Abstract
Infective endocarditis is an infection of the heart with systemic consequences, both infectious and non-infectious. Infective endocarditis can affect several systems, one of which is the central nervous system. The most common form of presentation is ischemic stroke; however, intracranial hemorrhage can occur due to immune-mediated damage to the vessel wall. The former further complicates cardiac surgical procedures when necessary. We present here the case of a 21-year-old male patient, with no personal medical history, who presented with intracranial hemorrhage due to a vasculitis phenomenon, caused by Streptococcus gordonii infective endocarditis. The patient underwent emergency drainage of the intracranial hemorrhage and minimally invasive valve surgery in 17 days, with satisfactory postoperative recovery and follow-up.
Introduction
Infectious endocarditis (IE) is characterized by the following sequence: endothelial injury, sterile vegetation in cardiac valves, vegetation colonization by a microorganism, and finally, cardiac tissue destruction. 1
Central nervous system (CNS) is among the potential systems which may be affected by IE. CNS signs or symptoms, present in up to 55% of patients, 2 include: encephalopathy, seizures, stroke (both ischemic and hemorrhagic), cerebral abscess, meningitis, and infectious aneurysms (also known as mycotic aneurysms). 3 These infectious aneurysms, or blood vessel wall damage through an immune-mediated phenomena, could precede an hemorrhagic stroke, 4 being both events of interest for the present work.
The etiology of mycotic aneurysms is comprised mainly of Staphylococcus aureus, and in second place, Streptococcus spp. 5 Streptococcus gordonii is within streptococcus viridans group, which is a commensal bacteria that dwells in the oral cavity, the skin, gastrointestinal tract, and the upper respiratory tract. 6 Furthermore, through the expression of pro-inflammatory molecules, it could lead to an infectious vasculitic phenomena, which could conclude with the development of an aneurysm. 6
We present the case of a patient who presented to the emergency department with a cerebral hemorrhage. Arteriovenous malformations were ruled out in the diagnostic workup, and a possible vasculitic etiology versus infectious involvement of the blood vessel wall (mycotic aneurysm) was suspected. This led to the diagnosis of a native mitral valve IE due to S. gordonii infection, which required early minimally invasive valvular replacement surgery and management of other systemic complications of IE.
Case presentation
A 21-year-old male patient with no personal medical history attended the local rural hospital with thunderclap headache, projectile vomiting, and progressive weakness of the left lower limb of 2 h of evolution. The patient also presented neurological deterioration and decorticate posture; hence, orotracheal intubation and transfer by non-medicalized intercentre ambulance were performed. On admission, the patient was under sedation and mechanical ventilation, hemodynamically stable, with anisocoria due to mydriasis of the right eye. An emergency cranial computed tomography (CT) scan was performed and showed an intraparenchymal hemorrhage measuring 60 mm × 62 mm × 57 mm. The hemorrhage extended to the right parietal and occipital lobe, with surrounding edema, 7 mm midline shift, and intraventricular hemorrhage (Figure 1).

(a) Head CT with intraparenchymal and intraventricular hematoma of the right parietal and occipital lobes, associated with edema, and mass effect. (b) Brain MRI: Post-contrast T1: Normal vascular structures were observed within the circle of Willis. No significant stenosis, arteriovenous malformations, nor aneurysms larger than 3 mm were observed. Uptake in the blood vessel walls of the circle of Willis was observed (Yellow arrows and dotted lines). (c) Angiography: No aneurysms, arteriovenous malformations, nor dural fistulas were observed. No significant stenosis was observed. A slow capillary phase, with delay in the venous phase in both cerebral hemispheres was observed.
Admission laboratory results included a hemoglobin of 9 g per deciliter (g/dL), hematocrit of 29%, white blood cells of 24.200 cells per cubic millimeter (mm3), platelets of 412.000 per mm3, C-reactive protein of 5 mg/dL; coagulation tests included a prothrombin time of 14.5 s, international normalized ratio of 1.3, and partial thromboplastin time of 30 s. Human immunodeficiency virus (HIV) antibodies were non-reactive, calcium was 7.8 mg/dL, chloride 97 mg/dL, sodium 127 mg/dL, potassium 3.98 mg/dL, and creatinine 0.76 mg/dL. Arterial blood gasses had a pH of 7.34, bicarbonate of 20 mmol/L, base excess of −5.3, and lactate of 3.0.
The patient underwent emergency surgery due to his critical condition. Craniotomy and drainage of the hematoma was performed, with lobectomy of the necrotic brain tissue. The procedure was completed without complications, and the patient was transferred to the intensive care unit (ICU).
After admission to the ICU, the intensivist identified a holosystolic murmur at the base of the heart. In addition, the patient’s family reported a 6-month history of depressive symptoms and multiple episodes of syncope preceded by chest pain. Therefore, transthoracic echocardiography was performed, showing 23 mm mitral valve vegetation in the anterior leaflet, associated with perforation and severe insufficiency (Figure 2). Given the severity of the valvular damage, acute infectious damage was suspected. Aerobic and anaerobic blood cultures were performed, and empirical antibiotic coverage was initiated.
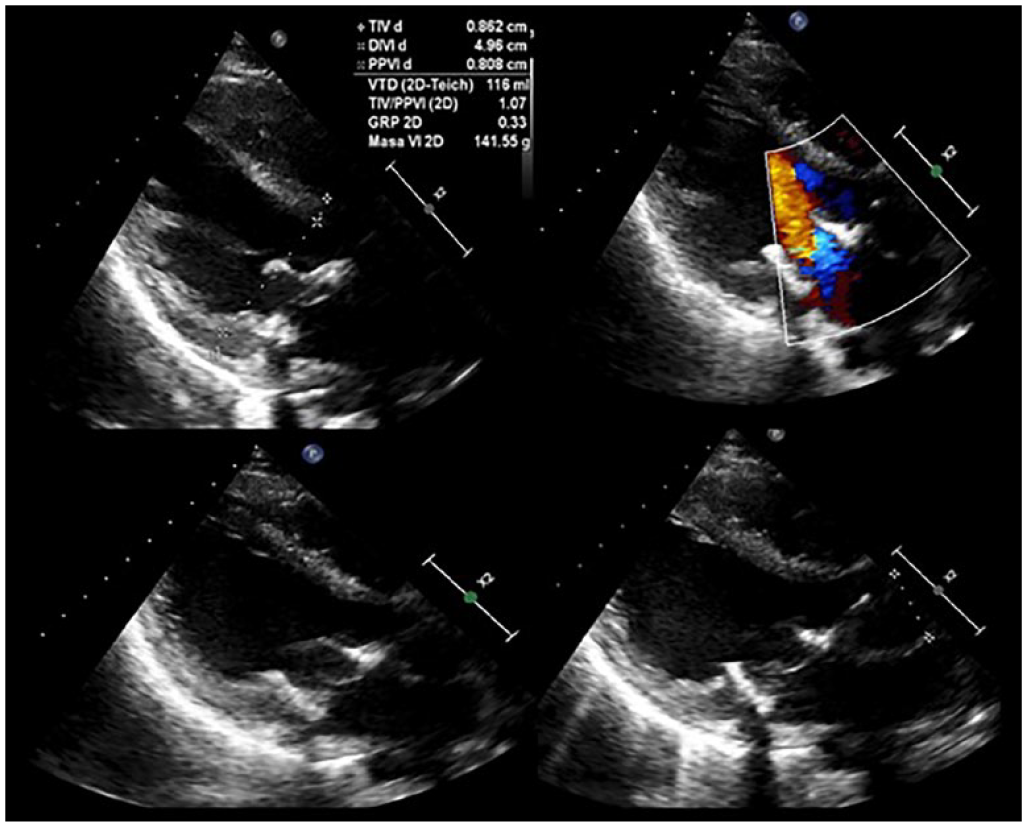
Mitral valve with a mobile, ecodense image attached to the ventricular surface of the anterior veil, compatible with vegetation, veil perforation, and severe insufficiency.
Cardiovascular surgery was consulted. Surgical treatment was clearly indicated due to the demonstrated embolism and echocardiographic findings. The staff meeting between neurosurgery and cardiovascular surgery decided that, given the recent massive cerebral hemorrhage and possible complications of systemic anticoagulant therapy, cardiac surgery should be postponed for 2 weeks.
Antibiotic treatment was adjusted to ampicillin given an isolation of S. gordonii in blood cultures. Moreover, left iliac pyomyositis and sacroiliitis were observed. Surgical lavage was performed by orthopedics, and cultures were negative.
Cranial magnetic resonance imaging (MRI) showed enhancement of vascular structures in the circle of Willis, suggestive of infectious/inflammatory vasculitis (Figure 1).
Pan-angiography showed no aneurysms, AVMs, or dural fistula (Figure 1). Therefore, according to a new joint meeting of cardiovascular surgery, cardiology, infectious disease, internal medicine, and neurosurgery staff, intracerebral hemorrhage (ICH) was considered a byproduct of the vasculitic process secondary to IE (cerebral vasculitis versus mycotic aneurysm).
Other imaging studies, including CT of the chest and abdomen revealed pneumomediastinum related to interstitial emphysema in the left lung, and ground-glass opacities in the upper and lower lobes suggestive of an aspiration process; splenic septic infarcts and air bubbles in the left iliac muscle were observed.
Based on MRI findings, no further neurosurgical procedures were deemed necessary, so minimally invasive valve replacement surgery with a biological valve was performed 2 weeks after the initial brain surgery, without complications.
The patient completed targeted antibiotic therapy and anticoagulation with low-molecular-weight heparin, with progressive neurological and clinical improvement. Therefore, the patient was safely discharged.
The patient was followed up by telephone. Ten months after the initial event, the patient resumed her daily life and work, with occasional headaches with adequate response to over-the-counter analgesics.
Discussion
Causes of ICH in young patients include AVM, arterial hypertension, cavernomas, and less than 2% are associated with systemic involvement of IE. 7 However, up to 40% of patients with IE present with neurologic involvement or sequelae. IE-related ICH has been reported in up to 30% of patients with ischemic stroke due to hemorrhagic transformation. Mycotic aneurysms or vasculitic phenomena have been described in 2%–5% of patients with IE-related ICH. 8 The most important mortality-related factor is the rupture of said aneurysms, rising from 30% to 80% in those with rupture compared with those without. 9 Some studies have described a higher frequency of aneurysmal rupture and ICH in patients with viridans streptococci infections. 4
We present the case of a young immunocompetent male patient, who was transfered to the emergency department for an intraparenchymal hematoma, in the context of a native mitral valve IE due to S. gordonii infection (Figure 1). This germ belongs to the Sanguinis group of the viridans streptococci. 6 To our knowledge, this is the second report available with similar characteristics. 8
Regarding native valve IE, it is known as an infrequent entity, with the male sex with higher likelihood of developing the disease. 10 Up to 80% of cases are caused by Gram-positive bacteria such as S. aureus, viridans streptococci, and enterococci. 1 Even though most symptoms the patient had are usually described in IE literature, 10 it is remarkable that no recent literature mentions neuropsychiatric symptoms, 3 which were described in the subacute onset of the disease in our patient. Other studies have described neuropsychiatric symptoms with a 2% frequency. 11
The main pathogenic mechanism involved in IE-related complications is vegetation development. The embolism risk is higher in the following situations: left-sided vegetation and anterior leaflet of the mitral valve involvement, vegetations larger than 10 mm, delayed antibiotic therapy onset, and positive blood cultures for S. aureus and S. bovis.2,3
The most common embolic complication of IE is stroke, which occurs in up to 20%–30% of cases. 10 These strokes usually have higher risk of hemorrhagic transformation, with the medial and posterior cerebral artery being the most common vascular territories. Prompt antibiotic therapy and cardiac surgery are keystones of IE treatment and prevention of complications. 2
Over 50% of patients with IE will require valve surgery. Most guidelines agree on surgery indications. However, surgery timing in the context of ICH is a matter of controversy, as current guidelines advise for cardiac surgery to be performed 30 days after brain surgery, based on observational studies with limited patient data. 12 Up to 40% of IE patients with surgical indications do not undergo surgery in the proper time, which leads to higher 30-day mortality. In 15% of patients, surgery was not performed due to the presence of an ICH. 1 Our patient underwent minimally invasive valvular surgery 17 days after brain surgery, with proper post-operative evolution. This was performed in concordance with different studies, which report the safety of the procedure when performed within the first months, and even within the first 2 weeks after ICH drainage.13,14
Conclusion
The present case involved a young, immunocompetent patient who was admitted to the emergency department for an intracerebral hematoma secondary to suspected vasculitis due to severe multisystem involvement secondary to IE due to S. gordonii, a germ species rarely reported in the literature. A disadvantage of the present case is that a histopathological diagnosis was not possible, given the severity of the patient on admission and therefore the initial objective of the health personnel was to save the patient’s life. Therefore, the diagnosis of vasculitis was retrospective and within the context of IE given the MRI findings. A fact to highlight is the time in which it was decided to perform the valve replacement after the bleeding, given that most guidelines recommend performing this procedure after 1 month; however, in the present case, it was performed within 2 weeks later, showing an adequate recovery even 10 months after the event.
Footnotes
Acknowledgements
The authors acknowledge their working relationship with the Emergency Department of the Pablo Tobón Uribe Hospital.
Author contributions
CAAV, JEQA, and TUC contributed to the conception and design of the study. CAAV, JEQA, and TUC wrote the manuscript in Spanish, and TUC translated it into English. CAAV performed the literature search, and JEQA performed the medical history and imaging search. CAAV contributed in the clinical assistance to the patient, and JEQA performed the follow-up. All authors reviewed the manuscript. CAAV made revisions and edited reviewers’ requests. All authors read and approved the final manuscript.
Data availability
Clinical data.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from the Ethical and Research committee of the Hospital Pablo Tobón Uribe, as stated in the Acta 23-2022.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) to publish the patient’s anonymized information in this article because of the patient’s baseline condition at the time of the request it was not possible to obtain the patient’s own consent.
