Abstract
Neurofibromatosis type I and multiple sclerosis, when considered separately, are associated with a higher risk of cerebrovascular accident. The coexistence of neurofibromatosis type I and multiple sclerosis may lead to a further increase in cerebrovascular risk; however, this has not been reported in the literature. We report the case of a 37-year-old woman affected by both neurofibromatosis type I and multiple sclerosis: she was referred to our rehabilitation department because of a recent event of ischemic stroke. Moreover, we provide a comprehensive and updated review of all published cases reporting the coexistence of neurofibromatosis type I and multiple sclerosis to gather information regarding their association with cerebrovascular accidents.
Introduction
Neurofibromatosis type I (NF-1) is a rare complex multisystem human disorder caused by the mutation of a gene on chromosome 17 responsible for neurofibromin production, a protein that is needed for normal function in many human cell types, including neurons. 1 Multiple sclerosis (MS) is the most frequent demyelinating disease affecting young people; characterized by widespread myelin damage and neurodegeneration in the brain and spinal cord. 2 Cerebrovascular accidents (CA) encompass a variety of medical conditions that affect cerebral perfusion or vasculature. 3 These three pathologies cause functional impairment and disability.
Compared to the general population, the odds of any type of stroke are increased in both adult and pediatric patients with NF-1. 4 It has not been reported whether the coexistence of NF-1 and MS possibly determines a further increase in cerebrovascular risk.
We report the case of a 37-year-old woman suffering from NF-1, MS, and a recent ischemic stroke. Moreover, we provide a comprehensive and updated review of all published cases reporting the coexistence of NF-1 and MS to gather information on their association with CA.
Case report
A 37-year-old woman with both NF-1 and MS was referred to our rehabilitation department because of a recent ischemic stroke.
The patient’s neurological history began at the age of 19: NF-1 was diagnosed after the removal of a plexiform neurofibroma in the right foot; a glioma of the left optic nerve and café au lait macules were also detected. No genetic confirmation of the NF1 was performed.
Ten years later, she underwent a neurological reevaluation due to the onset of balance impairment, gait ataxia, and mild paresis of the left lower limb. Brain and spinal cord magnetic resonance imaging (MRI) revealed several demyelinating lesions (left

(a) Axial T2 brain MRI showing multiple white matter inflammatory lesions. (b) Coronal brain MRI showing hypodensities in the left thalamus. (c) Sagittal brain MRI showing multiple white matter inflammatory lesions.
Over time, the patient also developed hypertension and dyslipidemia, known risk factors for stroke.
No family history of stroke, smoking, atrial fibrillation, and/or atrial heart disease were reported.
At the age of 37 years, the patient was referred to the emergency room because of the sudden onset of right upper limb paresis, speech and swallowing impairment, and gait ataxia. The first computed tomography scan excluded hemorrhagic lesions. The acute clinical presentation suggested a cerebrovascular event rather than an MS
Ten days after the stroke, the patient was transferred to our post-acute rehabilitation facility due to left-sided hemiparesis, balance impairment, speech, and swallowing disorders. Functional follow-up was conducted up to 3 years after the stroke.
The most evident improvements in balance control, stride length, and walking speed were recorded during the first 5 months after the stroke. The patient’s functioning remained stable at follow-up visits performed 1 and 2 years later (Table 1).
Most evident progressive improvements in balance control, stride length, and walking speed were recorded during the first 5 months after the stroke.

NP: not performed.
For m-FIS: the higher the score, the worse the fatigue.
For Equiscale and FIM: the higher the score, the better the performance.
We conducted a literature review following the PRISMA guidelines. The study population consisted of patients with a confirmed diagnosis of MS and clinical NF1. We searched the following databases for relevant studies: PubMed, Medscape, and Embase. Since NF-1 is a rare disease, we also searched the
The diagnostic criteria for NF1 are met in an individual who does not have a parent diagnosed with NF1 if two or more of the following are present: six or more café-au-lait macules over 5 mm in greatest diameter in prepubertal individuals and over 15 mm in greatest diameter in postpubertal individuals, freckling in the axillary or inguinal region, two or more neurofibromas of any type or one plexiform neurofibroma, optic pathway glioma, two or more iris Lisch nodules identified by slit lamp examination or two or more choroidal abnormalities, a distinctive osseous lesions such as sphenoid dysplasia, anterolateral bowing of the tibia, or pseudarthrosis of a long bone, a heterozygous pathogenic NF1 variant with a variant allele fraction of 50% in apparently normal tissue such as white blood cells. 6
Differential MRI features between MS lesions and Focal Areas of Signal Intensity (FASIs) of NF1 are summarized in Table 2.
Differences in the NMR of MS lesions from FASIs in NF1.
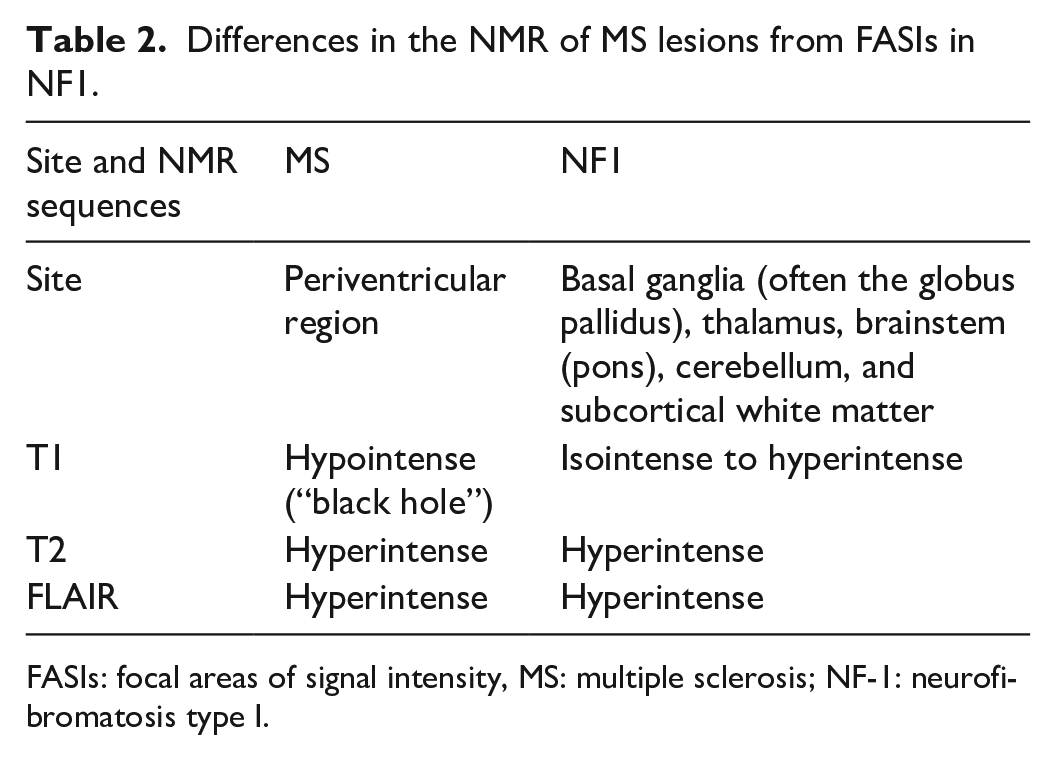
FASIs: focal areas of signal intensity, MS: multiple sclerosis; NF-1: neurofibromatosis type I.
The selected studies included 42 patients (23 women, 11 men, and 8 unspecified). The main inclusion criteria were a confirmed diagnosis of NF1 and MS (
Genetic data.

MS: multiple sclerosis; NF-1: neurofibromatosis type I; PPMs: primary progressive.
An American administrative claims analysis reported 25 cases of MS among the 8552 subjects affected by NF-1: no clinical and/or genetic information was available for these patients; therefore, we excluded them from the subsequent review. 11
Only one study out of 23 reported two cases of CA: a 42-year-old woman with a hemorrhagic lesion in the right posterior limb of the internal capsule 12 and a 62-year-old woman who died due to ruptured brain aneurysm (Table 4). 12
Literature selection for the synthesis of results.

Discussion
We reported an uncommon case of a patient affected by NF1, MS, and ischemic stroke. Including our patient, only 43 cases of coexisting NF-1 and MS have been reported in the literature, of which three cases of associated NF-1, MS, and CA.
The coexistence of NF-1 with MS has been reported to be more common in the primary progressive form than in the relapsing-remitting form. 20
From a rehabilitation perspective, patients with NF-1 and MS face a progressive functional worsening, which current treatments can only counteract for a short period. Differently, CA-related impairment and disabilities, if properly and timely treated 30 in the post-acute phase, improve and stabilize over time, as seen in this case.
According to existing literature, treatment with subcutaneous interferon beta 1a (IFN-β1a) does not increase the risk of stroke in patients with MS. 31 Muhannad et al. reported a case of alemtuzumab-induced simultaneous onset of autoimmune hemolytic anemia, alveolar hemorrhage, nephropathy, and stroke in a 52-year-old man that occurred 8 months after treatment initiation. 32 On the other hand, dimethyl fumarate (DMF) is hypothesized to provide clinical benefit to MS patients in case of ischemic stroke. 33
A small body of evidence suggests that the association between NF1 and MS might have a molecular explanation. The NF1 gene is located on chromosome 17q, contains 60 exons, and spans 350 kb of genomic DNA. This gene is highly expressed in the myelin-forming oligodendrocytes which are the primary targets of the inflammatory and immune attacks in MS. 34
Conclusion
The association of NF1, MS, and CA can be considered as an expression of multiple factors: molecular explanation and the hiring of pharmacological treatments for MS that can increase the risk of stroke.
The onset of new neurologic symptoms and signs in patients with both NF-1 and MS should not always be considered an expression of the natural history of these two progressive diseases. Other overlapping acute pathologies should be kept in mind so as not to miss or postpone the correct diagnosis, and the consequent management (interventional, pharmacological, and rehabilitative treatment) should be appropriately applied.
Footnotes
Acknowledgements
The authors would like to thank the patient for her participation.
Author contributions
Laura Perucca (conceived and designed the article, revised it critically for important intellectual content), Francesca Morello (collected the data, performed the analysis, and wrote the paper), Antonio Robecchi Majnardi (conceived and designed the analysis, wrote the paper, and revised it).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Italian Ministry of Health - Ricerca Corrente.
Ethics approval
Ethical approval for this study was obtained from Instituto Auxologico Italiano, IRCCS, within the RESET research project 3/12/18 24C822_2018 (Ricerca Corrente 2020, Italian Ministry of Health).
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article; the patient could provide the written informed consent themselves.
