Abstract
Chronic pulmonary aspergillosis is a lung disorder characterized by the presence of single or multiple cavities with or without an aspergilloma or nodules on chest imaging, with mycological evidence of and/or demonstration of immunological response to Aspergillus spp. The affected patient should manifest relevant symptoms for at least 3 months. Chronic cavitary pulmonary aspergillosis is the most common subset of chronic pulmonary aspergillosis, which is often reported in patients previously treated for pulmonary tuberculosis, having residual cavities in their lungs. We present a case of a 55-year-old male patient treated for pulmonary tuberculosis 2 years back, now presenting with fever, shortness of breath, and hemoptysis with overt radiological changes from the baseline, positive direct microscopy, and serology for Aspergillus spp. and thus meeting the criteria for chronic cavitary pulmonary aspergillosis. Treatment with oral antifungal was initiated, but the follow-up data are unavailable due to patient noncompliance and lack of resources. We aim to emphasize the radiological and microbiological features of this condition to aid the early diagnosis and prompt treatment, as this may mimic similar pulmonary disorders and pose a significant challenge in the diagnosis and management outcomes.
Introduction
The most common form of chronic pulmonary aspergillosis (CPA) is chronic cavitary pulmonary aspergillosis (CCPA). 1 The definition of CCPA requires the presence of single or multiple pulmonary cavities, which can be either thin- or thick-walled, containing one or more aspergilloma, with microscopy or culture-proven Aspergillus spp., significant pulmonary, and/or systemic symptoms and overt radiological progression over the last 3 months, which can include new cavities, increased pericavity infiltrates, or fibrosis. 2 The patients are usually immunocompetent and present with worsening shortness of breath, weight loss, and hemoptysis. Fever is unusual and, if present, indicates an accompanying bacterial or viral infection. Diagnosis usually involves radiological evidence from chest X-rays or high-resolution computed tomography (HRCT) of the chest, confirmed by Aspergillus IgG, which is highly sensitive and specific for the diagnosis of CCPA. 1 The culture of sputum for Aspergillus also provides diagnostic evidence. However, it should be supported with a positive culture of the bronchoscopic specimens, as many conditions can yield Aspergillus in the sputum. The treatment of CCPA requires long-term therapy with oral azole antifungal agents, either with voriconazole or itraconazole.3,4
Case presentation
A middle-aged man in his 50s presented to the outpatient department of a teaching hospital with complaints of fever, progressive shortness of breath, weight loss, and worsening hemoptysis over the last 7–8 months. There was a history of pulmonary tuberculosis treated with 6 months of the first-line regimen, that is, 2 months of isoniazid (H), rifampicin (R), pyrazinamide (P), and ethambutol (E), followed by a continuation phase with 4 months of isoniazid (H) and rifampicin (R), (2HRZE/4HR) 2 years back. No history of smoking was present. He was a known hypertensive and diabetic with no other comorbidities. He was previously seeking care from a local practitioner who put him on antituberculosis treatment 2 months back, but he presented with worsening complaints.
On arrival, his blood pressure was 140/90 mmHg, pulse was 75 bpm, respirations were 25/min, and temperature was 100°F. Oxygen saturation was 92% on room air. There was reduced air entry bilaterally on auscultation, and heart sounds were normal. Routine blood investigations revealed mild leukocytosis (WBC = 13.2 cells/mL) with neutrophilic predominance, hemoglobin was 9.8 g/dl, and platelet count was 275 cells/mm3. The erythrocyte sedimentation rate was 90 mm/hr. Renal and liver function tests were within normal limits. Arterial blood gas revealed uncompensated respiratory alkalosis. The patient was HIV-negative and had never been on chemotherapy or immunosuppressive drugs.
A radiological workup was ordered and compared with the previous records available to document the progression of the disease and assist in the diagnosis. The chest X-ray performed 5 months back suggested poorly defined bilateral cavitations with progression in the new chest X-ray to multiple cavitations with dense pericavity infiltrates on the left side (Figure 1). HRCT of the chest revealed similar comparative findings. A 5-month-old computed tomography (CT) chest was suggestive of well-defined right-sided cavitation with an aspergilloma and relative sparing of the left side. In the present-day CT chest, this had progressed to a static cavitary lesion on the right side with findings compatible with an aspergilloma, with the formation of a confluence of multiple cavities having an aspergilloma and pericavity infiltrates on the left side (Figure 2).
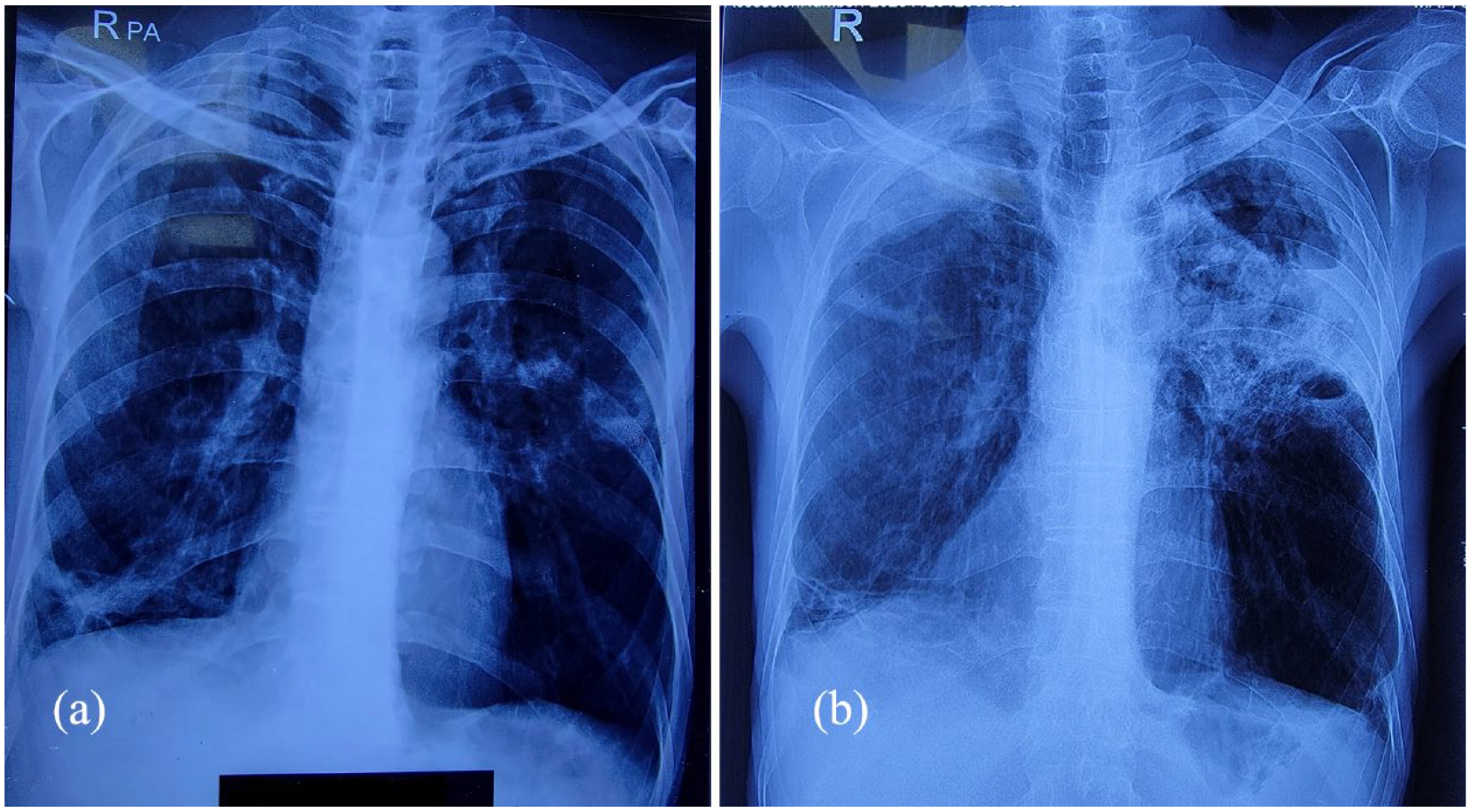
X-ray chest suggestive of bilateral ill-defined cavitations (a). Progression to left-sided multiple cavitary lesions with dense pericavity infiltrates (b).
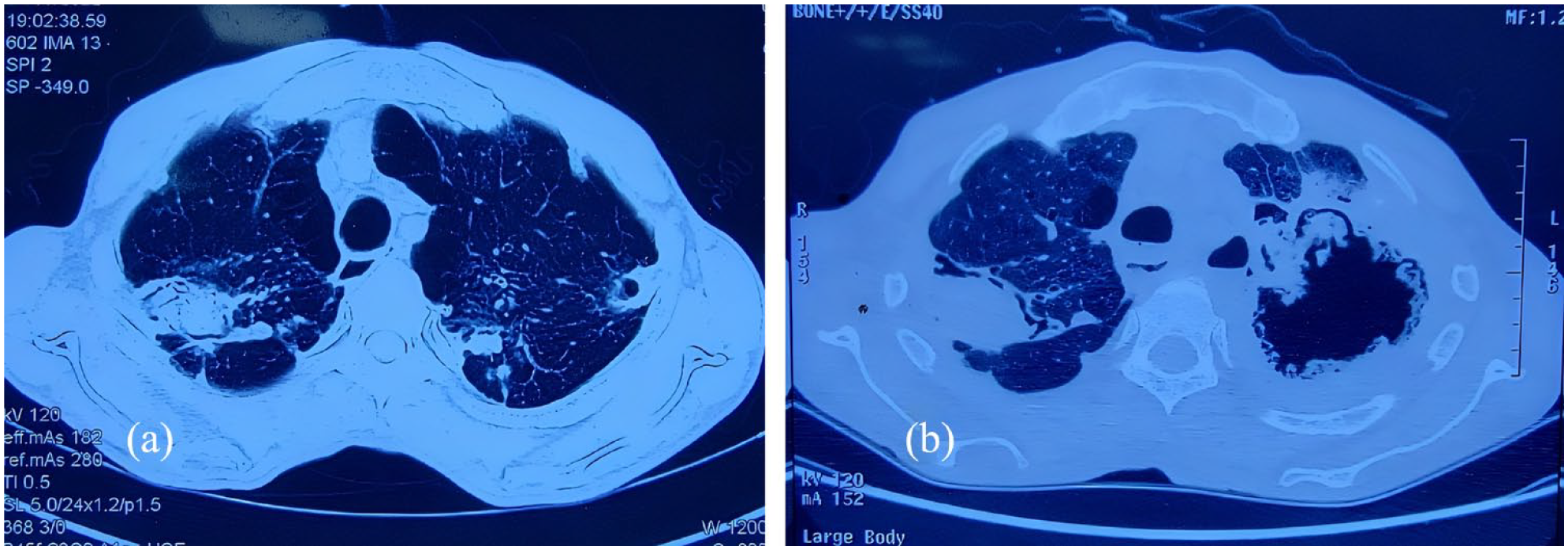
High-resolution computed tomography chest suggestive of right lung cavity with an aspergilloma (a). Progression to the left-sided confluence of cavities with an aspergilloma and pericavity infiltrates (b).
Considering a high suspicion of fungal etiology, Aspergillus IgG and sputum culture for fungus were sent. Aspergillus IgG, performed by automated enzyme immunoassay (Siemens Immulite 1000), came out positive, and thus, the diagnosis of CCPA was made considering the radiological progression and the positive Aspergillus IgG, which was later reinforced by the presence of Aspergillus niger in the fungal culture performed by the conventional method. The patient was initially misdiagnosed as a case of pulmonary tuberculosis relapse as the sputum for GeneXpert came out to be negative in three successive samples. The patient was started on oral itraconazole 200 mg twice daily for 6 months and was advised to follow-up after 3 months. Although the sputum culture for bacteria was negative, antibiotics were prescribed for the superadded bacterial infection as the patient was febrile on presentation and neutrophilic leukocytosis was present. Other treatment options, including intravenous antifungal therapy, local cavity therapy, or surgery, were not employed due to the poor socioeconomic status of the patient.
Discussion
CPA represents a rarely encountered group of pulmonary disorders manifesting as a consequence of a long-term pulmonary fungal infection. 5 The routinely encountered form of CPA is CCPA, which, if left untreated, can transform into chronic fibrosing pulmonary aspergillosis which in addition to the radiological features of CCPA comprises fibrotic destruction of a minimum of two lung lobes, fibrosis of a single lung lobe will be still considered as CCPA. 1 Other manifestations of CPA can include simple aspergilloma and Aspergillus nodules on thoracic imaging. All of these features mentioned above are present in immunocompetent patients. A severe form of CPA can occur in immunocompromised individuals and is referred to as subacute invasive aspergillosis, also known as chronic necrotizing pulmonary aspergillosis, hallmarked by the invasion of fungal hyphae into the lung tissue and positive Aspergillus galactomannan in the blood. 6 The topmost differential includes pulmonary tuberculosis, which should be adequately investigated, but both can occur simultaneously.7,8
The diagnosis of CCPA requires radiological evidence of a progression of lesions within 3 months of observation, including the formation of new cavities, pericavity infiltrates, or fibrosis from preexisting single or multiple cavities with one or more aspergillomas. 9 If the radiological progression is highly suggestive of CCPA, then any of the following investigations can be done to confirm the diagnosis: a positive Aspergillus IgG, strongly positive Aspergillus antigen or DNA in respiratory secretions, biopsy showing fungal hyphae on microscopy, or growing Aspergillus spp. from a cavity. Aspergillus IgG by enzyme immunoassay is highly specific and sensitive in patients with characteristic radiological findings. People residing in areas where the prevalent Aspergillus species are nonfumigatus, can have a false-negative serology. IgG tests that target the nonfumigatus species can be helpful in these patients. A positive Aspergillus PCR supports the diagnosis but is not enough alone because colonization and contamination could result in a positive Aspergillus PCR beyond the other forms of aspergillosis. The culture of sputum, growing Aspergillus, is supportive of the diagnosis but not confirmatory, as other conditions may yield Aspergillus in sputum. However, the culture of bronchoscopic specimens for Aspergillus is more indicative of CCPA. Aspergillus galactomannan assay in bronchoalveolar lavage specimens can also support the diagnosis of CCPA.10,11
Treatment in CCPA aims to manage hemoptysis and prevent the progression to fibrosis effectively. Oral antifungal therapy is with either voriconazole or itraconazole, as they are considered active against the Aspergillus species. If these are not available, posaconazole or isavuconazole may be considered. Although not statistically significant, the latest clinical literature favors the use of voriconazole over itraconazole, due to better clinical outcomes and tolerance.12,13 The duration of oral therapy is 6 months, which can be prolonged in nonresponders and immunocompromised patients. Other treatment options include intravenous antifungal therapy, instillation of an antifungal agent into an aspergilloma cavity, adjunctive interferon-gamma therapy to cure the immune defects noticed in CPA, and the surgical removal of aspergilloma as a last resort. Hemoptysis can be treated by oral tranexamic acid, but extreme cases may require bronchial artery embolization. The effect of treatment in the case of CCPA is slow; the response can be seen as long as 3 months. Therefore, regular radiological follow-ups every 3–6 months are required to monitor the duration of oral therapy. 14
The limitation of this study is the loss of the follow-up data due to various reasons, including patient relocation, noncompliance, and lack of resources.
Conclusion
To mitigate the consequences of this severe illness, CPA must be appropriately diagnosed and followed up on, mainly in low-income nations where the prevalence is much higher. In these situations, where healthcare resources are limited, early detection of CPA can significantly enhance patient outcomes and ease the load on already overwhelmed healthcare systems. The implementation of easily accessible diagnostic tools and increased awareness among healthcare providers is pivotal due to the challenges posed by underdiagnosis and lack of awareness.
Footnotes
Acknowledgements
Not applicable.
Author contributions
M.W. contributed to design, analysis, drafting, critical revision, and final approval of the manuscript; N.I.B. contributed to design, drafting, critical revision, and final approval of the manuscript; A.U. contributed to design, drafting, and final approval of the manuscript; Z.T. contributed to design, drafting, and final approval of the manuscript; A.I. contributed to design, drafting, and final approval of the manuscript; O.A. contributed to design, drafting, and final approval of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
