Abstract
High-grade gliomas (HGGs) are the most aggressive of brain tumors and are one of the most common primary intracranial malignancies. The poor prognosis after aggressive treatment of HGGs makes these gliomas a challenge to treat with curative intent. Proton radiation therapy is a recent radiation modality that is being explored for the treatment of HGGs. Proton radiation therapy provides improved sparing of critical normal structures while giving an ablative dose of radiation to the tumor, which can be performed more accurately than photon beam radiation therapy. We report a case of a diffuse HGG treated with proton radiotherapy and chemotherapy after previously being treated with photon irradiation. A complete radiographic response was seen on MRI imaging after proton irradiation.
Introduction
Gliomas are one of the most common types of brain tumors originating from the glial cells. The glial cells are non-neuronal cells in the central nervous system (CNS) and the peripheral nervous system (PNS) that do not produce electrical impulses. The glial cells help to facilitate communication between neurons, assist in the regulation of inflammation in the CNS and PNS, and help form the blood-brain barrier. The World Health Organization (WHO) grading system assigns a grade from I to IV for gliomas, with a grade I 1 glioma being the least aggressive and a grade 4 glioma being the most aggressive. Grades I and II are defined as low-grade gliomas and grades III and IV are defined as high-grade gliomas (HGGs). HGGs are the most common primary intracranial malignancies, accounting for approximately 47.1% of malignant brain tumors. 1 The poor prognosis after aggressive treatment of HGGs makes these gliomas a challenge to treat with curative intent. Proton radiotherapy is a recent radiation modality that is being used to treat HGGs. It uses high-energy protons to damage and destroy cancer cells. Because of its unique properties, proton radiation therapy significantly limits the radiation dose to surrounding tissues.
We report a case of a 20-year-old male with a past medical history of melanotic neuroectodermal tumor of the right parietal occipital lobe status post gross total resection of the tumor, craniospinal irradiation, and chemotherapy, completed 15 years previously who now presents with a diffuse HGG. The patient was treated with proton radiotherapy and chemotherapy and achieved a complete radiographic response. Unfortunately, 6 months after treatment, the tumor recurred with leptomeningeal spread, and the patient succumbs to his disease shortly afterward.
Case presentation
A male in his early twenties with a past medical history of melanotic neuroectodermal tumor of the right parietal occipital lobe status post gross total resection of the tumor, craniospinal irradiation, and chemotherapy, completed 15 years previously, presented with a new right-sided facial palsy noticed by his mother. He denied any new history of headaches, emesis, gait or vision changes, back pain, extremity weakness, tingling/numbness of toes and fingers, changes in bladder or bowel habits, or seizures. A brain MRI was performed approximately 1 week after presentation, which revealed an interval increase in the size of the areas of contrast enhancement in the posterior fossa along the margins of the fourth ventricle. The lesion, which had been stable for almost 15 years, measured 9.5 × 8.7 × 6.0 mm, and on the recent brain MRI (Three Tesla Phillips MRI) using a T1-TSE sequence, the lesion had increased in size and measured 16.0 × 16.3 × 15.9 mm (Figure 1). An MRI of the cervical, thoracic, and lumbar spine revealed no evidence of cerebrospinal fluid (CSF) metastasis or seeding of the spinal cord.
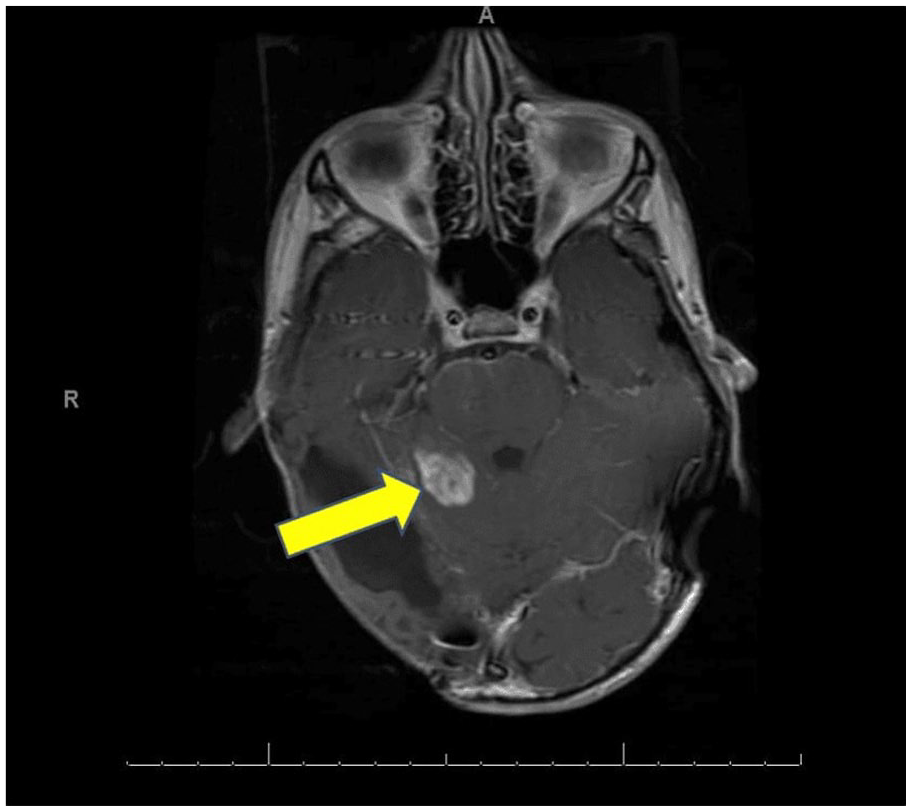
Axial T1 MRI showing the right cerebellar lesion.
He underwent a stereotactic biopsy of the right cerebellar lesion, and histopathology was consistent with a diffuse HGG WHO grade IV. The biopsy revealed relatively uniform small neoplastic glial cells infiltrating the cerebellar cortex and underlying white matter with a high Ki-67 labeling index 2 and early microvascular proliferation. A 1p36 deletion was identified, but no IDH1, IDH2, or BRAF mutations were detected.
The patients had undergone previous radiation therapy approximately 15 years ago and received 36 Gray (Gy) in 20 fractions to the craniospinal axis with a boost to the posterior fossa to an additional 19.8 Gy in 11 fractions, for a total dose of 55.8 Gy to the posterior fossa in 31 fractions. He underwent re-irradiation with proton radiation to the brain lesion to a total dose of 54 Gray equivalents (GyE) in 1.8 GyE per fraction in 30 fractions (Figures 2 and 3).

Sagital view of proton radiation plan. Gross tumor volume (GTV) is in red, clinical target volume (CTV) is in orange, Brainstem is light blue, 95% isodose line is in light green, 50% isodose line is light purple, 25% isodose line is dark green and 5% isodose line is in blue.

Axial view of proton radiation therapy plan. The brainstem is light blue, the CTV is in orange and is almost completely covered by the 100% (yellow) and completely covered by the 95% (green) isodose line.
A dose-volume histogram is shown (Figure 4) for the proton therapy. The maximum dose (Dmax) to the brainstem was 55.9 GyE, and the mean dose was 36.8 GyE. The Dmax to the brainstem from the previous photon radiation therapy 15 years prior was 50 Gy. The total combined dose to the brainstem from the photon and the proton radiotherapy was approximately 105.9 GyE. The Dmax to the structures at risk, including the right optic nerve, was 6.8 GyE, the left optic nerve was 2.2 GyE, the optic chiasm was 18.5 GyE, the pituitary gland was 25.8 GyE, and the Dmax to the left cochlea was 9.1 GyE and the right cochlea was 41.6 GyE. The patient received concurrent chemotherapy with Temozolomide (TMZ) 140 mg daily and was prescribed a pentamidine infusion (antiprotozol agent) for pneumocystis jiroveci pneumonia (PJP) prophylaxis. (PJP can be found in the lungs of immunosuppressed patients). The patient tolerated treatment well, with no significant side effects from either the proton radiotherapy or the chemotherapy. After completion of the proton radiotherapy, the patient was initiated on maintenance chemotherapy with TMZ 160 mg and Lomustine 90 mg for six cycles, with each cycle lasting 6 weeks. After starting the first cycle of chemotherapy, the patient developed diarrhea, which lasted approximately 2 weeks. No abdominal pain or fever was associated with diarrhea, and his stool was negative for

Dose-volume histogram for proton therapy treatment. Red is the GTV, dark orange is the CTV, light blue is the brainstem, and yellow is the optic chiasm. The green and light orange lines in the low-dose area represent the optic nerves.

Axial T1 MRI showing a complete radiographic response to treatment.
Approximately 4 months after treatment, he developed new-onset numbness of the left side of his body as well as decreased hearing in his left ear after the completion of his maintenance chemotherapy. A brain MRI was performed, which revealed the progression of the disease with evidence of leptomeningeal disease and interval development of enhancement within the internal auditory canal concerning metastasis. He was initiated on avapritinib chemotherapy but unfortunately passed away less than 1 month after the MRI scan and 5 months after undergoing proton radiotherapy for his recurrent disease.
Discussion
Gliomas account for approximately 27% of all primary brain tumors and most often present themselves in adults, with males being more susceptible than females. 3 The WHO grading system divides gliomas into four grades, with grade 1 gliomas being the least aggressive and grade 4 gliomas being the most aggressive. 4 The treatment and prognosis of gliomas mainly depend on the grade of the glioma, which can be dramatically different. Low-grade gliomas are typically treated with surgical resection of the tumor, followed by observation or adjuvant therapy. The standard treatment approach for HGGs is to obtain maximal surgical resection, which is followed by radiation therapy with concurrent and adjuvant TMZ. 5
Surgical resection of HGGs can sometimes be challenging since they are highly infiltrative tumors that can invade the surrounding brain tissue beyond the tumor margins that are not visible on neuroimaging. Therefore, microscopic disease can be present beyond the surgical margins even when a gross total resection is achieved. Multifocal or diffusely infiltrative tumors, tumors deep in the brain, and tumors adjacent to or within eloquent areas of the brain may limit the extent of resection or even prevent any attempt of surgery beyond a biopsy. 6 TMZ chemotherapy and radiation therapy are commonly used after surgery as adjuvant treatment or as the primary treatment when surgery is not possible.
Radiation therapy is essential in the treatment of gliomas but can also damage normal brain tissue. This, in turn, can result in neurocognitive impairment, neurologic deficits, neurovascular compromise, neuroendocrine deficiency, and second malignancies. 5
Compared to photon therapy, a unique property of proton radiotherapy is its ability to significantly decrease the radiation dose to the surrounding tissues outside the target. The entrance and exit dose is minimal compared to conventional photon irritation, allowing most of the energy from the proton therapy to be deposited into the tumor. The rate of energy loss of the protons can be plotted on a curve called a Bragg curve. The highest point on the Bragg curve is called the Bragg peak, which occurs when the protons deposit most of their energy in the tumor volume. The proton beam’s initial energy determines the Bragg peak’s depth. The position and depth of the Bragg peak can be adjusted and modified and placed anywhere in the patient. Several Brag peaks can be shifted in-depth and weighted to create a well-defined dose distribution. This is an essential property of proton radiation therapy and aids in decreasing the dose to the critical structures in the brain, such as the hypothalamus, pituitary, cochlea, and optic pathways, while still treating the tumor. 5
To successfully treat a patient with proton therapy, the proton beam must be broadened longitudinally and laterally to conform to the target shape. Several methods can be used to modify the proton field, but the two most common methods are passive scattering of the protons and magnetic scanning of narrow “beamlets” of protons of different energies. 7
With passively scattered proton therapy, the initially monoenergetic narrow beam of the appropriate beam energy is spread longitudinally, laterally, and then shaped appropriately to conform to the target volume. With magnetic scanning, thin “beamlets” of protons of a variety of energies are used to achieve a proton dose distribution that conforms to the shape of the target volume while optimally sparing normal tissues. 7
Another factor that must be considered in treating patients with proton therapy is the radiosensitivity of the different brain structures. The alpha-beta ratio measures the radiosensitivity and radio-resistance of normal tissues and tumors. This ratio expresses how resistant a cell line is to radiation damage. The higher the ratio, the more radioresistant the cell line is thought to be.
The alpha-beta ratio for glioblastoma multiforme (GBM) cells is estimated to be approximately 8 Gy compared to normal tissues within the CNS, which are estimated to have an alpha-beta ratio of approximately 2 Gy, suggesting that GBMs are more radioresistant than normal brain tissue. 8
The inherent radiosensitivity of normal brain tissue manifests as late toxicity in the form of radionecrosis and is evident in some patients after completing CNS-directed radiotherapy. 9 One key mechanism contributing to radionecrosis is radiation-triggered endothelial cell and oligodendrocyte death through apoptosis. Post-irradiation vascular insufficiency leads to chronic ischemia, which in turn leads to worsening hypoxia, increased oxidative stress, and reactive oxygen species generation, toxic to normal brain tissue near the tumor being treated. By decreasing the dose to normal brain tissue with the use of proton therapy, late toxicity of normal brain tissue can also be reduced. 9
Current radiotherapy indications for proton therapy are limited but are increasing, and currently at about 16%.
5
In a clinical study by Kong et al., 50 patients with either GBM (
An additional case report examined a 42-year-old male who presented with a one-week history of headache and vomiting. A brain MRI displayed an obstructive hydrocephalus and a ring-enhancing lesion located in the right posterior thalamus. 11 A biopsy of the brain lesion was obtained via an endoscopic third ventriculostomy and stereotactic biopsy. Pathologic review of the biopsy specimen revealed endothelial proliferation and areas of necrosis, which were consistent with a diagnosis of glioblastoma. Surgical resection was not performed, and the patient was treated with conventional photon radiation therapy and received a dose of 50.0 Gy in 2.0 Gy fractions to the tumor and surrounding edema. This was followed by a proton radiotherapy boost to the tumor with 10 GyE in 2.0 GyE fractions. 11 In this case report, a complete radiological response was observed on MRI 1 year after the end of radiation therapy.
Tumor-treating fields (TTFields) are alternating electrical fields that are pulsed through the skin of the scalp, which can interfere with cellular processes critical for cancer cell viability and tumor progression. 12 TTFields and TMZ chemotherapy were initiated concurrently 4 weeks after completion of chemoradiation. TMZ therapy was completed after six cycles, and a brain MRI obtained approximately 4 weeks after the completion of photon and proton radiation therapy revealed an increase in T1 and T2 fluid-attenuated inversion recovery (FLAIR) in the tumor. The patient was asymptomatic and overall stable, suggesting that this was most consistent with a pseudoprogression of the tumor. TTFields were continued, and the tumor decreased in size on serial MRI scans. Approximately 1 year after photon and proton radiation therapy was completed, a complete radiographic response was noted, with no evidence of the tumor seen on imaging. The patient was healthy on clinical examination and had no new neurological deficits.
In another case, a 45-year-old male presented to the Emergency Room with seizures, and a brain MRI revealed a diffuse lesion involving the right medial temporal, right occipital, and posterior parietal lobes of the brain. He underwent a right parieto-occipital navigational guided burr hole biopsy of the occipital lesion, and histopathology was consistent with an infiltrative glioma grade III. 13 The molecular markers were consistent with an IDH mutation, IP19q codeletion, and a telomerase reverse transcriptase mutation. Due to his age and some adverse features of the high-grade component of his glioma, he underwent treatment with proton radiotherapy to a dose of 54 GyE in 30 fractions at 1.8 GyE per fraction, along with concurrent TMZ chemotherapy. 13 He tolerated the treatment, and a response assessment scan after 4 weeks demonstrated an approximate 50% reduction in the brain lesion.
In a third case, a male in his thirties underwent an evaluation for new generalized tonic-clonic seizures. A brain MRI revealed a hyperintense area in the anterior occipital and posterior temporal lobe, which was suspicious of a malignancy. He underwent a left temporoparietal craniotomy and resection of the brain lesion. A histopathology review of the surgical specimen revealed an oligodendroglioma WHO grade II (Ip19q deleted). He received regular follow-ups, and a brain MRI was obtained every 6 months. Imaging over time showed an increase in the size of the lesion involving the left temporal lobe. At that time, he also began to experience occasional episodes of tonic-clonic seizures. A brain MRI demonstrated an infiltrative lesion measuring 5 × 3.8 × 5.56 cm in the left posterior temporal lobe of the brain with ill-defined patchy enhancement, which was concerning for disease progression. Due to his age and favorable histology, he was treated with proton radiotherapy at a dose of 55.8 GyE in 31 fractions of 180 GyE per fraction, along with concurrent TMZ chemotherapy. 13 He tolerated the treatment, and an MRI scan approximately 4 weeks after treatment displayed a nearly 50% reduction in the brain lesion.
A study by Jhaveri et al. evaluated photon versus proton radiation therapy for patients with WHO grade I–IV glioma treated with definitive radiation therapy between 2004 and 2013.
14
The patients were identified from the National Cancer Data Base (NCDB) and were stratified into two main groups: proton beam therapy (
Concerning Toxicity and proton beam radiation therapy, a study by Vora et al. 15 retrospectively reviewed 63 patients who underwent definitive proton beam therapy for grade 3 or 4 gliomas at six institutions between 2009 and 2017. A total of 89% of the patients received concurrent TMZ with radiation. The median dose delivered was 59.4 GyE (range 40–66 GyE), given over 15–33 fractions. Proton therapy was well tolerated, with Common Terminology Criteria for Adverse Events (CTCAE version four) grade 1/2 for symptoms such as alopecia, headache, ataxia, confusion, insomnia, and dermatitis. Three patients (4.6%) developed grade 3 acute toxicity. In conclusion, the authors stated that proton beam radiation therapy was well tolerated, and outcomes were comparable to photon-based historical controls. 15
Concerning recurrence patterns for HGGs, a study by Zhou 16 retrospectively evaluated recurrence patterns in 54 patients with recurrent HGGs. The clinical data and serial MRIs of these patients were assessed, and all the patients received adjuvant radiation and TMZ chemotherapy after total resection, subtotal resection, or a biopsy. 16 Local recurrence (central and in-field) was found to be the main recurrence pattern. The rate of marginal recurrence was low, being less than 5%. 16 At a median follow-up of 14 months, 54 HGG patients developed tumor recurrence. The median overall and progression-free survival were 14 and 10.5 months, respectively. A total of 34 patients developed central recurrence; 8 presented with in-field recurrence, 2 developed marginal recurrence, 2 developed distant recurrence, and 11 developed cerebrospinal fluid dissemination, 2 of whom developed central recurrence, with 1 patient simultaneously developing marginal recurrence. 16
Finally, in addition to surgery, adjuvant radiation therapy, and TMZ chemotherapy, other treatment options, including molecularly targeted therapies, have been developed as a potential therapy for treating HGGs. In a paper by Begagic et al., a systemic review of the current molecular targeted therapies for treating HGGs was examined. 17 A total of 166 studies with 2526 patients were analyzed. The most common critical molecular targets currently being investigated and explored in clinical and laboratory studies to treat HGGs were identified. These included Epidermal Growth Factor Receptor (EGFR), Mammalian Target of Rapamycin (mTOR), Vascular Endothelial Growth Factor (VEGF), and Mitogen-Activated Protein Kinase (MEK). 17 All of these therapies are promising, according to the paper. Most studies discussed the need for a comprehensive therapeutic approach to treating HGGs. They also discussed many promising therapies for the various molecular targeted therapies as mentioned. Targeted therapies now offer an advantage in efficacy and reduced collateral damage. Potential side effects of molecular targeted therapies are being investigated. Finally, the authors state that molecular targeted therapies’ potential efficacy and tolerance still need to be examined. 17
Conclusion
The standard of care for the treatment of HGGs is surgical resection followed by radiation therapy with concurrent and adjuvant TMZ. Technological advances in radiation therapy, especially in proton radiotherapy, have shown promise in treating HGGs, especially in the recurrent setting after photon radiation therapy. One of the several advantages of treating patients with proton radiotherapy is its ability to give a focused dose of radiation to the tumor and avoid nearby critical tissues, especially in the brain. Besides surgery, radiation, and chemotherapy, other potential treatment modalities, including molecular therapy, are being explored for HGGs. Finally, photon radiation therapy and proton radiotherapy should always be considered in the treatment regimen for HGGs. Proton radiotherapy should always be considered when retreating HGGs. Other treatment modalities and the potential retreatment of HGGs are also being explored.
Footnotes
Acknowledgements
Not Applicable.
Author contributions
J.H.: substantial contributions to concept or design, acquisition, analysis, or interpretation of data, drafting of the manuscript, has agreed to be accountable for all aspects of the work and will review the final version to be published. C.H.: acquisition, analysis, or interpretation of data, critical review of the manuscript for important intellectual content, has agreed to be accountable for all aspects of the work and will review the final version to be published. M.J.: acquisition, analysis, or interpretation of data, critical review of the manuscript for important intellectual content, has agreed to be accountable for all aspects of the work and will review the final version to be published. S.M.: substantial contributions to concept or design, acquisition, analysis, or interpretation of data, drafting of the manuscript, has agreed to be accountable for all aspects of the work and will review the final version to be published and supervise the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series
Informed consent
Written informed consent from the legally authorized representative of the DECEASED subject for the publication of this case report was obtained.
