Abstract
Reversed flow in the basilar artery can be acquired or congenital. Acquired reversed flow in the basilar artery can result from acute thrombosis of the basilar artery or retrograde vertebral artery flow. Congenital continuous retrograde basilar artery flow has not been described. We report a 2-day-old male presenting with hypocalcemic seizures which led us to obtain a Duplex echoencephalogram. An echocardiogram was subsequently ordered. In the coronal plane through the anterior fontanelle, retrograde flow was seen in the basilar artery and the right vertebral artery. In the axial plane through the temporal window, the flow was anteroposterior in both posterior communicating arteries. In the posterior cerebral arteries, the flow was retrograde in the P1 segment and anterograde in the P2 and P3 segments. An interrupted aortic arch was suspected. The echocardiogram showed a large perimembranous ventricular septal defect with bidirectional shunting, a hypoplastic and bicuspid aortic valve, an aortic arch interrupted between the left common carotid artery and the left subclavian artery (type B interrupted aortic arch), and a 5 mm patent ductus arteriosus with predominant right to left flow. Because of the patency of the large patent ductus arteriosus, our patient showed no sign of posterior circulation insufficiency. Prostaglandin E1 therapy was initiated immediately. Diagnosis of DiGeorge syndrome was proven. The infant underwent interrupted aortic arch repair and anterograde flow was established in the basilar artery. We conclude that congenital asymptomatic continuous retrograde flow in the basilar artery and left vertebral artery is a medical emergency as it implies the presence of type B interrupted aortic arch with large patent ductus arteriosus in a neonate.
Introduction
Acquired reversed flow in the basilar artery (BA) can result from acute thrombosis of the BA or retrograde vertebral artery (VA) flow. In patients with acute vertebrobasilar ischemia, continuous flow reversal in the BA is considered a favorable indicator of recovery. 1 Reversed flow in the BA can be intermittent in patients with proximal subclavian/innominate artery occlusion or stenosis. When significant disease is present in the innominate/brachiocephalic or proximal subclavian arteries, blood must course up the contralateral VA, cross over at the BA, and course down the ipsilateral VA to perfuse the arm. Retrograde VA flow increases with ipsilateral upper extremity exercise or by placing a blood pressure cuff on the arm for a few minutes, then releasing it. When retrograde VA flow increases, the subclavian steal phenomenon can become symptomatic due to the acute basilar flow reversal. 2 A congenital continuous retrograde basilar flow has not yet been described.
Case report
A large for gestational age (4184 g) Caucasian female was born via spontaneous vaginal delivery after artificial rupture of membrane at 38 weeks and 2 days, to a 31-year-old gravida 4 para 4 with A2 gestational diabetes mellitus. The Apgar scores were 8 and 9 at 1 and 5 min, respectively. At delivery, the infant was vigorous, and interventions included bulb syringe suctioning, warming, and stimulation. Hypoglycemia prophylaxis with oral glucose gel was administered successfully. The baby was admitted to the neonatal intensive care unit on day 2 of life for seizure-like activity characterized by rhythmic intermittent multifocal jerking of extremities that was associated with episodes of desaturations. Diagnosis of neonatal seizure prompted a septic workup including a spinal tap as well as echoencephalography.
The infant was loaded with phenobarbital and started on antibiotics and acyclovir. An emergency duplex echoencephalography (at 49 h of life) showed normal brain anatomy with non-specific lenticulostriate vasculopathy (not shown). In the axial plane through the temporal window (Figure 1), the flow was anteroposterior in both posterior communicating arteries (PCOMs). In the posterior cerebral arteries (PCAs), the flow was retrograde in the P1 segment and anterograde in the P2 and P3 segments. In the coronal plane through the anterior fontanelle, Duplex power Doppler echoencephalography (Figure 2(a)) showed retrograde flow in the BA. In the same plane, color Doppler echoencephalography (Figure 2(b) with blue indicating flow away from the transducer) showed retrograde flow in the BA and left VA. Flow remained anterograde in the right VA. An echocardiogram was recommended to evaluate the aortic arch, as an interrupted aortic arch (IAA) was suspected. Blood pressures were noted to be low in the lower extremities (systolic BP/diastolic BP: 50/30 mmHg; mean arterial pressure: 37), but blood pressure in the right arm could not be checked due to the IV site.

Duplex echoencephalography using transcranial power Doppler through the anterior fontanelle (green) and left temporal window (red). Blood supply to the entire brain depends on the internal carotid arteries (ICAs). There is anteroposterior flow in the posterior communicating arteries. Flow is maintained in the P2 segment of the posterior cerebral arteries (PCAs). Flow reversal is seen in the P1 segment of the PCAs and the basilar artery.

Duplex power Doppler echoencephalography (a) through the anterior fontanelle showed reverse flow in the basilar artery (BA). Color Doppler echoencephalography (b), with blue indicating flow away from the transducer, showed that reverse flow in the BAs was accompanied by reversed flow in the left vertebral artery (VA) while flow remained anterograde in the right VA. Diagnosis of interrupted aortic arch is suspected.
Prenatal ultrasound did not show an IAA. The echocardiogram showed a large perimembranous ventricular septal defect (VSD) with bidirectional shunting (not shown), a hypoplastic and bicuspid aortic valve (not shown), a 5 mm patent ductus arteriosus (PDA) with predominant right to left shunting, and an aortic arch interrupted between the left common carotid artery and the left subclavian artery (type B IAA) (Figure 3). There was no clinical evidence that the ductus arteriosus was getting restricted as saturation measurements in the lower extremities and left arm were not diminished. Because of the patency of the large PDA, our patient showed no sign of posterior circulation insufficiency.

Suprasternal echocardiogram in normal control ((a) and (b)). The ascending aorta branches into the brachiocephalic trunk, the left common carotid artery, and the left subclavian artery before becoming the descending aorta. Suprasternal echocardiogram in the patient with type B interrupted aortic arch (IAA) ((c) and (d)). The hypoplastic ascending aorta becomes a single innominate artery (bovine aortic arch) that terminates in a brachiocephalic artery and a left carotid artery. The white arrow indicates where the aortic arch is interrupted. Below the IAA, the patent ductus arteriosus provides deoxygenated blood to the lower extremities.
Laboratory showed normal glucose (68 mg/dL) and low ionized calcium (0.7 mmol/L;
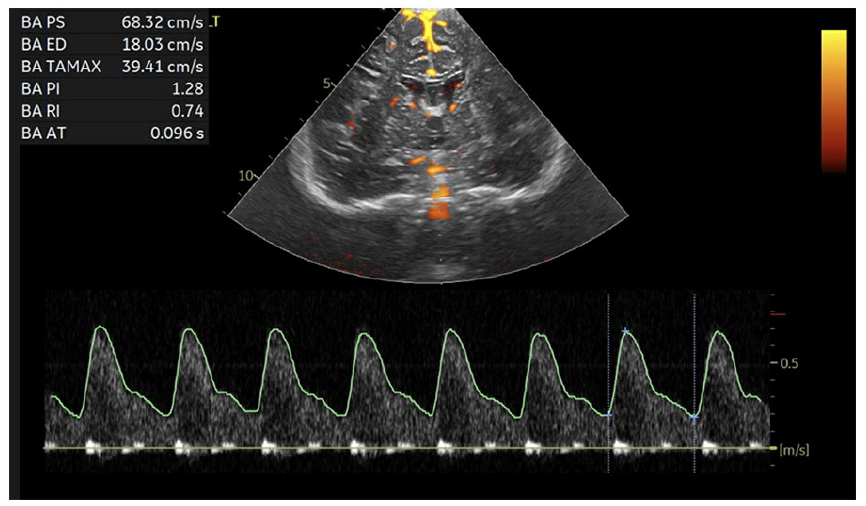
Duplex power Doppler echoencephalography through the anterior fontanelle showing anterograde flow in the basilar artery after surgical correction of the interrupted aortic arch.
Discussion
In our institution, duplex echoencephalography is performed before electroencephalographic evaluation in neonates with seizures. A nonspecific lenticulostriatal vasculopathy most likely related to maternal diabetes was found (not shown). Power Doppler Duplex echoencephalography through the temporal window showed retrograde flow in the PCOMs suggesting that the predominant blood supply to the PCAs arose from the ICAs. If our patient had the fetal PCOM variant, we would have expected that the P1 segment of the PCAs would have been hypoplastic. 3 Instead we discovered that the P1 segment was normally developed and flow was retrograde indicating that the predominant blood supply to the BA arose from the ICAs (Figure 1). Power Doppler through the anterior fontanelle confirmed the presence of continuous retrograde flow in the BA (Figure 2(a)). An emergency echocardiogram was requested to confirm our initial intuition, the presence of an IAA. (Figure 1(a)).
IAA is defined as an interruption in anatomical continuity between the ascending and descending aorta. Clinically, IAA should be suspected when differential pulses or blood pressure are noted in the extremities. Such a differential was not appreciated by our patient. IAA accounts for about 1.5 % of all congenital heart defects. The classification made by Celoria and Patton 4 is the most widely used, based on the position of interruption that can be distal to the left subclavian artery (type A), between the left common carotid and left subclavian arteries (type B), or between the innominate and left common carotid arteries (type C). In most cases, IAA is associated with additional cardiovascular anatomic defects. PDA, VSDs, bicuspid aortic valve, and hypoplasia of the ascending aorta are frequently associated with IAA. Approximately 50% of all IAA cases are associated with a chromosome 22q11.2 deletion. This chromosomal abnormality is seen in up to 75% of patients with type B IAA. 5 IAA affects up to 42% of individuals with DiGeorge syndrome. 5 In our patient, diagnosis of DiGeorge syndrome was suggested due to the combination of cleft palate, hypocalcemia-induced seizures, and IAA. The diagnosis was confirmed by FISH analysis.
Abnormalities of the subclavian arteries are seen in more than 60% of patients with DiGeorge syndrome and the most frequent abnormality is the aberrant origin of the left subclavian artery from the descending aorta. 6 Cervical origin of the right subclavian artery is detected in most patients with type B IAA and exclusively in patients with monosomy 22q11. 6 In rare cases, right VA originates from the aortic arch distal to the origin of the left subclavian artery and has an aberrant retro-esophageal course. 5 In our patient, continuous retrograde flow was seen on color Doppler echoencephalography in the BA and left VA consistent with either an aberrant origin of left VA from the descending aorta or a normal origin from the left subclavian artery. Flow remained anterograde in the right VA (Figure 1(b)) excluding the possibility of an aberrant retropharyngeal right VA arising from the descending aorta. After surgical correction of the IAA, flow in basilar and vertebral arteries became anterograde.
The signs and symptoms of IAA occur when the ductus arteriosus begins to close. This occurs 3–4 days after birth. Mean patient survival is approximately 4–10 days if untreated. 5 Posterior circulation insufficiency is expected after closure of the ductus arteriosus, leading to hypoperfusion-related complications and eventually death. 5 Our patient was still asymptomatic when the diagnosis was made. Saturation measurements in the legs were not diminished as ductus arteriosus was still widely patent. Treatment with prostaglandin was initiated as soon as diagnosis was established, and surgery was performed within days.
Conclusion
We conclude that congenital asymptomatic continuous retrograde flow in the BA and left VA is a medical emergency and evaluation of the aortic arch should be performed without delay. Congenital asymptomatic continuous retrograde flow in the BA and left VA or both VAs implies the presence of type B IAA with a large PDA in a neonate.
Footnotes
Author contributions
P.M. is the principal senior author, who conceptualized the article, searched literature, and drafted the initial manuscript. K.S. contributed to the literature review, critical review, and construction of the manuscript. D.S., K.D., and J.S. contributed to the literature review, manuscript writing, and data gathering. All authors approved the final manuscript as submitted and agreed to be accountable for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient’s legally authorized representative, in this case parent for their anonymized information to be published in this article.
