation models have been widely used to address these questions and provide essential proof-of-concept findings which can then be extended to other experimental models, including mice and large animal models, as new concepts are translated to the clinic. Here, we review the large body of rat hind limb vascularized composite allotransplantation models in the literature, with a focus on the various surgical models that have been developed, contrasting the characteristics of the specific model and how they have been applied. We hope that this review will assist other researchers in choosing the most appropriate rat hind limb transplantation model for their scientific interests.
Keywords
Introduction
The first human forearm transplant in Ecuador in 1964 was a surgical success, but the less effective available immunosuppressive treatment (azathioprine and prednisone) before the development of calcineurin inhibitors likely caused the rejection and graft amputation only 3 weeks after surgery.1,2 In the era of modern immunosuppression, the first successful hand transplantation was performed in France in September 1998. 3 Four months later, in January 1999, the firsthand transplant in the United States occurred, 4 and this recipient continues to do well and is now more than 21 years post-transplant. To date, more than 100 hand transplants have been performed worldwide, 5 and the field of vascularized composite allotransplantation (VCA) now extends beyond the upper extremity and face to abdominal wall, urogenital and uterine transplants. 6
While the success of upper extremity transplantation has exceeded early expectations, there are multiple challenges to be addressed, such as rejection and vasculopathy and the impact of environmental factors.7 –12 The number of VCA transplants performed each year is still quite low (seven per year on average from 2014 to 2018); 6 due to multiple ethical and logistical challenges, potential treatments and investigative studies are best approached in an experimental animal model. Rodents offer a uniquely resilient, malleable, and cost-effective research method. Here, we review the strengths and weaknesses of the various rat hind limb VCA models, and how they might be used to address unanswered questions in clinical cases of VCA.
The first experimental models of hind limb flaps
In the 1960s and 1970s, clinical microsurgery underwent several major advances, culminating in the first digital anastomosis 13 and first arm replantation 14 which laid the groundwork for the field of reconstructive transplantation. The first rodent VCA models developed in the 1970s focused on the feasibility of performing successful microsurgical transplantation in rats. Harashina and Buncke 15 explored the need for graft wash out and perfusion in experimental autogenic rat hind limb amputation and replantation. In their model, they amputated the hind limb at the level of the femoral shaft, following replantation using intramedullary nail and microvascular anastomosis (without nerve anastomosis). They demonstrated good success rates and surprisingly found that no perfusion of the donor graft resulted in better outcomes than perfusion with either heparinized saline or the kidney organ preservation fluid used at the time. Success rates were 90% in animals with grafts that were not perfused, falling to around 50% with perfusion. 15 Shapiro and Cerra 16 described a similar replantation model with a longer 4-month follow-up and a more proximal amputation of the donor graft. Donor graft amputation below the inguinal ligament and pelvic girdle allowed the inclusion of anastomosis of sciatic and femoral nerves, as well as preservation of the profunda branch of the femoral vein. This group was one of the first to document whether any functional movement of the grafted limb returned, although they did not check for a sensory response or pain withdrawal response. No movement of the grafted limb was observed; however, the authors noted good passive movement of the joints at 4 months post-transplant.
Advent of allogeneic hind limb transplantation
In 1979, K Doi, 17 an orthopedic surgeon from Yamaguchi University in Japan was one of the first to perform allogeneic hind limb transplants in rats. In this model, a mid-thigh amputation was performed followed by bone fixation using K-wire and muscle approximation, followed by vascular and sciatic nerve anastomosis. The donor animals were outbred Wistar rats, and recipients were inbred Fischer 344 (F344) rats. The animals were relatively small at 200 g. This was also the first report of using immunosuppression to prevent graft rejection. Doi tested different immunosuppressant drugs such as azathioprine, 6-MP and prednisolone in various combinations and their effect on the allograft survival. The technical success rate was similar to previous syngeneic studies, but the allogeneic survival rate under immunosuppression was only 3 weeks, 17 strikingly similar to the first clinical hand transplant performed 15 years earlier. 1 As such, although sciatic nerve repair was performed, it was not possible to evaluate function of the grafts.
Titration of immunosuppression
Black and colleagues18,19 were the first to report prolonged rat limb allograft survival with Cyclosporin A (CsA) immunosuppression. They used Lewis/Brown Norway (BN) F1 hybrid rats as donors and Lewis rats as recipients and performed an orthotopic transplant similar to Doi, 17 except they repaired the femoral nerve in addition to the sciatic nerve. The rats were treated with a dose titration of CsA for 20 days when it was discontinued and animal were monitored until rejection occurred. At lower doses (2–8 mg/kg/day CsA), rejection followed approximately 20 to 35 days post-transplantation. Prolonged survival was found with a dose 25 mg/kg/day CsA, where rejection occurred approximately 100 days after transplant. Of note, the primary criteria for rejection was reported as a drop below 32° C in the internal temperature of the transplanted limb which occurred at 101 days in the experimental group and at 18 days in the untreated control group. 18 In addition, the investigators noted recovery of response to pain stimuli approximately 2 months after the transplant.
Lipson et al. 20 described syngeneic and allogeneic 21 orthotopic distal-femur hind limb transplantation; unlike previously described models, this graft contained the distal femur, knee and leg, but without the foot and without the donor’s skin. The focus of their studies was a model for joint reconstruction. The authors performed a complete major histocompatibility complex (MHC) mismatch (BN to F344) in the absence of immunosuppression. Surprisingly, they were able to follow grafts for up to 1 month, which is a longer survival than expected for this strain combination without immunosuppression compared to other reports. 22 It is possible that the lack of donor skin may have contributed to the increased survival time of the graft in the absence of immunosuppression. Bone union was delayed but did occur. In addition, thrombosis of the large vessels did not occur until relatively late in the rejection process. The concept of transplanting vascularized skeletal tissue without skin provides unique opportunities to investigate the role of skin in VCA graft rejection. Shortly thereafter, Kim et al. 23 performed similar experiments, but with a model that included the donor skin of the lower limb and foot. This study validated the use of CsA in allotransplantation of rat limbs and also tested other conventional immunosuppressive agents such as azathioprine and prednisolone. In 1989, Arai et al. used the same orthotopic model with Brown Norway as donor and Fischer 344 as recipient to explore immunosuppression of limb allografts using FK506. This group was the first to report long-term survival of over 300 days, but did not comment on functional outcomes despite the fact that sciatic nerve repair was done. The model was also notable in that less donor skin (only skin on foot) was included.24,25 These early studies in the 1970s and 1980s demonstrated that the rat model was technically and immunologically well-suited as an experimental model of VCA and had the additional benefit of being cost- and resource-effective.
In 1991, Lee et al. 26 investigated the relative immunogenicity of the various tissues of the hind leg in an elegant paper that used six different surgical models to compare the allogeneic rejection pattern of tissue compartment in isolation as well as an intact hind limb. A strong MHC mismatch (Buffalo to Lewis) was used. Fluorochrome DCAF was injected post-operative day 1 to monitor viability of bone. Five separate models of vascularized grafts of isolated skin, subcutaneous tissue, muscle, bone (isolated knee joint) and blood vessels (isolated 1.5 cm segments of the femoral artery and vein as interpositional grafts) were connected to the recipient femoral vessels. A sixth model of heterotopic hind leg with no nerve repair or bone anastomosis was used to study rejection of intact tissues. Three major groups were studied: Buffalo to Lewis (allogeneic), Lewis to Lewis (syngeneic) and Buffalo to Lewis (allogeneic) treated with CsA. In addition, non-vascularized skin and bone allografts without immunosuppression were also performed in two smaller groups. Briefly, the investigators were able to establish a hierarchy of sensitivity to rejection for the various models and found differences between cellular and humoral immune responses. While humoral responses reflected the expected hierarchy of strongest responses to skin, subcutaneous tissue, intact limb, muscle and then vessel as the least antigenic, a different pattern was observed for cellular responses. Isolated muscle, and not skin, elicited the strongest cellular immune response. Lipson et al.20,21 had previously reported that vascularized grafts showed superior survival to non-vascularized grafts in the absence of immunosuppression, whereas results from this study suggested increased rejection when isolated non-vascularized skin and non-vascularized bone grafts were compared to similar vascularized grafts. However, the reduced survival of non-vascularized grafts in the Lipson reports were due to overwhelming sepsis and not rejection. The larger amount of necrotic tissue in the avascular grafts of the Lipson reports may also have contributed to the different findings. Lee et al. compared isolated grafts and also checked the magnitude of the anti-donor cytotoxic T-cell response, which was increased in the vascularized grafts. The results shown by Lee et al. also suggested that the intact limb in general was less immunogenic than the isolated tissues. 26
Subsequently, Benhaim et al. 27 tested a new immunosuppressant (RS-61443) also known as mycophenolic acid (MMF) in comparison to CsA using an orthotopic BN to F344 model and a surgical procedure similar to Doi. 17 Animals were followed for 32 weeks. The CsA group appeared to have more rejection and morbidity than the MMF group which was relatively free of rejection. 27 Jones et al. 28 used the same model to compare limb transplant survival in rats treated with CsA, MMF, and FK-506. This report confirmed that FK-506 (tacrolimus) was superior to the other two agents in preventing rejection of the skin component, with significantly improved average graft survival times of 296 days vs 61 days and 43 days for CsA and MMF, respectively.28,29 While significant survival of over 300 days was obtained and nerve repair was performed, the authors did not comment on graft function. Also in 2001, Foster et al. performed the first studies using tolerogenic stem cells to achieve graft survival in the absence of systemic immunosuppression. They established mixed allogeneic chimeras using bone marrow transplantation (BMT) from donor ACI rats to recipient inbred Wistar Furth (WF) rats. Twelve months after chimerism was established with the BMT, an ACI hind limb transplant was performed using the mid-femur orthotopic model. The mixed allogeneic chimeras induced by BMT showed no sign of rejection of donor limb allografts, and the tolerance was donor specific, as third-party skin grafts were rejected. In addition, this study documented return of graft function with weight bearing, toe spread and sensory re-innervation returning after 60 days in the majority of animals. 30 This study demonstrated that donor-specific tolerance can be achieved in a vascularized composite allograft after previous induction of durable chimerism.
At this point in the evolution of rat hind limb models, prolonged graft survival has resulted in tacrolimus being the most commonly used immunosuppressant for rat VCA models, although CsA is still used as a reliable immunosuppressant. 31 That said, the usefulness of CsA in experimental models of VCA is diminished by the fact that all clinical VCA programs base immunosuppression protocols on tacrolimus, and not CsA. A recent study by Hautz et al. in 2018 showed that an early, transient treatment with anti-lymphocyte serum (ALS) combined with a low dose of tacrolimus (0.3 mg/kg/day) also leads to prolonged graft survival, which was further augmented by injections of anti IL-1β. 32 A summary of the published doses, administration frequency, and time to first rejection event for each immunosuppressant described here can be found in Table 1.
Immunosuppressive regimens following hind limb VCA. a
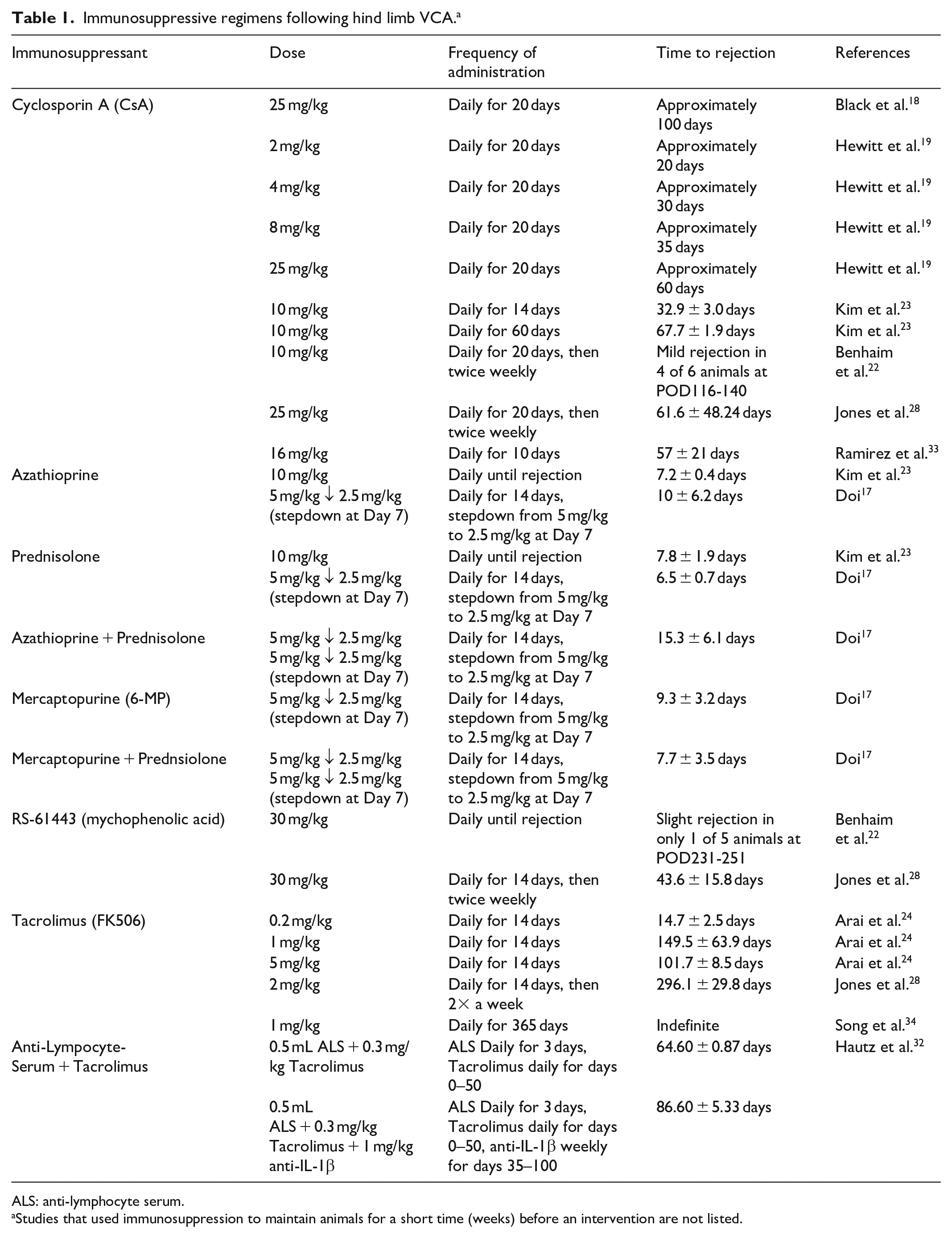
ALS: anti-lymphocyte serum.
Studies that used immunosuppression to maintain animals for a short time (weeks) before an intervention are not listed.
Heterotopic models
Until the early 2000s, with the exception of the heterotopic hind leg model of Lee et al., 26 most reports focused on an orthotopic graft with most of the hind leg and repair of the sciatic or sciatic/femoral nerves included, although function was often not documented. In 2001, Liao et al. 35 presented a simplified modification of heterotopic hind limb transplant in a syngeneic Lewis rat model. Their procedure was the first to introduce an osteomyocutaneous (OMC) flap model, which decreased the graft mass by including only the disarticulated tibia and fibula rather than the entire leg and utilized only related muscles and their associated skin. The graft was anchored to the surface of the recipient gracilis muscle. In 2004, Nazzal et al. described an allogeneic hind limb transplant where a mid-femoral osteotomy was performed approximately 5 mm above the knee joint and another osteotomy at the level of the ankle joint, above the tarsus. The graft included the adjacent musculature inserted to distal femur and proximal tibia and fibula, not including gastrocnemius muscle that was removed in order to decrease the size of the implant. The graft was inserted in the recipient groin area in a subcutaneous position, with donor skin externalized to monitor viability. 36 The focus of these studies was immunological outcomes and effects of immunosuppression, rather than graft function. The reduced surgical complexity and morbidity of the heterotopic model lends itself well to these type of studies. In 2005, Ulusal et al. described transplantation of the entire hind limb, including its skeletal and musculature components, but with much of the skin on the limb removed. In this model, the hind limb was disarticulated at the hip joint. The graft also included an epigastric cutaneous flap, albeit larger in size than that used by Nazzal et al., 36 and the surgeons also insert the graft in the inguinal area. 37 These models left the native recipient limbs intact.
These studies suggest that if graft function is not a focus of the study, heterotopic transplants are easier to perform, have lower ischemia times and result in a less severe surgical insult to the recipient. Precise anatomic reconstruction is not needed, and reattachment of muscle, nerve, and bone component is not required. Because bone fixation is not required, the problems encountered in orthotopic models of femoral bone length discrepancies after fixation and the negative effect on vascular anastomosis are irrelevant. There is less physiological stress as the native limb amputation is not required. Investigators reported ischemic times reduced by approximately 30%–58% using the heterotopic model compared to the orthotopic hind limb model.36 –38 In addition, higher success rates (94.4% vs 66.7%) were observed with the heterotopic model in direct comparison to the orthotopic model. 38 However, many of these comparisons would likely be impacted by the surgical experience of the group, and the superiority of the heterotopic graft to orthotopic grafts with respect to morbidity has been challenged. In 2012, Sacks et al. published outcomes of a dual surgeon orthotopic technique (N = 60) to a traditional single-surgeon orthotopic rat limb transplant (N = 15). 39 The technique allowed for two animals to be transplanted at the same time and resulted in ischemic times similar to that reported for heterotopic models, with morbidity and mortality equivalent to or better than outcomes reported for heterotopic models. In addition, the authors noted that autophagy was rarely seen unless the graft was necrotic, unlike their experience with heterotopic grafts. They hypothesized that the lack of innervation in heterotopic models increased the potential for self-mutilation. The authors also discussed a cost savings of the dual-surgeon approach. However, the report did not factor in the labor costs of two highly skilled microsurgeons, or the fact that many labs have difficulty retaining one highly skilled microsurgeon, let alone two. More recently, studies utilizing cuff techniques for vascular anastomoses in rodent orthotopic models have reported significantly reduced ischemic time for the procedure, offering another possible avenue for researchers interested in full hind limb transplantation.40,41
Open-bone versus closed-bone models
An essential difference for immunological studies between the models described in this review lies in the bone compartment. For example, while the Nazzal et al. 36 model involved osteotomy of the long bones, the model used by Ulusal et al. 37 did not interfere with the bone cavity. This is an important difference, as transection of the long bones exposes the marrow, potentially leading to excessive bleeding, bone marrow embolus, infection and graft loss. 37 The loss or reduction of bone marrow viability or stem cell niches relates to the important concept of allograft tolerance and may affect the interpretation of results with this model for the induction or study of donor-specific tolerance. Multiple groups have investigated the use of closed-bone models to address immunoregulation questions. In 2006, Ozmen et al. 42 suggested that a vascularized disarticulated closed femur isograft with an epigastric skin island could be used to address bone marrow–based tolerance studies without the need for immunosuppression. The authors used the model to test a combined 7-day αβTCR plus CsA treatment protocol. The combination of vascularized bone and skin had longer survival times (61–125 days) than skin alone (43–61 days) suggesting a beneficial effect of the vascularized bone. It would be interesting to determine whether an open vascularized bone model would change observed survival times reported in this study.
Ulusal et al. discussed the advantage of a “closed-bone” model, in which a bone osteotomy is not performed. 37 Previous studies suggested that the bone marrow inside the transplanted hind limb had an impact on prolongation of allograft survival.30,43,44 Tsuchida et al. showed that skin allografts in rat recipients that received limb allograft (containing bone marrow cells) survived much longer than recipients that received VCA not containing bone marrow cells as well as recipients that received bone marrow cells alone. The authors concluded that bone marrow cells contained in a closed-bone model of limb allotransplantation may assist in the induction of tolerogenic activity. 43 Other authors raised the concern that “open bone” model might have an altered or reduced tolerogenic activity, as osteotomy and intramedullary fixation might alter the quantity of bone marrow. 45 Moreover, they support heterotopic models for chimerism-based tolerance studies because, by keeping the bone closed, researchers can ensure that a relatively constant amount of bone marrow is introduced. Adamson et al. 46 designed a modified heterotopic hind limb model to study tolerance to VCA. This report confirmed previous studies that mixed chimerism induces tolerance to the highly antigenic tissues of VCA and examined the effect of T cell depletion of donor marrow on the incidence of graft versus host disease (GvHD). In this study, the femur was divided at the distal shaft and the tibia was cut at the level of the fibular bifurcation. Following curettage and flushing of the bony medullary cavity, the flap was inset and fixed in the gluteal region of the recipient and the vascular pedicle passed through a subcutaneous tunnel into the inguinal area where it was anastomosed. The authors concluded that this heterotopic model was superior to previous orthotopic hind limb models for the study of mixed chimerism tolerance based on fewer complications and reduced animal loss. The authors reported that by curetting and flushing away the graft’s bone marrow compartment, significantly reducing the presence of host reactive T cells in the graft, GvHD was avoided in all recipients. 46 However, a control group that received T-cell depleted bone marrow transplants and a heterotopic donor graft that had bone that was not curetted and flushed was not performed. The authors stated that previous transplants in this model suffered from GvHD, but the animals from the previous study received more bone marrow in the form of an orthotopic hind limb, 47 not a heterotopic graft with a much smaller bone component. In 2008, Chang et al. 45 suggested that a drawback in the previous rat hind limb models was the large percentage of bone marrow resident in the graft, whereas there is a relatively small percentage in clinical human hand transplant. To address this, the group performed a modified flap with a lower percentage of bone marrow, since only two-third of the femur was recovered. An additional cited benefit was reduced operative and ischemic times. Zhang et al. 38 suggested a modification in which the donor whole leg was inset and fixed to the inner aspect of the recipient leg, without bony fixation. However, this model might be associated with disadvantages of friction trauma as the transplanted limb is being dragged, as well as the potential risk to the vascular anastomosis via shearing trauma. To date, there are no studies directly comparing the levels of chimerism achieved on open- versus closed-bone VCA models; the chimerism and tolerance experiments discussed above, while informative, remain somewhat speculative and deserve further investigation.
Functional studies in orthotopic hind limb transplants
Interestingly, although many groups that performed orthotopic limb transplants also repaired at least the sciatic nerve, the majority failed to document or report functional outcomes. One of the earliest reports documenting graft function was Shapiro and Cerra, 16 in which the authors monitored the transplanted rats for any return of motor function; while passive movement of the joints was maintained, no movement was observed in any of these animals. Performing and monitoring VCA limb transplants, especially in allogeneic models where immunosuppression must also be given, is a time-intensive endeavor; insufficient resources to perform the additional monitoring required to document function may be the cause of this lack of reporting. A few groups were careful to comment at least briefly on functional outcomes. Lipson et al. 20 described normal joint function and bone growth following a mid-femoral orthotopic syngeneic transplant. Benhaim et al. 22 observed toe spread and ambulatory weight bearing in an allogeneic mid-femoral transplant procedure, as well as clinically normal protective sensation, while knee function was regained following a mid-femoral allotransplantation study conducted by Arai et al. 25 Foster et al. 30 described gross motor and sensory re-innervation in a model of chimerism following mid-femoral allograft. It is important to note that all successful functional outcomes described above occurred in surgical models utilizing mid-femoral anchoring of the grafted hind limb. Some groups focused on this important issue. Yeh et al. 48 described a hind limb transplant procedure in a syngeneic model that used a pin and cement method to increase stability of the fixation. In this model, researchers also repaired the tibial, peroneal and sural nerves and did not connect the saphenous nerves as a control. This carefully performed study used cutaneous pinch of the skin areas served by these nerves, as well as walking track analysis to look at functional recovery over 4 months. Excellent results were obtained, with sensory recovery for all three connected nerves in six out of six animals and walking track analysis that ranged from near normal to more than 30% of normal. In 2005, Song et al. used the standard orthotopic model with sciatic nerve repair only in an allogeneic and syngeneic model and also performed extensive functional tests including cutaneous reaction test, gait analysis, and electromyography (EMG) and nerve conduction recordings. Interestingly, there was little difference in recovery times of the allogeneic versus syngeneic group, with some recovery in all animals occurring from months 1 to 6 for walking analyses and between months 1 and 9 for electrophysiological studies. 34
Following up on these early studies on functional outcomes in hind limb transplantation, Tsuji et al. 49 explored the effect of prolonged ischemic time on muscular atrophy and nerve regeneration in mid-femoral orthotopic syngeneic transplants. This group’s work took advantage of green fluorescent protein transgenic animals to visualize the donor sciatic nerves regenerating across the recipient/donor nerve anastomosis. The investigators focused on the effect of ischemia on nerve recovery as well as muscle atrophy in the graft. No other functional tests were performed. With the exception of Shapiro et al. in 1978, 16 none of the previously described functional studies performed addressed the issue of physical therapy, which is known to be crucial for success of clinical hand transplantation and replantation. 50 In clinical hand transplantation, failure to perform physical therapy in the first 3 months is associated with significantly reduced graft function.
In 2007, Bueno et al. 51 demonstrated that training or physical therapy of animals who have received an orthotopic graft significantly affects functional outcome. Post-transplant training on a treadmill resulted in resolution of “drop foot,” where the animal bears weight and walks on the dorsal surface of the foot. With training, many animals recover normal gaits using the plantar surface. This study was performed with a syngeneic model, but results are likely applicable to allogeneic models as well. More recently, Liu et al. 52 demonstrated in a model of rat sciatic nerve repair that misdirection of the regenerating axons can be mitigated using tabulation repair with a Chitosan/poly(lactic-co-glycolic acid) (PLGA) nerve guidance conduit. The authors argue that misdirection is unavoidable after nerve anastomosis via epineurial or perineurial suturing, regardless of whether direct end-to-end suturing or nerve autografting is used; their report is the first direct evidence that nerve guides will mitigate axonal misdirection and improve function recovery. 52 This technique may improve functional outcomes in rat hind limb transplants going forward.
Alternative rat VCA models applicable to limb transplantation
Other VCA models have been developed that address the mass or make up of the graft on immunologic processes following allotransplantation. In 2014, Ramirez et al. explored the influence of vascularized bone marrow content on VCA survival by using a novel combined flap model in rats incorporating full-thickness hemi-abdominal wall and a hind limb OMC flap. 33 The authors concluded that grafts containing vascularized marrow versus abdominal wall grafts were associated with tolerance induction. This effect appeared to be lost by the combination of limb VCA with abdominal wall grafts, suggesting graft mass had an effect on tolerance induction. However, it was not clear that tolerance had been achieved in the study, and conclusions may have only been valid for prolongation of the grafts versus true tolerance induction. 33
The skin island in a VCA flap is a key component to address mechanisms of graft acceptance and rejection in clinical VCA. Skin appearance, as well as re-growth of hair is a clinical sign of healthy perfusion; conversely, poor skin appearance or loss of hair indicate possible rejection events within the transplant. Unlike solid organ transplants that can be monitored by different lab tests, VCA has no such marker; clinicians rely heavily on the skin appearance to assess graft health. Furthermore, the skin island makes it possible to take a small skin biopsy at multiple time points during the experiment, documenting the progression of rejection. In addition, imaging of superficial vasculature with multiple methods such as Doppler or high-resolution ultrasound is possible when the flap has a skin island. Several experimental models incorporated a minimal skin component, which intuitively should result in less immunogenic burden on the graft25,28,45 and limit the potential damage to the transplant’s skin by friction trauma and self-mutilation. A number of researchers have addressed this issue by designing the skin component as an island flap that was inset in the groin area. In this manner, the skin is protected and the animal’s ambulation is less affected.45,46,53
Arav et al. 54 published a novel use of rat hind limb models to study potential cryopreservation of intact vascularized composite allografts. If successful, this technique could potentially revolutionize the field of VCA, and indeed organ transplantation by allowing the development of organ banks, similar to blood and stem cell banks. The investigators developed a protocol for cryopreservation and transplantation of rat hind limb VCA by using a mid-femur heterotopic syngeneic model. 54 Two methods of cryopreservation were used. Directional freezing (DF) is a procedure where the graft is placed in tight contact with two brass blocks as controlled thermal conducting masses in a freezing solution containing 10% dimethyl sulfoxide (DMSO). The graft was then inserted in a cryobag with additional freezing solution and stored at −80 C for 7–30 days. The investigators also used vitrification, where the graft was manually perfused for 2 min with a series of solutions with increasing percentages of DMSO, from 1.6% to 25%. The graft was then sealed with minimal air in a cryobag and inserted into liquid nitrogen (LN) slush and then stored in LN for 7–30 days. Remarkably, when the grafts were thawed and transplanted on a syngeneic Lewis recipient, investigators noted that the vessels of the cryopreserved grafts were indistinguishable from fresh grafts, and anastomosis was performed without difficulty. The animals were followed for only 3 days, but no obvious signs of ischemia were noted, and the grafts bled even distally upon removal on day 3. 54 These early results, if successfully reproduced and extended, would significantly change the field of VCA.
In solid organ transplantation, the graft is usually protected from direct environmental influences, with the exception of lung transplants. VCA grafts containing skin are in constant contact with the environment, including toxins, ultraviolet (UV) irradiation from the sun, thermal burns, excessive cold and trauma in the form of direct injury or vibrational trauma. Anecdotal evidence in clinical hand transplantation has suggested that these types of traumatic insult can trigger alloimmune rejection episodes. Recently, Messner et al. 55 developed a rat hind limb transplant model to address the question of whether vibrational trauma can induce graft rejection. The investigators performed an orthotopic allogeneic transplant and developed a device to deliver repetitive 10-minute sessions of mechanical irritation of the planta pedis of the transplanted hind limb, using a Sonicare™ electric toothbrush. In fact, compared to syngeneic controls, which also sustained some dermal injury from the treatment but healed quickly, allogeneic transplants did not heal well and progressed to grade III rejection in some animals. Of note, the authors also included syngeneic transplants treated with tacrolimus to control for drug toxicity. This is an excellent example of how the rat hind limb model can be used to quickly address questions that have been raised in the clinic.
Discussion
Large animal models such as non-human primates, canine and swine are the gold standard for translational research. These models enable safety and efficacy studies in novel treatments of transplantation before use in humans. 56 However, these models suffer from significant cost limitations and share many of the same barriers to clinical translation as rodent models. 57 Clinically relevant or even higher doses of immunosuppressant drugs such as tacrolimus are required to maintain the grafts; the daily dosing can be cost prohibitive and may also result in unacceptable toxicity. Rodent models offer the advantages of greater efficiency of cost and relative ease of genetic manipulation. 58 Experimental groups with sufficiently large N to power statistical analysis of multi-arm studies are more feasible in rodents and require fewer resources for handling, transportation and housing. Furthermore, the time frame for experiments with rodents is significantly shorter compared to models using swine and non-human primates.59,60
Mouse VCA models have also been used extensively.40,61 –63 Mice represent the gold standard in basic immunologic research because there are a greater number of genetically defined inbred and knockout strains, as well as more commercially available monoclonal antibodies and molecular probes. Based on previous work64,65 in rat liver transplantation, researchers developed similar non-suture “cuff technique” for mouse limb transplantation. Several groups have used this technique to develop successful orthotopic and heterotopic limb transplant models in mouse. 66 Sucher reported 62% success rates in orthotopic models and 90% in heterotopic models. 40 Kim et al. 41 modified the cuff technique and used the abdominal aorta for anastomosis because its diameter is 2–3 times larger than the femoral artery. These publications present an elegant solution for super-microvessel (0.2–0.4 mm) anastomosis and renewed interest in mouse VCA models. However, another consideration in mouse models is the physical fragility of mice. The mouse is less tolerant of surgical trauma than the rat, especially when a long procedure such as hind limb VCA transplant is performed. As was shown by Zhang et al., 63 a large percentage of experimental mice were lost to hypovolemic shock in the peri- or post-transplant period following the technically challenging non-cuff super-microsurgical anastomoses. While these challenges as well as the technical skill required for super-microsurgery should be taken into consideration when choosing a model for VCA research, the availability of more diverse strains and reagents in mouse models is attractive.
The development of rat hind limb transplant models was first investigated by Harashina and Buncke 15 in 1975 and has evolved in a number of areas over the last 45 years. Table 2 summarizes the evolution of rat hind limb VCA models, as well as the research trends that led to various improvements and modifications. As the field progressed, experimental models evolved with clinical VCA practice and adapted to the changing needs of basic and translational research. The first models concentrated on the technical microsurgical challenge, and once achieved, the models were modified to focus on the immunobiology of VCA and the methods required to control rejection. As graft survival, rather than graft function was the focus of studies designed to explore the effects of mixed chimerism and tolerance, the less complicated heterotopic model was often chosen. The most recent models further refined the approach to place the skin island in a well-protected area, reducing post-transplant morbidity. Heterotopic grafts are also ideal to answer questions directed at vasculopathy and chronic rejection, as well as cellular and humoral rejection.
Rat hind limb transplantation model.

ACI: ACI rat; WF: Wistar Furth rat—inbred rat strain based on Wistar rat; Wistar: outbred rat strain; SD: Sprague–Dawley rat; CsA: Cyclosporin A; BN: Brown Norway rat; DA: Dark Agouti rat; Lewis: Lewis rat; GFP-Tg: green fluorescent protein transgenic rat; MMF: mychophenolate mofetil; VCA: vascularized composite allograft; OMC: osteomyocutaneous; F344: Fischer 344 rat.
Extensive summary of the rat hind limb VCA models utilized throughout the years. Note that this table focuses on changes to the surgical procedure and design of the rat hind limb model, as well as outcomes and advancements in the field. It does not include all of the very important rat hind limb transplantation studies which used surgical models combined with different tolerance induction protocols or biological or pharmaceutical interventions that investigated mechanisms of graft rejection, as these have been discussed elsewhere in this review.
Most studies using an allogeneic model also performed syngeneic models as a control.
Harashina and Buncke did not designate strain, but photos show a hooded rat, likely the outbred Long–Evans strain.
The focus of this review is on rat hind limb models for VCA research. However, as the field continues to grow, models focusing on forelimb function in the rat are being developed.67,68 Kern et al. developed a rat allogeneic (BN to Lewis) midhumeral forelimb transplant model that includes cooptation of the median, ulnar and radial nerves. 67 Importantly, this model allowed the investigators to study recovery of not only grip strength but also of behavioral function. Taking advantage of the propensity of rats to eat a piece of Fruit Loops™ cereal while rolling the treat in a circle using movement of elbow, forearm, wrist and paw, they have developed the first VCA rodent model that can assess behavioral functional recovery in conjunction with histologic evaluation of nerve regeneration and reinnervation. The investigators demonstrated that coaptation of all three forearm nerve (median, ulnar and radial) animals had superior grip strength and forearm function in the Fruit Loop test, compared to animals without nerve approximation. 67 More recently, Hanwright et al. developed a model of stimulated grip strength by combining aspects of isometric tetanic force testing (ITFT) and volitional grip strength testing (vGST). 68 While ITFT is very accurate, serial measurements are precluded, and vGST is associated with significant variability. Using a rat forelimb median nerve repair model, percutaneous stimulation median nerve stimulation resulted in maximal tetanic contraction of the digital flexors. In this manner, serial measurements of maximal grip strength can be measured, in the absence of behavioral variability. 68 Both of these forelimb models should contribute significantly to the study of VCA graft function in the rat model.
As Dubernard et al. 3 stated, the expansion of the field of VCA can be attributed to three areas of advancement: development of microsurgical instruments and techniques, more effective and better tolerated immunosuppression, and the development of animal models. Over the last two decades, the field of clinical VCA has expanded and new indications for VCA are being implemented. However, the number of clinical transplants performed each year are still low. Experimental models make it possible to address other mechanistic research issues that cannot be studied in humans for ethical or logistical reasons. In designing research projects to address clinical challenges, investigators should focus on the advantages and disadvantages with respect to translation, technical difficulty and finally logistical and financial feasibility.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Office of the Assistant Secretary of Defense for Health Affairs through the Reconstructive Transplantation Research Program (RTRP) under Award No. W81XWH-13-2-0057. Opinions, interpretations, conclusions, and recommendations are those of the authors and are not necessarily endorsed by the Department of Defense.
