Abstract
Primary lymphoepithelial carcinoma of the salivary glands is an exceptional oncological condition that predominantly emerges within the parotid gland. A significant prevalence of lymphoepithelial carcinoma has been reported in the Inuit population, along with an associated positive serology for Epstein-Barr virus in these endemic regions. In this paper, we present a case of primary lymphoepithelial carcinoma of the parotid gland in a 68-year-old female patient with a history of diabetes and hypertension residing in a non-endemic area. Histology plays a critical role in the definitive diagnosis, and confirming the primary origin of lymphoepithelial carcinoma after ruling out metastasis from undifferentiated nasopharyngeal carcinoma. The mainstay of treatment is a combination of surgical resection and adjuvant radiotherapy. Inoperable forms are typically managed with chemoradiotherapy.
Introduction
Salivary gland lymphoepithelial carcinoma (LEC) is uncommon and accounts for less than 1% of all salivary gland malignancies. 1 It has an ethnic predilection for Southeast Asian, Japanese, and Inuit population 2 with a significant association with the Epstein-Barr virus (EBV).3,4 The histological study reveals undifferentiated epithelial cells with a prominent non-neoplastic lymphoplasmacytic cell infiltration, displaying a remarkably similar histological appearance to undifferentiated nasopharyngeal carcinoma. 5
We report a case of a parotid LEC in a non-endemic country for salivary gland LEC.
Case presentation
A 68-year-old female patient with a history of diabetes and hypertension presented to our department with a painless right parotid swelling that had been progressively enlarging over several months. The patient did not report any decline in her overall health status or any instances of pyrexia.
Clinical examination showed a non-well-defined swelling measuring 4 cm in the right parotid gland. The mass was firm, adherent to the deep tissue planes with intact overlying skin. The patient had a grade 2 facial palsy based on the House-Brackmann scale. There was no involvement of cervical lymph nodes, no oropharyngeal bulging, and the Stensen’s duct was patent.
Magnetic resonance imaging (MRI) detected an irregularly contoured right parotid mass involving both lobes of the parotid gland, with hypo T1 signal and intermediate heterogeneous T2 signal. Moreover, a pronounced early enhancement with subsequent washout, demonstrating a characteristic type C curve pattern, was identified. The apparent diffusion coefficient value measured 0.6. 6 The mass exhibited extension into the intracranial space through the foramen ovale and infiltration of the retrostyloid and prestyloid spaces, as well as the infra-temporal fossa (Figure 1). Invasion was noted in the ascending branch of the mandible and the right pterygoid process. Multiple lymph node metastases were identified in the right II, III, IV, and V lymph node chains.

Magnetic resonance imaging (MRI) showing a right tumoral mass of the parotid. (a) T1-weighted sequence MRI frontal image showing the intracranial extension of the parotid tumor. (b) a T1-weighted sequence MRI axial image showing the infiltrating the right infra-temporal fossa by the parotid tumor.
Endoscopic examination and staged biopsies of the nasopharynx excluded the presence of a tumor at that site.
Given radiologic localization within the parotid gland, a parotid biopsy under general anesthesia along with a level IIa excision was performed.
Histological examination showed a poorly differentiated tumor proliferation arranged in nests and sheets exhibiting a syncytial appearance (Figure 2). The tumor cells were ovoid or polygonal with ill-defined eosinophilic cytoplasm and large atypical hyperchromatic nuclei. Prominent eosinophilic nucleoli were noted. The stroma was heavy rich in lymphoid cells (Figure 3). The tumor nests were infiltrated by lymphocytes, and follicles were frequently seen (Figure 4). Examination of the lymph node revealed tumor metastasis (Figure 5(a)) associated with a non-necrotizing granulomas of epithelioid cells and Langerhans giant cells (Figure 5(b)). Immunohistochemical analysis of the parotid tumor showed a strongly positive staining of tumor cells for pancytokeratin (Figure 6(a)), p63 (Figure 6(b)) and epithelial membrane antigen (EMA) (Figure 6(c)). The tumor cells were negative for CK7 (Figure 6(d)), Alpha Smooth Muscle Actin (SMA) (Figure 7(a)) and S-100 (Figure 7(b)). The Ki 67 proliferation index was high (Figure 7(c)). The lymphoid stromal cells stained positive for CD20 antibody (Figure 7(d)). Immunostaining for pancytokeratin highlighted tumor cells in the lymph node metastasis (Figure 8).
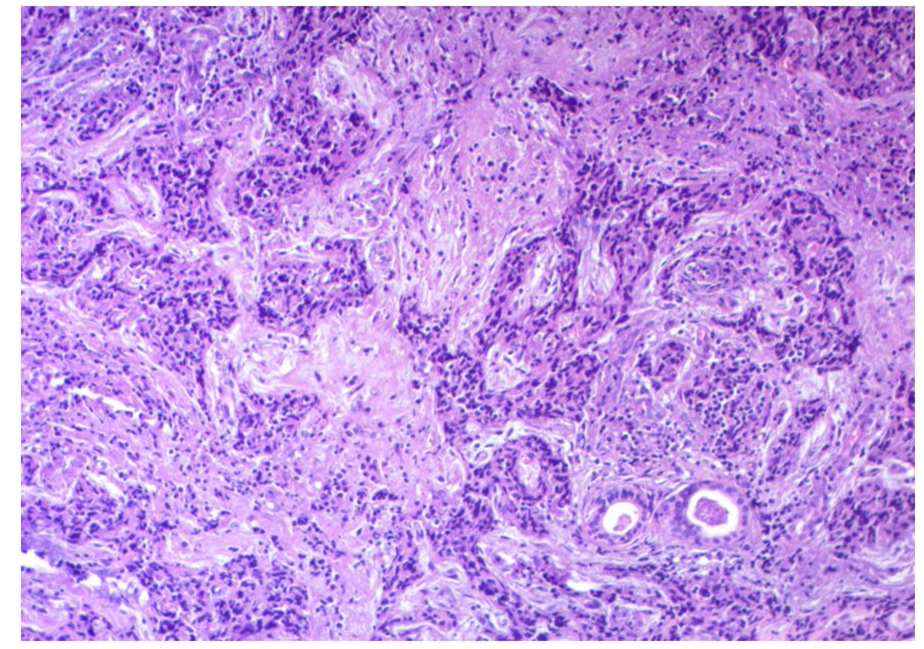
Poorly defined tumor nests and sheets separated by fibrosis and infiltrating between the residual entrapped salivary ducts (HE 100× magnification).

Tumor cells with large atypical nuclei and poorly defined borders (syncytial appearance) intermixed with dense lymphoid stroma (HE 400× magnification).

Lymphoepithelial carcinoma with lymphocytic cell infiltration in and around tumor nests; note the presence of follicles ( ) (HE 200× magnification).
) (HE 200× magnification).

(a) lymph node metastasis ( ) of syncytial tumor sheets and accompanying lymphocytes (HE 200× magnification) (b) Epithelioid granuloma with Langerhans giant cells involving the lymph node (HE 200× magnification).
) of syncytial tumor sheets and accompanying lymphocytes (HE 200× magnification) (b) Epithelioid granuloma with Langerhans giant cells involving the lymph node (HE 200× magnification).

(a) Tumor cells stain positive for pancytokeratin (×100 magnification) (b) Immunostaining for p63 shows diffuse nuclear positivity in tumor cells (×100 magnification) (c) Positive staining for epithelial membrane antigen in the majority of tumor cells (×100 magnification) (d) Negative immunoreactivity for CK7 in tumor cells (×100 magnification).

(a) Negative staining of tumor cells for SMA (×200 magnification) (b) Negative staining of tumor cells for S-100 (×100 magnification) (c) High Ki 67 proliferation index (×200 magnification) (d) lymphocytes stain positive for CD20 with presence of follicles ( ) (×100 magnification).
) (×100 magnification).

Immunostaining for pancytokeratin highlighting the tumor cells in the lymph node metastasis (×100 magnification).
The EBV serology revealed the presence of VCA IgG antibodies, while VCA IgM antibodies were negative. This serological profile is indicative of a past infection with EBV.
The diagnosis of primary LEC of the parotid with lymph node metastasis was established, and the tumor was classified as T4b N2c M0 according to the American Joint Committee on Cancer TNM Classification (8th edition). 7 The patient was subsequently referred for chemoradiotherapy. The progression has shown a reduction in the parotid swelling. A follow-up parotid MRI is scheduled in 3 months to monitor the improvement.
Discussion
Salivary gland LEC is considered an uncommon histological malignancy. Parotid localization is the most common non-nasopharyngeal location for LEC to arise.2,5,8 In the English literature, there are few reports describing parotid LEC clinicopathological particularities.
Parotid LEC exhibits an evident ethnic predisposition determining endemic and non-endemic regions. Endemic regions include Inuit and Southeast Asian populations.2,9 In these regions, parotid LEC accounts for 1.7% of all parotid gland tumors and 10% of all parotid malignancies.9–11 Non-endemic regions LEC accounts for approximately 0.4%–0.66% of all malignant salivary gland tumors.1,12 This difference of incidence is related to the rate of EBV infection as it has been reported a strong association with EBV infection and a positive serodiagnosis in almost 100% of patients in the endemic regions.2,4,8,9 In non-endemic regions, it is noteworthy that EBV-negative LECs can also be detected. 3 A relationship of the salivary gland LEC with the HIV infection 13 and the Human Papilloma Virus (HPV) infection 14 was suggested and needs to be further confirmed. In our case, the EBV serology was positive indicating a past infection by EBV, but it cannot confirm the causality of the virus in the oncogenesis of the tumor.
The most common clinical presentation of parotid LEC includes a non-tender, chronic, painless swelling of the parotid gland, as observed in our patient. The presence of facial nerve paralysis is observed in 20% of the cases, 15 and the presence of concurrent cervical lymph nodes is reported in 15% of cases.15,16
The diagnosis of LEC depends mainly on morphologic features, which exhibit sheets and cords of syncytial-like growth pattern. The tumor cells show poorly defined borders with large vesicular nuclei and prominent nucleoli. This tumor is typically characterized by lymphocytic cell infiltration, with variable density in and around tumor nests. Two main patterns of growth were described. The Schmincke-type pattern depicts a subtle epithelial component in an intimate relationship with a dense lymphoid stroma. However, the Regaud-type displays large tumor nests and sheets that are arranged within and even separated by the lymphoid cells.
Squamous differentiation, spindle cell, and basaloid features are occasionally present. Tumor necrosis may be seen. Granulomas and amyloid deposits have been reported.2,3,17,18 Immunohistochemical study is useful to confirm the epithelial phenotype of neoplastic cells and to show positive reactivity to pancytokeratin and EMA. P40 and p63 immunostaining are generally observed. 2 In LECs associated with EBV infection, tumor cells express latent membrane protein-1, a factor that perturbs the cell proliferation cycle, along with Epstein-Barr virus-encoded small RNA (EBER). In this case, tumor cells are typically positive for EBER by in situ hybridization. 19 This study was not conducted due to its unavailability in our department.
Given the similar histological and immunohistochemical features of this oncological entity with undifferentiated nasopharyngeal carcinoma, the possibility of metastasis in the salivary gland should be ruled out, along with other poorly differentiated neoplasms. 17 The differential diagnosis also arises with other lymphoepithelial lesion such as lymphadenoma, Warthin tumor, lymphoepithelial sialadenitis, and lymphoma. 2
The standard treatment for salivary gland LEC involves a combination of radical surgical excision and postoperative irradiation.9,20 This approach has been shown to be effective in achieving acceptable local control and a range of the over all survival in 5 years from 50% to 90%.2,9,20,21 In cases where the tumor is advanced and not amenable to surgery, a combination of radiation therapy and chemotherapy represents an acceptable treatment option. 22 Overall, this specific tumor category of undifferentiated carcinomas exhibits a favorable prognosis compared to other types of undifferentiated carcinomas with an approximate 80% 5-year survival rate. 1 However, regrettably, in our case, we do not have follow-up information for our patient.
Conclusion
The LEC of the parotid gland is a rare neoplasm that exhibits ethnic and geographic predilection. Its association with EBV infection suggests an etiopathogenic relationship. However, the detection of seronegative EBV LECs in non-endemic regions may raise doubts about this causality relationship.
Footnotes
Author contributions
K.S. contributed to Conceptualization, Data Curation, writing- Original Draft Preparation, supervision and Validation; A.S. contributed to Conceptualization, Data Curation, writing- Original Draft Preparation, writing-review and editing, supervision and Validation; N.S. contributed to Conceptualization, Data Curation, writing- Original Draft Preparation, writing-review and editing; M.Z. contributed to Data Curation, writing- Original Draft Preparation, writing-review and editing, and Validation; B.A.M. contributed to Conceptualization, Data Curation, writing- Original Draft Preparation; L.A. contributed to Data Curation, writing- Original Draft Preparation, Supervision and Validation; M.M. contributed to Supervision and Validation; B.T. contributed to Supervision and Validation; C.I. contributed to Supervision and Validation.
Data availability statement
The data are available with the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Patient perspective
The patient is satisfied with the treatment received.
