Abstract
Single atrium is a rare congenital heart disease characterized by complete absence of the interatrial septum. It occurs as an isolated cardiac defect, or as a part of skeletal, muscular, ophthalmologic, and vascular malformations to signify congenital syndromes. A 78-year-old male patient presented with generalized body swelling of 2 weeks duration. He had associated shortness of breath, orthopnea, productive cough, and palpitation. Upon physical examination, blood pressure = 150/75 mmHg, pulse rate = 50 bpm, respiratory rate = 24 bpm, and T° = 36.7 °C. He had signs of pleural effusion on left lung field. Cardiovascular examination revealed mean heart rate of 50 beats/min with irregularly irregular pulse rhythm, raised jugular venous pressure, and pansystolic murmur at left lower sternal border. He had tender hepatomegaly, ascites, and pitting leg edema. Chest X-ray showed cardiomegaly, prominent pulmonary trunks, and left-sided pleural effusion. Electrocardiography revealed atrial fibrillation, bifascicular block (right bundle branch block and left anterior fascicular block) with mean heart rate of 50 beats/min. Two-dimensional transthoracic echocardiography showed complete absence of interatrial septum (single atrium) without atrioventricular defect and interventricular communication. Diagnosis of heart failure secondary to congenital heart disease (single atrium) with atrial fibrillation and bifascicular block was made based on clinical and imaging evaluation. Well-tolerated symptoms of a single atrium until late adulthood could be explained by the presence of streaming or incomplete mixing of blood within the atrium, in which the more oxygenated blood is directed to the systemic circuit.
Introduction
Single atrium (SA) is defined as complete or near complete absence of the interatrial septum, and has clinical and hemodynamic similarities with large-size atrial septal defect of fossa ovalis type.1–3 It is a rare form of congenital heart defect accounting for 0.5%–1% of all congenital heart diseases. 4 SA is a term often used interchangeably with common atrium in medical literatures. However, presence of atrioventricular valve malformations with or without interventricular communication is accompanying abnormalities in common atrium.1,3,5 Most patients with SA tend to experience exertional dyspnea, palpitation, fatigue, and frequent respiratory infections during childhood.3–5 However, we described a patient with SA who experienced his first obvious symptoms at 78 years of age.
Case presentation
A 78-year-old male patient presented to Emergency Medical Department of University of Gondar Hospital, Northwest Ethiopia, with generalized body swelling of 2 weeks duration associated with shortness of breath, orthopnea, productive cough, and palpitation. He had waxing and waning bilateral leg swelling for the last 3 years. He had recurrent episodes of respiratory tract infections in the past 6 years. He had no previous hospital admissions for similar complaints. Upon physical examination, blood pressure (BP) = 150/75, pulse rate (PR) = 50 beats/min, respiratory rate (RR) = 24 breaths/min, and temperature (T°) = 36.7 °C. His arterial oxygen saturation (SaO2) was 92% while breathing ambient air. On respiratory system examination, he had grade two clubbing but no evidence of cyanosis. He had decreased air entry with relative dullness on left basal lung field. Cardiovascular examination revealed a mean heart rate of 50 bpm with irregularly irregular pulse rhythm, raised JVP, pansystolic murmur at left lower sternal border, but no summation gallop. On abdominal examination, he had ascites and tender hepatomegaly. He had bilateral pretibial and pedal pitting edema. No skeletal abnormalities were detected on locomotor system examination. Complete blood counts, liver biochemical tests, renal function tests, and serum electrolytes were within normal limits. Chest X-ray showed cardiomegaly, prominent pulmonary trunks, and left-sided pleural effusion (Figure 1). Electrocardiography revealed atrial fibrillation, bifascicular block (right bundle branch block and left anterior fascicular block) with mean heart rate of 50 beats/min (Figure 2). 2-D transthoracic echocardiography showed an absence of interatrial septum with SA diameter measuring 106 mm. No mitral valve defect or cleft was seen. The atrioventricular valve attachments to the interventricular septum were on the same anatomic plane. No interventricular communication. Left ventricular ejection fraction was 55%. There was also severe tricuspid regurgitation [tricuspid regurgitation pressure gradient (TRPG) = 68 mmHg] and dilated main pulmonary artery (Figure 3). Diagnosis of congestive heart failure secondary to congenital heart disease (SA) with atrial fibrillation and bifascicular block was made. The patient was treated with furosemide (40 mg po twice daily), spironolactone (25 mg po daily), and anticoagulated with warfarin (5 mg po daily). His cardiac symptoms improved and edema subsided, and INR value was within the therapeutic range. He was referred to a cardiac center in Addis Ababa, capital of Ethiopia, for surgical evaluation. Patient’s follow-up after referral to cardiac center was not available.
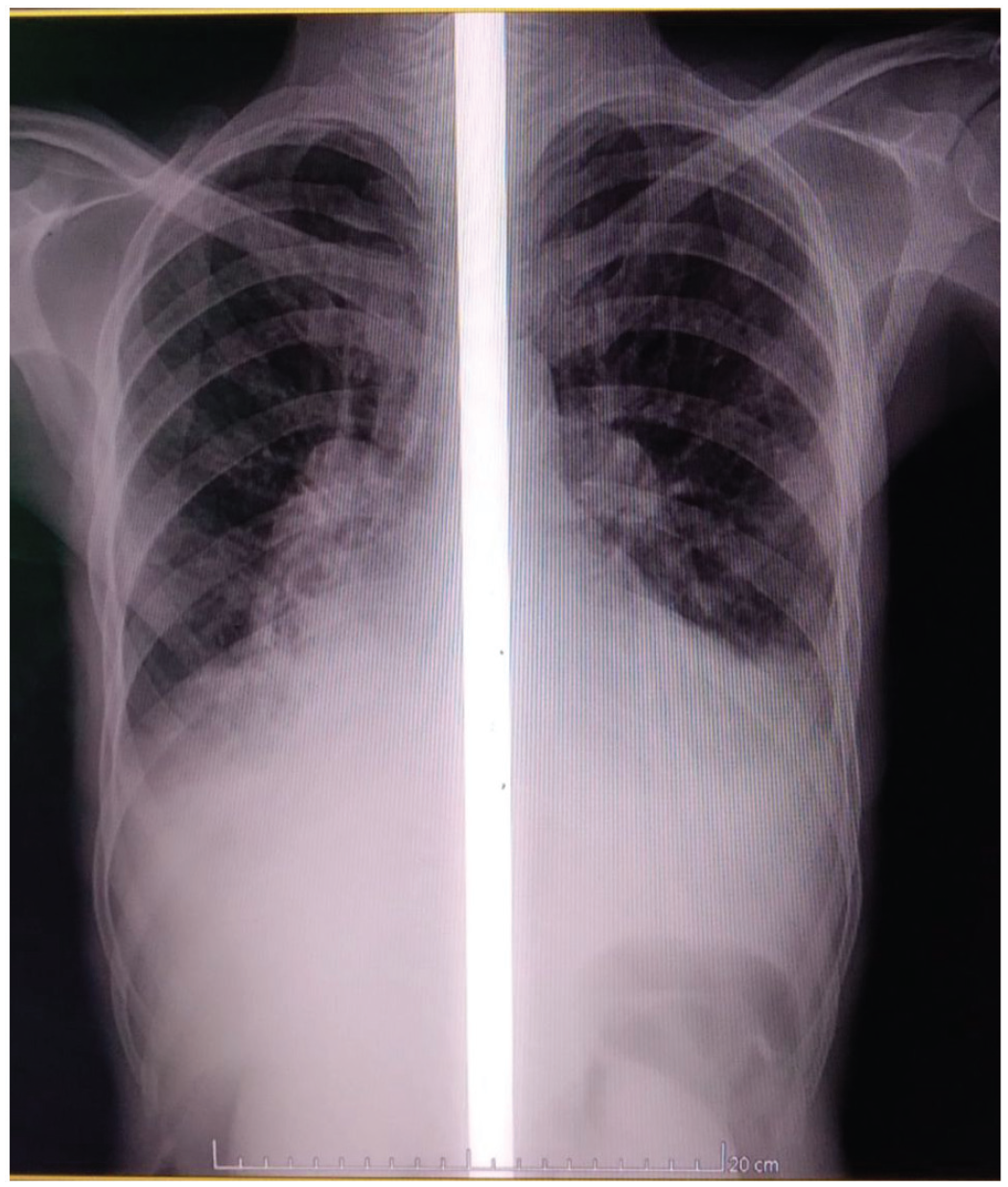
Chest X-ray showing cardiomegaly, prominent pulmonary trunks, and left-sided pleural effusion.
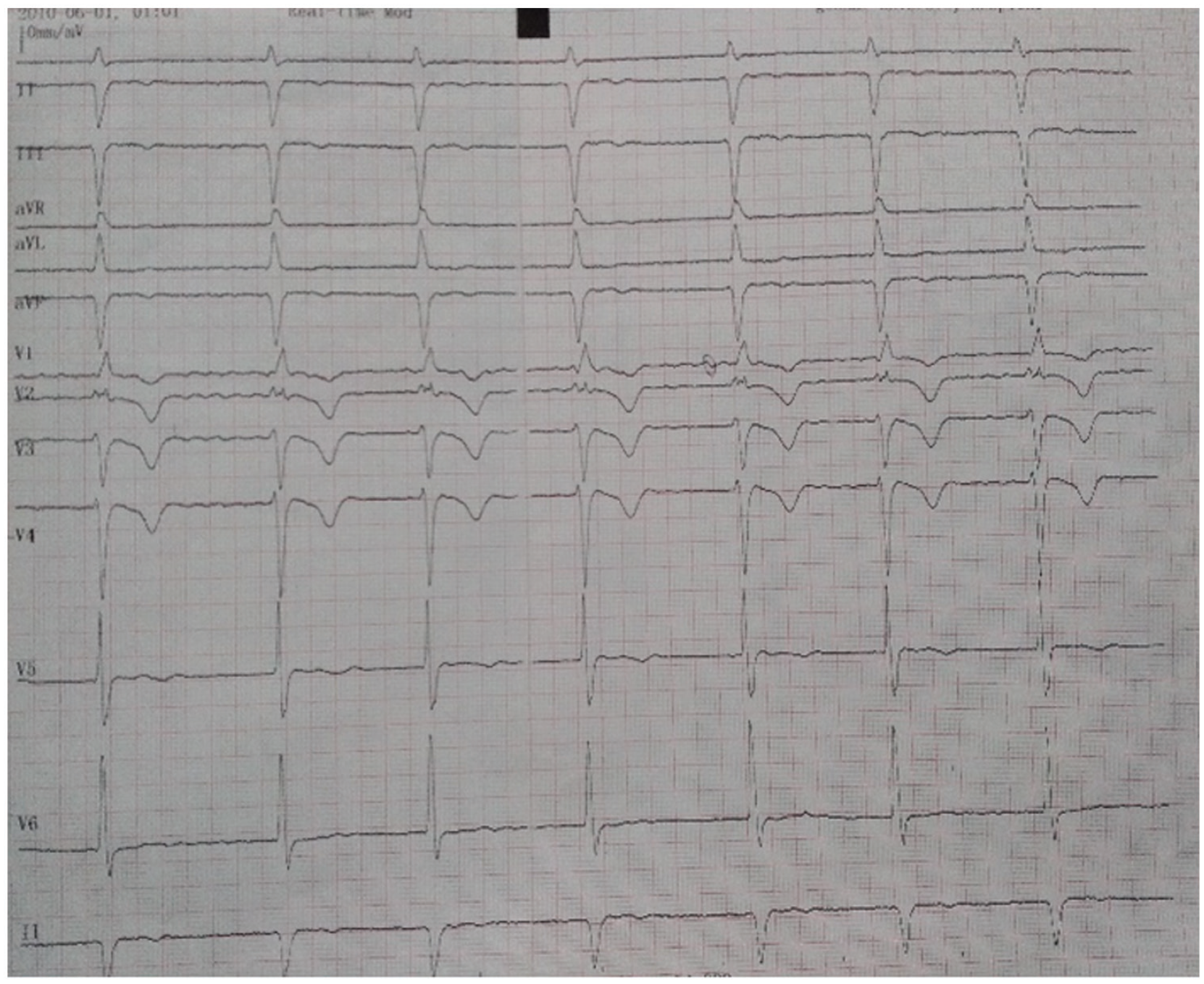
Electrocardiography showing atrial fibrillation, bifascicular block (right bundle branch block and left anterior fascicular block) with mean heart rate of 50 beats/min.

Two-dimensional apical four-chamber view showing SA and its size (a and b); color Doppler image showing TR, TRPG = 68 mmHg, and TR velocity = 4.5 m/s (c). (a) SA with biventricle, (b) SA size of 106 mm, and (c) color flow Doppler showing TR, and TRPG.
Discussion
Young and Robinson reported the first case of SA in 1907 G.C., after similar cases had been reported in individuals at different age groups. 6 SA occurs as an isolated congenital heart anomaly, or as part of Ellis–Van Creveld Syndrome, trisomy 21, or heterotaxy syndrome with asplenia.4,7,8 Most patients with SA experience symptoms like shortness of breath on exertion, palpitation, recurrent upper respiratory tract infection and physical underdevelopment early in their life time. But there are also reports of adults surviving with SA. Bun et al. 3 documented a 57-year-old woman, who presented with common atrium and mitral valve cleft. Likewise, Kim et al. 7 diagnosed common atrium with atrioventricular defect in a 48-year-old man. Altıntaş et al. 4 described a 43-year-old woman, who had SA without mitral valve cleft. Similarly, Rao et al. 8 reported a 56-year-old woman, who was diagnosed with Ellis–Van Creveld syndrome, with a common atrium and common atrioventricular valve. Our case might be the oldest patient, who survived with undiagnosed SA till the age of 78 years. Reported patients with SA had no or mild cyanosis (arterial hypoxemia), despite receiving blood from both systemic and pulmonary circulation. This could suggest that there is some degree of streaming or incomplete mixing of blood within a SA, in which the more oxygenated blood is directed to the systemic circuit.1,9 Well-tolerated symptoms in the absence of arterial hypoxemia results in delayed diagnosis to late adulthood. Late diagnosis of SA increases the possibility of progression to pulmonary hypertension, heart failure, and arrhythmia, as observed in our case. Even though 2-D transthoracic echocardiography is adequate to visualize SA, 3-D transesophageal echocardiography is preferred to define anatomy of common atrium and associated anomalies.4,5,10
Conclusion
Well-tolerated symptoms of SA until late adulthood could be explained by the presence of streaming or incomplete mixing of blood within the atrium, in which the more oxygenated blood is directed to the systemic circuit.
Footnotes
Acknowledgements
We are grateful to the medical personnel who were caring for the patient.
Author contributions
All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agreed to be accountable for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual case reports.
Consent for publication
Written informed consent was obtained from the patient for publication of the case report and any accompanying images. A copy of the written consent was available for review by the Editor-in-Chief of this journal.
Informed consent
Written informed consent was obtained from the study subject.
Trial registration
Not applicable.
