Abstract
Postpartum cardiomyopathy is a type of heart failure that occurs during late pregnancy or early postpartum without clear causes. It poses significant health risks. Recognition and management are crucial for better outcomes. We describe a case of a 23-year-old Ugandan woman who developed heart failure symptoms 1 month after giving birth. Physical examination revealed signs of congestive heart failure. Chest X-ray showed cardiomegaly, leading to a diagnosis of postpartum cardiomyopathy based on clinical criteria. Treatment involved diuretics, beta-blockers, angiotensin receptor blockers, SGLT2 inhibitors, and bromocriptine. This case underscores the importance of considering postpartum cardiomyopathy in the postpartum period. Prompt diagnosis and comprehensive management through a multidisciplinary approach can improve patient outcomes. Further research is needed to deepen our understanding of this condition.
Background
Postpartum cardiomyopathy is defined by the Heart Failure Association of the European Society of Cardiology Working Group on peripartum cardiomyopathy (PPCM)1,2 as an idiopathic cardiomyopathy characterized by heart failure secondary to left ventricular (LV) dysfunction occurring toward the end of pregnancy or in the months following delivery, in the absence of any other identifiable cause of heart failure. Postpartum cardiomyopathy represents a rare form of heart failure manifesting toward the latter stages of pregnancy or within the months following childbirth, devoid of other identifiable causes of cardiac dysfunction. 3 Clinically, postpartum cardiomyopathy is characterized by diminished LV systolic function, often delineated by an LV ejection fraction (LVEF) below 45%, accompanied by associated phenotypes such as LV dilatation, Bia trial dilatation, compromised systolic and diastolic functions, and elevated pulmonary pressures. 1 The etiology of postpartum cardiomyopathy remains incompletely elucidated, hypothesized to stem from a complex interplay of factors encompassing the hemodynamic stresses inherent to pregnancy, vasculo-hormonal dynamics, inflammatory processes, immunological responses, and genetic predispositions. 4 Diagnosis of postpartum cardiomyopathy encompasses a spectrum of modalities including electrocardiography (ECG), echocardiography, magnetic resonance imaging, endomyocardial biopsy, and specific blood biomarkers. 5 Therapeutic strategies for postpartum cardiomyopathy hinge upon the pregnancy stage, postpartum duration, disease severity, and lactation status, encompassing conventional pharmacological interventions tailored to heart failure management, with emerging agents such as bromocriptine demonstrating potential efficacy in initial investigations. 6 In instances of profound postpartum cardiomyopathy, interventions may escalate to encompass mechanical circulatory support and heart transplantation. Encouragingly, a significant proportion of affected women achieve normalization of LV function within the first year following diagnosis.
The global prevalence of postpartum cardiomyopathy exhibits considerable variation, with notably higher incidence rates documented in regions such as Nigeria compared to lower rates reported in countries such as Japan. 7 Within sub-Saharan Africa, dilated cardiomyopathy, including postpartum cardiomyopathy, emerges as a significant contributor to heart failure burden, ranking second only to hypertensive heart disease. 8 In Uganda specifically, postpartum cardiomyopathy prevalence is relatively elevated, with a notable proportion of women presenting with acute heart failure during the peripartum period diagnosed with postpartum cardiomyopathy. 9 Notably, multiple pregnancies serve as a predictive factor for postpartum cardiomyopathy occurrence in this population. 10 However, the prognosis for postpartum cardiomyopathy in Uganda remains unfavorable, as evidenced by a substantial percentage of patients experiencing incomplete recovery of LV function at the 6-month mark. 11 Given the substantial impact of postpartum cardiomyopathy on maternal and fetal health outcomes, further comprehensive investigations are warranted to elucidate the long-term implications of this condition in Uganda and inform targeted interventions aimed at improving patient outcomes. We present a case that we believe is rare in Uganda and holds significant clinical implications.
Case presentation
A 23-year-old female Ugandan, with no known chronic illnesses, admitted at Kayunga Regional Referral Hospital with complaints of generalized body swelling persisting for 3 weeks, accompanied by shortness of breath upon exertion, orthopnea, paroxysmal nocturnal dyspnea (PND), and easy fatigability. Subsequently, the patient developed a history of cough, particularly pronounced at night, with a small amount of blood-tinged sputum that was odorless and not associated with night sweats, weight loss, or fever. Additionally, the patient reported experiencing abdominal pain, distension, and loss of appetite. Review of other systems revealed no remarkable findings. Medical history indicated that this was the patient’s second admission, with no known chronic illnesses, and she tested negative for HIV. She had not undergone any previous surgeries or blood transfusions. There was no family history of chronic illness among her relatives. In terms of social history, the patient is a mother of two children, with her most recent childbirth being 6 weeks prior, delivered normally. She is employed at a restaurant, does not consume alcohol, and doesn’t smoke.
During the examination, by then, a young female sitting on the bed without apparent distress. On tactile assessment, the patient was found to be afebrile, with moderate conjunctival pallor and tinge scleral jaundice noted, and no lymphadenopathy, however generalized edema was observed. Vital signs recorded, blood pressure of 90/58 mmHg, a pulse rate of 112 beats per minute (bpm), oxygen saturation of 90%, a temperature of 36.7°C, and a respiratory rate of 24 breaths per minute. Cardiovascular examination revealed a pulse rate of 112 bpm, characterized as regular and weak, with synchronous pulsations. The apex beat was displaced and palpated at the 6th intercostal space, along the anterior axillary line, without evidence of thrills or heaves. Jugular venous pressure was noted to be distended, and auscultation revealed a pansystolic murmur of grade two radiating to axilla. And bilateral basilar crackles noted. Abdominal examination revealed distension with generalized tenderness on palpation and no organomegaly appreciated. Central nervous system examination findings were unremarkable.
Diagnostic investigations, including a complete blood count, renal function tests, liver function tests, electrolyte levels, lipid profile, abdominal ultrasound, and chest X-ray, were conducted. However, due to limited resources, ECG and echocardiography were not available. Chest X-ray (Figure 1) revealed enlarged heart (cardiomegaly). Other laboratory investigations returned within normal ranges except cardiac blood test that revealed an elevated level of N-terminal prohormone of brain natriuretic peptide (NT-proBNP) at 8670 pg/ml. and prolactin level of 49 ng/mL). Based on clinical findings and diagnostic evaluations, the patient was diagnosed clinically with congestive heart failure secondary to postpartum cardiomyopathy, as per Framingham criteria. 12 Treatment was initiated with intravenous furosemide 40 mg twice daily, oral bisoprolol 5 mg once daily, oral dapagliflozin 10 mg once daily, and oral bromocriptine 5 mg once daily, with close monitoring of vital signs (Figure 3(a)). Subsequent improvement was noted after 1 week of treatment, and the patient was referred for an echocardiogram. Next appointment was scheduled for 2 weeks.
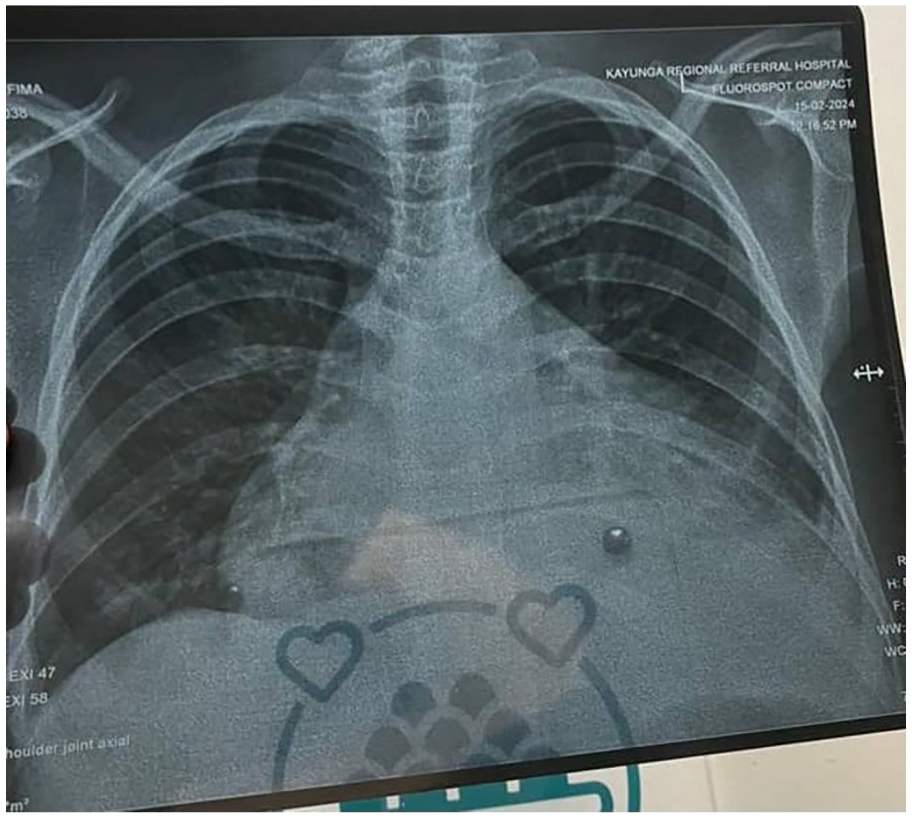
Chest X-ray that showed cardiomegaly.
At the 2-week follow-up, the patient presented with echocardiography (Figure 2) conducted at Mulago National Specialist Hospital, revealing an ejection fraction of 30% and LV dilatation. Clinical symptoms had improved, and concurrently, laboratory tests indicated a decrease in biomarkers (NT-proBNP and prolactin levels) (Figure 3(b)), suggesting a favorable trajectory in cardiac function. In light of these encouraging developments, medications were continued to maintain the therapeutic benefit.

Echocardiography showing reduced ejection fraction of 30% and left ventricular dilatation.

(a) Combining weight, respiratory rate, oxygen saturation, and medication duration. (b) Changes in brain natriuretic peptide and prolactin levels over time. (c) Clinical course, including treatment and symptom onset.
By the 2-month visit, the patient had become asymptomatic (Figure 3(c)), with NT-proBNP and prolactin levels decreasing further, marking a significant milestone in their recovery journey. Capitalizing on this clinical improvement, a cautious approach was adopted, and medications were gradually tapered over subsequent months to align with the patient’s evolving needs and ongoing progress. This sustained improvement underscores the efficacy of the treatment regimen and bodes well for the long-term prognosis of the patient.
Discussion
Postpartum cardiomyopathy presents as a rare form of heart failure emerging toward the culmination of pregnancy or in the postpartum period. Its risk profile encompasses diverse factors including multifetal gestation, high gravidity, extremes of reproductive age, prolonged tocolysis, race, ethnicity, multiparity, advanced maternal age, pre-eclampsia, lack of formal education, unemployment, underweight status, and selenium deficiency.1,3 Moreover, vasculo-hormonal dynamics, inflammation, immunological perturbations, and genetic predispositions constitute additional contributory elements to postpartum cardiomyopathy pathogenesis. 5 It’s noteworthy that customary postpartum practices previously linked to postpartum cardiac failure, like regular hot baths and pap enriched with dried lake salt, lack association with postpartum cardiomyopathy occurrence. 1 Vigilant monitoring for potential cardiac complications is warranted in patients with gestational hypertension and other identified risk factors.
Postpartum cardiomyopathy emerges as a life-threatening cardiomyopathy characterized by LV systolic dysfunction occurring late in pregnancy, during delivery, or within the initial postpartum months, notably in women without preexisting heart disease. The intricate pathophysiology of postpartum cardiomyopathy involves a constellation of factors, encompassing genetic predisposition, activation of inflammatory pathways within the immune system, and the impact of prolactin metabolites, which induce vasoconstriction, apoptosis, inflammation, and capillary destruction. Additionally, oxidative stress and antiangiogenic activity contribute to the disease process. Treatment modalities for postpartum cardiomyopathy entail medical management employing bromocriptine therapy, anticoagulant drugs, and nonpharmacological interventions. Prospective therapies targeting dysregulated oxidative stress and angiogenesis hold promise for future treatment avenues. Optimal management of women with postpartum cardiomyopathy necessitates the involvement of a multidisciplinary team proficient in both high-risk pregnancy and heart failure management. While ventricular function recovery in postpartum cardiomyopathy surpasses that of other forms of dilated cardiomyopathy, select cases may necessitate mechanical circulatory support and/or cardiac transplantation for definitive management. 13
Postpartum cardiomyopathy manifests as a rare manifestation of heart failure occurring during late pregnancy or in the early postpartum period. Clinical presentation includes atypical chest pain, abdominal discomfort, signs of pulmonary edema, orthopnea, PND, and fatigue. Given the resemblance of these symptoms to those of normal pregnancy, diagnosis delays can transpire, underscoring the importance of prompt recognition and medical intervention tailored for pregnancy and lactation to avert adverse outcomes. Management of Postpartum cardiomyopathy mirrors that of other forms of congestive heart disease, encompassing medications such as diuretics, aldosterone antagonists, and angiotensin II receptor blockers. Ongoing cardiologist follow-up is paramount for all postpartum cardiomyopathy patients, while preconception counseling and collaborative care between obstetrics and cardiology are imperative for women contemplating subsequent pregnancies.14–17
Delays in diagnosing postpartum cardiomyopathy may arise due to the resemblance of heart failure symptoms to those commonly experienced during pregnancy. Therefore, clinicians should maintain a high index of suspicion for postpartum cardiomyopathy in pregnant or postpartum women presenting with symptoms suggestive of heart failure. 15 Postpartum cardiomyopathy diagnosis involves a comprehensive approach integrating clinical assessment and diagnostic modalities. Manifestations of heart failure in pregnant or postpartum individuals should prompt consideration of postpartum cardiomyopathy, prompting the need for diagnostic evaluation. Laboratory investigations, including N-terminal pro-BNP, serve as valuable adjuncts in confirming suspicions, 1 while echocardiography remains pivotal in postpartum cardiomyopathy diagnosis, enabling the assessment of LVEF and other relevant cardiac parameters. 15 Complementary to echocardiography, cardiovascular magnetic resonance imaging offers a detailed evaluation of myocardial structure, function, and tissue characteristics, encompassing features such as late gadolinium enhancement and fibrotic burden. 18 Speckle tracking echocardiography emerges as an additional diagnostic modality, facilitating the evaluation of LV function through the measurement of global longitudinal and circumferential strains. 19 Moreover, cardiac biomarkers such as NT-proBNP and cardiac troponin I (cTn-I) contribute to postpartum cardiomyopathy diagnosis by providing additional insights into cardiac function and injury. 20
The treatment approach for postpartum cardiomyopathy encompasses a range of conventional pharmacological heart failure therapies tailored to address the underlying pathophysiology. Commonly employed medications include diuretics, angiotensin-converting enzyme inhibitors, vasodilators, digoxin, β-blockers, and anticoagulants, which collectively aim to alleviate symptoms and improve cardiac function. 21 Notably, bromocriptine, a dopamine-receptor antagonist, has emerged as a promising therapy for postpartum cardiomyopathy by suppressing prolactin release, thereby offering potential benefits in select cases. 22 Similarly, therapies targeting angiogenesis, such as vascular endothelial growth factor agonists, hold promise in augmenting cardiac function through the promotion of vascular growth and repair. 4 In cases where immune dysregulation contributes to disease pathogenesis, high-dose intravenous steroids and immunosuppressants may be considered adjunctive therapies to attenuate inflammation and mitigate cardiac injury. 1 Dopamine agonists such as cabergoline have also demonstrated efficacy in select patients, further expanding the armamentarium of postpartum cardiomyopathy treatment options. 23 However, in severe or refractory cases, mechanical circulatory support and cardiac transplantation may be warranted to provide temporary or definitive management solutions. Given the complex nature of postpartum cardiomyopathy management, a multidisciplinary approach involving specialists in high-risk pregnancy and heart failure therapy is essential to optimize patient outcomes and ensure comprehensive care.
While postpartum cardiomyopathy typically carries a favorable prognosis, it is essential to remain vigilant due to the potential for serious complications such as cardiac dysrhythmias, progressive heart failure, and thromboembolic events. 24 Timely recognition and management of postpartum cardiomyopathy are paramount to mitigate these risks. 25 The prognosis of postpartum cardiomyopathy is variable, ranging from complete resolution to progression to heart failure and even death. Long-term outcomes depend on factors such as age, ejection fraction, and the presence of comorbidities. While some cases may improve or resolve completely, others may experience residual LV systolic dysfunction or adverse outcomes.2,26,27
Conclusion
This case highlights the importance of maintaining a high index of suspicion for postpartum cardiomyopathy in the postpartum period. Timely diagnosis and management guided by multidisciplinary care can help improve outcomes in these patients. The diagnosis of postpartum cardiomyopathy can be challenging due to its overlapping clinical presentation with other cardiac conditions and the lack of specific diagnostic criteria. The development of reliable biomarkers or imaging modalities specific to PPCM could improve early detection and facilitate timely intervention.
Footnotes
Acknowledgements
Not applicable.
Author contributions
M.J. Conceptualization and writing—Original draft preparation; A.M. Supervision and proofreading; F.D. Reviewing and editing.
Availability of supporting data
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from the Ethics Committee at Kayunga Regional Referral Hospital (Approval Number: KRRH-EC-2024-001).
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
