Abstract
Tuberculosis, being an airborne disease with a broad spectrum of symptoms and signs, is a rare cause of chylothorax. Mortality due to tuberculous chylothorax is 6%, and bilateral chylothorax has a higher mortality with a poor prognosis. A previously well 28-year-old male with a household contact history of tuberculosis exposure presented with a 1-month history of shortness of breath, fever, pleuritic chest pain, and constitutional symptoms. He was found to have bilateral chylothorax, generalized lymphadenopathy, hepatosplenomegaly, and miliary nodules in chest x-ray. Sputum acid-fast bacilli, pleural fluid TB PCR (Tuberculosis polymerase chain reaction) and culture became negative. Sputum culture became positive for Mycobacterium tuberculosis at 6 weeks. He had marked clinical and radiological improvement within 2 months of starting anti-tuberculosis treatment. Diagnosis of pleural tuberculosis is difficult as pleural fluid acid-fast bacilli detection has poor sensitivity. It is important to consider tuberculosis in patients with chylothorax and initiate prompt treatment.
Keywords
Introduction
Tuberculosis is an airborne disease caused by bacillus Mycobacterium tuberculosis and occasionally Mycobacterium bovis and Mycobacterium africanum. Once infected with M. tuberculosis, a person remains infected for life. Only 10% of those infected will develop active disease and the risk gradually lessens with time.1–5
Tuberculosis has become a global health burden and has become the 13th leading cause of death and the second most common cause of death due to an infectious disease next to COVID-19. 3 According to the World Health Organization, in 2020, an estimated 10 million people developed tuberculosis, and 1.5 million people died. The global tuberculosis incidence is gradually reducing by 2% per year, and approximately 66 million lives were saved through proper diagnosis and treatment.3,5
Chylothorax is the accumulation of chyle from the thoracic duct in the pleural space. 6 Although traumatic causes are the most common cause of chylothorax, they can rarely occur in non-traumatic causes such as malignancy, sarcoidosis, congestive heart failure, cirrhosis, and diseases of lymph vessels.7–9 Any disease that can cause mediastinal lymphadenopathy can compress the thoracic duct, causing chyle to extravasate into the pleural space. 10 Mortality due to chylothorax has been reduced from 50℅, which was recorded in 1948, and is assumed to be much less currently due to aggressive management and early diagnosis. Bilateral chylothorax carries the highest mortality rates and poor prognosis. 11
We describe a rare case of tuberculosis presenting as bilateral chylothorax, an interesting clinical condition due to the infrequent occurrence and the diagnostic challenge it poses.
Case report
A 28-year-old Sri Lankan male residing in the Middle East was admitted with high-grade fever (102–104°F) with chills, night sweats, progressive dyspnea, pleuritic chest pain, malaise, loss of appetite, and unintentional loss of weight (11 kg) for 1 month, after returning to Sri Lanka. He has been previously well and denied active or passive smoking. His mother had pulmonary tuberculosis 15 years ago. He had been well during contact tracing at that time and his Tuberculin skin test (Mantoux) had been negative.
He was ill-looking, pale, febrile, and dyspneic with peripheral oxygen saturation of 90% on room air. Mobile, non-matted small lymph nodes were palpable in the left anterior triangle of the neck and bilateral inguinal regions. Chest examination suggested possible bilateral pleural effusions, while abdominal examination found hepatomegaly of 2 cm and mild splenomegaly. These findings were confirmed with a bedside ultrasound scan.
Chest x-ray showed moderate right and small left pleural effusions and <5 mm tiny nodules affecting all zones (Figure 1). Subsequent Computed tomography scans of chest and abdomen revealed bilateral pleural effusion (right > left), multiple nodules mainly involving posterior, basal, and apical segments of the right lower lobe, and posterior basal segment of the left lower lobe, bilateral hilar and mediastinal lymphadenopathy, hepatomegaly, and splenomegaly without evidence of ascites or para-aortic lymph nodes.
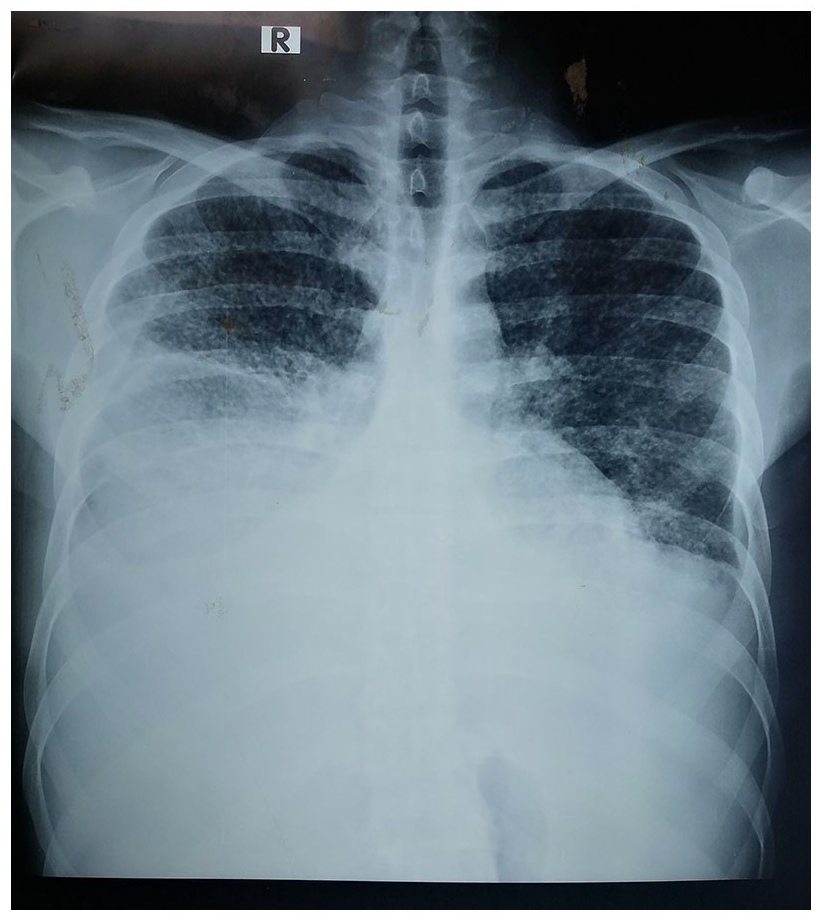
First chest x-ray shows bilateral pleural effusion with multiple lung nodules.
His Erythrocyte sedimentation rate was 9 mm/h, C reactive protein 28.9 mg/L, hemoglobin 10.8 g/dL, had mild neutrophil leukocytosis (White cell count 11.6 × 103/uL, neutrophils 90%, lymphocytes 10%), platelets 399 × 103/µL. Liver enzymes were mildly elevated (Aspartate transaminase 64 U/L, Alanine transaminase 55 U/L) and serum creatinine was normal (70.4 µmol/L).
The patient underwent therapeutic needle aspiration of approximately 550 ml of milky white pleural fluid on the right side. It was exudative with lymphocytic predominance (WBC count 3440, RBC 1600, polymorphs 18, lymphocytes 82, pleural protein level 3.69 g/dL, sugar 151 mg/dL). Pleural triglyceride (TG) was 1374 mg/dL, pleural total cholesterol was 70 mg/dL, and pleural Lactate dehydrogenase was 432 U/L. Serum TG was 137 mg/dL (pleural fluid: serum TG = 10) and cholesterol was 156 mg/dL (pleural fluid: serum cholesterol = 0.44). The serum LDH count was 386 U/L. Pleural fluid was negative for atypical cells and bacterial growth. Pleural fluid for Adenosine diaminase was 27.7 IU. Antibodies for melioidosis and HIV were negative.
A diagnosis of disseminated/miliary tuberculosis complicated with bilateral chylothorax was made on high clinical suspicion with biochemical and radiological evidence, and the patient was started on Anti tuberculous therapy according to the National Guidelines for Tuberculosis Control (2021) 2 on Day 06 of admission. To confirm the diagnosis, pleural fluid TB PCR (tuberculosis polymerase chain reaction), sputum for Acid fast bacilli and TB PCR was done, which was negative. Sputum for TB culture was sent. The patient’s Mantoux test became positive with 18 mm induration after 48 h. Due to cervical lymph nodes being small (0.5–1 cm), a biopsy could not be done.
The patient’s symptoms persisted with oxygen dependency despite pleural aspiration and continuation of ATT, and serial bedside ultrasound scans of the chest confirmed recurring bilateral moderate pleural effusions, which warranted bilateral intercostal tube insertions on Days 12 and 16 of admission for continuous pleural drainage. Both intercostal tubes tubes drained 300–700 ml/day per side for approximately 21 days, after which the drainage gradually subsided, and the tubes were removed on Day 28. During the intense period, he was provided with a high-calorie, low-fat, medium-chain triglyceride-containing diet, chest physiotherapy, and mobilization advice to prevent thromboembolism.
Meanwhile, a diagnostic thoracoscopy was performed on Day 15 (Figure 2), which revealed densely inflamed parietal and visceral pleurae and enlarged hilar and mediastinal lymph nodes with caseous discharge. Biopsies taken from the parietal pleura and mediastinal nodes showed inflamed pleural tissue and xanthogranulomatous lymphadenitis.

Mediastinal lymph nodes with caseous discharge in thoracoscopy.
The patient was discharged on Day 32 of ATT, and his sputum culture became positive for M. tuberculosis at 6 weeks of incubation, confirming the diagnosis of tuberculosis. At the 2-month review, the patient had much improved symptomatically, and the ultrasound scan was negative for pleural effusions. ATT continuation phase was extended to 10 months for disseminated TB.
Discussion
Tuberculosis is a rare cause of chylothorax, with only 37 cases reported in literature.12–14 Vennera, in 1983, was the first reported case of tuberculosis infection causing chylothorax. 15 Mortality due to tuberculous chylothorax (TC) is 6%. 12 Out of the 37 patients summarized, two died, while the remaining 35 had a good prognosis with appropriate treatment.
Diagnosis of pleural tuberculosis is often difficult and has poor sensitivity because AFB detection is mostly negative, pleural fluid cultures and blind biopsies are positive in only 17%, and ADA levels are positive in only 40%. 16 However, Christopher et al. in 2018 described that thoracoscopic guided biopsies are 95% positive for tuberculosis, which is also confirmed in cases of TC.
The pathogenesis of chylothorax in patients with tuberculosis is not well understood. It is assumed to be associated with the compression of the thoracic duct and cisterna chyli by mediastinal and hilar lymph nodes. Due to the compression exerted by the lymph nodes, the pressure inside the lymphatics increases, resulting in the leaking of chyle into the pleural cavity.16–18
Milky pleural fluid should be differentiated from chylothorax, pseudochylothorax, and empyema. 19 Pseudochylothorax is an accumulation of cholesterol crystals due to chronic pleurisy. Hence, the pleura is commonly markedly thickened. 7 Empyema develops commonly as a complication of bacterial pneumonia. It is of acute onset and has a strong odor, unlike chyle. Chylothorax and pseudochylothorax can be biochemically differentiated by lipid-level analysis of effusion. Chylothorax will have high triglyceride levels (more than 1.24 mmol/L or 110 mg/dL) and chylomicrones are usually present. In pseudochylothorax, cholesterol levels will be high (more than 5.18 mmol/L or 200 mg/dL) and cholesterol crystals will be present. The presence of cholesterol crystals is diagnostic of pseudochylothorax irrespective of triglyceride levels. 19 Tuberculosis is known to cause both chylothorax and pseudochylothorax. 19
The management of non-traumatic chylothorax includes drainage of the large or symptomatic effusions and decreasing the chyle production. Drainage can be done with repeated thoracocentesis, tube drainage, or pleuroperitoneal shunts 20 while chyle production can be decreased with surgical methods such as ligating the thoracic duct during thoracoscopy, thoracic duct embolization or by medical treatment, which includes chemical pleurodesis, chronic pleural catheters, low-fat nutrition, and octreotide.21,22
Needle thoracocentesis is the initial step in the management of symptomatic chylothorax, but intercostal tube drainage has become the preferred method of drainage in most of the centers. It is recommended to go for surgical treatment methods if the drainage exceeds 1.5 L. 10
It is important to maintain a proper diet in chyle loss as it can cause immunosuppression and malnourishment, which can be disabling. Tuberculosis itself is a disabling disease that causes rapid loss of weight and loss of appetite. Hence, nutritional support plays a major role in preventing malnutrition and minimizing chyle production and flow. Usually, patients are advised to take a diet rich in low-fat, medium-chain triglycerides for the management of chylothorax while it is important to take a high-calorie, high-protein diet for patients with tuberculosis.10,23
The initiation of ATT was done according to the guidelines of the National Tuberculosis Program conducted in Sri Lanka. 2 The patient was started on three tablets of FDC 4 (fixed-dose combination) daily for intensive phase treatment according to his current weight. The tablets are continued for 2 months of the intensive phase and converted to FDC 2 tablets for the continuation phase of treatment. FDC 2 should be continued for 10–12 months in patients with miliary TB, depending on response. ATT is associated with a survival rate of 94.4%.
Conclusion
Tuberculous infection should be considered in patients presenting with bilateral chylothorax without a history of trauma or thoracic surgery, especially in countries where tuberculosis is an endemic disease. It highlights the importance of having high-grade suspicion and vigilance in diagnosing tuberculosis, even when biochemical and microbiological investigations become negative, to start ATT in a timely manner.
Footnotes
Acknowledgements
None.
Author contributions
W.M.M.H.P.W. and R.M.S.N.R. were involved in the management of patients, data collection, and drafting of the manuscript. B.M.D.G.Y. was involved in the management of patients, supervision, and drafting of manuscript and revision of manuscript. All authors approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) before the collection of information for their anonymized information to be published in this article.
