Abstract
Haemophilus parainfluenzae, a HACEK organism, is one of five species known to less commonly cause infective endocarditis. This article presents a rare and noteworthy case of H. parainfluenzae endocarditis affecting the aortic valve, seemingly unique when Haemophilus species in endocarditis were previously known to more commonly involve the pulmonic or mitral valves. In the setting of H. parainfluenzae bacteremia, a 61-year-old male with no significant past medical history exhibited subtle systemic symptoms with a paucity of clinical signs associated with endocarditis and no evidence of embolic disease. A thorough workup demonstrated a large vegetation involving the aortic valve causing acute aortic insufficiency necessitating subsequent replacement of the aortic valve. In follow-up appointments with providers, the patient demonstrated excellent recovery returning to his baseline activity within 6 months. This case highlights the importance of considering endocarditis in Haemophilus bacteremia, especially in those with an unclear source of infection. Recognition of these rare presentations can assist with timely diagnosis and appropriate management, thus improving patient outcomes.
Keywords
Introduction
Infective endocarditis (IE) remains a significant cause of morbidity globally. Although incidence has decreased significantly in the past few decades, this diagnosis carries a high mortality rate. Commonly known risk factors for developing endocarditis include an immunocompromised state, intravenous (IV) drug use, underlying valvular disorders, implanted cardiac devices, or prosthetic valves. Skin flora, such as Staphylococcus, Enterococcus, and Streptococcus remain the most common causative organisms in IE, accounting for 80%–90% of cases. More rare causes of IE include the HACEK organisms: Haemophilus species, Aggregatibacter species, Cardiobacterium hominis, Eikenella corrodens, and Kingella species. 1
Haemophilus, an oropharyngeal flora, is an exceedingly rare cause of IE. HACEK organisms, including H. parainfluenzae, are known for their slow growth in routine blood cultures. However, mortality rates remain lower than common causes of IE such as Staph aureus. 2 Previously reported cases of H. parainfluenzae endocarditis were found to affect the mitral valve and almost all cases had prior valvular disease such as mitral valve prolapse.3–5 About 30% of all cases are noted to have either aortic stenosis or mitral valve prolapse. In one study, the affected valves in 51 patients were noted. Of these, 29 were noted to have mitral valve involvement, versus only 8 affecting the aortic valve. 5 Hence, mitral valve involvement is more commonly seen rather than other valvular disorders when managing IE. Patients reported to have mitral valve endocarditis due to H. parainfluenzae without any prior risk factors, systemic embolic events were present that led to the eventual diagnosis. For example, one case report discusses a 54-year-old female with no known past medical history, no recent procedures, and no risk factors for endocarditis who presented with fevers and headache; magnetic resonance imaging of the brain demonstrated multiple cerebellar, white matter, and sub-grey matter, low-signal T1-weighted and high-signal T2-weighted lesions consistent with embolic stroke. Transesophageal echocardiography (TEE) demonstrated two 20 mm mitral valve vegetations and the patient underwent mitral valve replacement with blood cultures later returning positive for H. parainfluenzae a few days after. 5 Another case report describes a previously healthy 26-year-old male with no risk factors for endocarditis with a chief complaint of abdominal pain. He was found to have multiple splenic infarcts concerning embolic disease, however, initial transthoracic echocardiography (TTE) demonstrated no valvular lesions. Given the concern for underlying cardiac pathology additional cardiac imaging was performed which demonstrated a right-to-left shunt and 1.5 cm left ventricular mass under the posterior leaflet of the mitral valve and large atrial septal defect. 6 Thus, as demonstrated in both cases, embolic manifestations and mitral valve involvement represent more common presentations of H. parainfluenzae infections.
This article discusses a unique occurrence of H. parainfluenzae IE in a patient with no known risk factors and the absence of embolic disease prior to diagnosis. Additionally, what makes this case increasingly unique is the demonstration of vegetation on a native aortic valve, a rarity given H. parainfluenzae’s predilection for the mitral valve. Hence, this demonstrates the elusive nature of the species and its ability to spread without alarm, potentially resulting in dire consequences from delayed diagnosis.
Case presentation
Case history and examination
A 61-year-old man with no significant past medical history presented to the emergency department with complaints of headache for 2 days with associated dizziness and chest discomfort. He also noted dysuria, abdominal cramping, and constipation for 5 days. Of note, the patient had just returned from a vacation in Hawaii 5 days prior. Vital signs were notable only for a blood pressure of 98/55 mm Hg with associated tachycardia, concerning for sepsis. Physical exam demonstrated mild abdominal tenderness to palpation with cardiovascular exam revealing a regular rate and rhythm without murmurs, rubs, or gallops. His lungs were clear to auscultation in all fields.
Methods
Labs demonstrated a leukocytosis of 13.5 cells/L, creatinine 4.69 mg/dL, BUN 54 mg/dL. There were also noted elevations in total bilirubin to 4.3 μmol/L and alkaline phosphatase level of 187 U/L. Cardiac troponin was minimally elevated and adynamic. D-dimer was 3.64 mg/L, lactate o2.6 mmol/L, and NT pro-BNP was 8171 pg/m. Urinalysis demonstrated leukocyte esterase, protein, and glucose. Upon this initial presentation to the hospital in the setting of sepsis, the patient was started on ceftriaxone for coverage of common infectious organisms.
Computed tomography (CT) abdomen and pelvis revealed a fluid-filled gallbladder and focal outpouching at the posterior lateral aspect of the left bladder. Further imaging was obtained with an abdominal ultrasound which demonstrated a thickened gallbladder wall with normal common bile duct diameter. Ventilation perfusion scan suggested a low likelihood of acute pulmonary embolism. Due to worsening acute kidney injury on the subsequent day with unclear etiology, nephrology was consulted, and recommended a full serological workup including ANA, ANCA, and complement studies. However, during this time, the patient did begin to demonstrate renal recovery with excellent urine output.
On day three of admission, blood cultures collected on day one of hospital visit returned positive for Haemophilus parainfluenzae. Two bottles were positive noting Haemophilus parainfluenzae in the aerobic bottle from the right and Haemophilus parainfluenzae in the bottle drawn from the left. Unfortunately, the patient continued to meet sepsis criteria, with the source yet to be identified. Results were discussed with infectious disease specialists who recommended repeat abdominal workup given signs of fluid-filled gallbladder on previous imaging as well a TTE. Repeat CT abdomen and pelvis remained unrevealing for source. Antibiotics were broadened to piperacillin/tazobactam. Moderate aortic valve stenosis was noted on TTE. Given the valvular abnormality and bacteremia, the decision was made to proceed with TEE. A large vegetation on the right coronary cusp of the aortic valve with associated moderate aortic regurgitation was observed (Figures 1 and 2). The presence of an eccentric jet suggested significant damage to the right coronary and non-coronary cusps as well (Figure 3). Per the modified duke criteria, two major criteria (blood cultures and echo finding of vegetation) were met. The patient also met three minor criteria with fever, positive blood cultures, and positive echo results meeting criteria for IE.

Demonstration of the vegetation on the right coronary cusp of the aortic valve during systole.
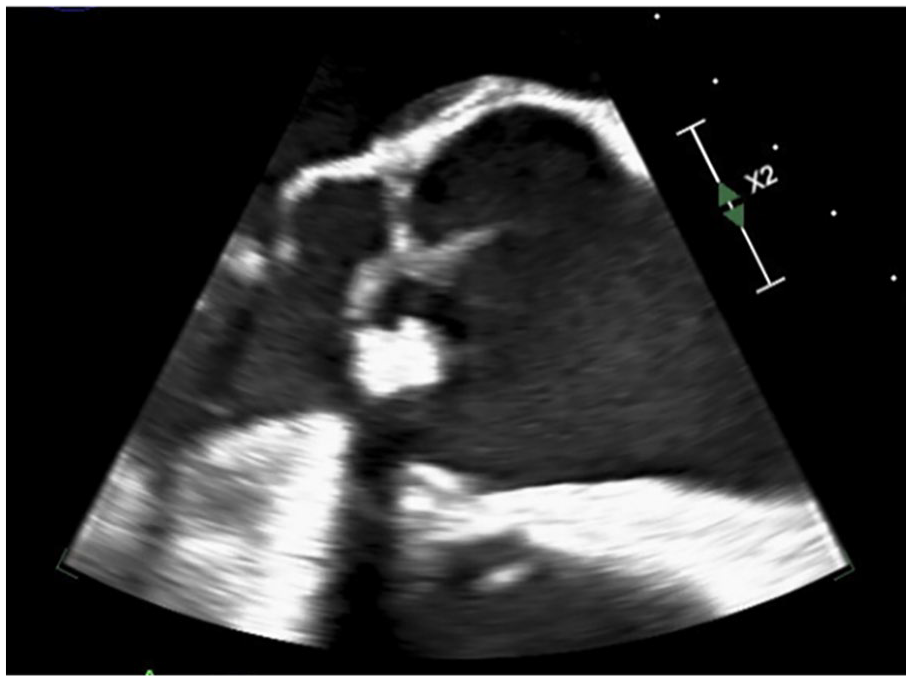
Redemonstration of the aortic valve vegetation at the end of systole.
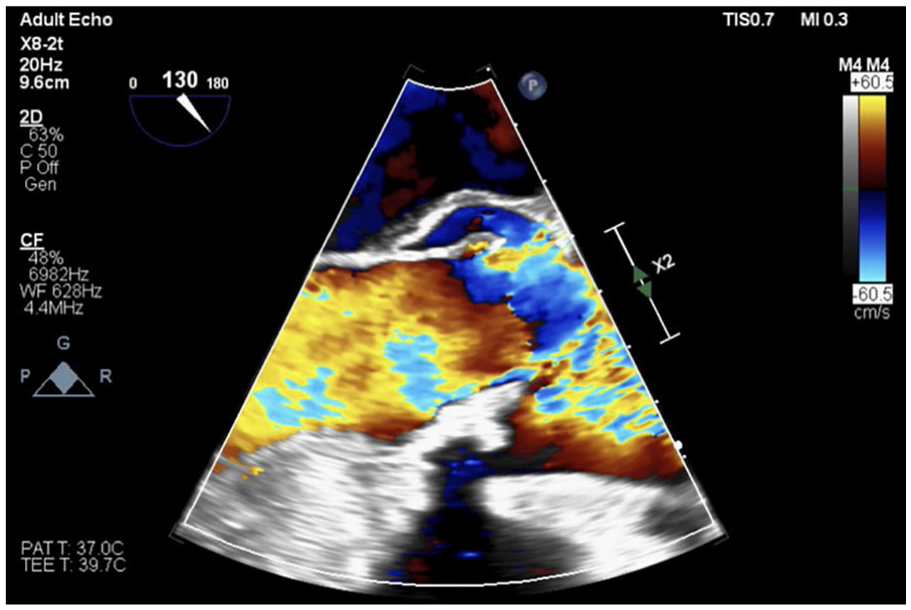
Regurgitant flow through aortic valve consistent with aortic valve insufficiency.
Conclusion and results
The patient was urgently transferred to a partner hospital for surgical aortic valve replacement. While awaiting transfer, he was also taken for cardiac catheterization which was unremarkable for occlusive coronary artery disease.
Cardiothoracic surgery recommended surgical aortic valve replacement. An infected aortic valve was excised from the annulus and debridement of devitalized tissue was performed. A replacement 29 mm bioprosthetic pericardial tissue valve was sewn into the super-annular position and left atrial appendage clipping was performed with a 35 mm Atri Clip. Repeat blood cultures and valve cultures demonstrated no growth. Pathology results of removed valve demonstrated acute inflammation and fibrinous exudates with calcification and degenerative changes.
The patient’s antibiotics were deescalated to ceftriaxone 2 g daily for 4 weeks. He continued to have a strong recovery with discharge home only 5 days after valve replacement. On the 4th of January, 5 days after discharge, the patient was seen in his primary care’s office and noted to have improvement in his pain and overall appetite. The swelling previously noted in his lower extremities was now resolving. On January 11th, the patient was seen in Cardiology clinic, 12 days post discharge, and noted to be doing well, walking 5–10 minutes at a time. No acute complaints including dyspnea, chest pain, or palpitations were reported. His last visit with Cardiology was 29th August 2024, and he was noted to have continued recovery with no complaints of lightheadedness, syncope or lower extremity edema.
Discussion
This case demonstrates a unique occurrence of IE in the setting of H. parainfluenzae infection. Although the development of endocarditis post Haemophilus infections has been seen previously, it has been more commonly found in mitral valves, and less commonly in pulmonic or tricuspid valves. However, the occurrence of aortic valve involvement has been rare, although has been recently increasing. 2
A systematic review done in 2022 investigated the prevalence of endocarditis in H. parainfluenzae. The majority of reviewed patients had at least one predisposing risk factor for endocarditis. These included prior history of endocarditis, a prosthetic or mechanical valve, dental caries, recent dental procedure, IV drug use, or steroid use. Although these are important for clinicians to identify to allow for early diagnosis, not all patients, such as the case discussed in this paper, may fit these criteria. 2
The above patient did not present with typical signs of endocarditis, such as Roth spots or Osler nodes, delaying the completion of a transesophageal echo to guide the diagnosis of endocarditis. Although this patient was on broad-spectrum antibiotics allowing for treatment of the infection, early source control with valve replacement is key to successfully managing the disease. Hence, early diagnosis of endocarditis in patients with Haemophilus bacteremia is vital. Despite delay in identification of the source, fortunately, this patient did not have dire effects of endocarditis such as septic emboli leading to cerebrovascular incidents and possibly life-changing events. 7 In fact, the rate of stroke in patients with HACEK endocarditis can be up to 25%, further demonstrating the importance of timely management. 1
H. parainfluenzae is responsible for only 0.5% of endocarditis cases. One unique characteristic of Haemophilus species endocarditis is a large vegetation with a high risk of embolization. For example, 35.7% of vegetations caused by Haemophilus species endocarditis embolize however 60% of cases where embolization has occurred are due to H. parainfluenzae species. 8 With these findings, IE should be considered in patients with H. parainfluenzae bacteremia of unknown source regardless of predisposing factors. As stated by the American Heart Association guidelines, “bacteremia caused by HACEK microorganisms in the absence of an obvious focus of infection is highly suggestive of IE even without typical physical findings of IE.” 9
Conclusion
This article demonstrates the rare yet consequential occurrence of aortic valve endocarditis in H. parainfluenzae bacteremia. The significance of this association lies in the elusive nature of the bacteria and its variable presentation. Despite previously reported cases of H. parainfluenzae endocarditis, there have been few documented cases of aortic valve involvement. The significance of early evaluation in the setting of bacteremia with transesophageal echocardiogram can be vital to prevent morbidity as well as mortality as evident in this patient who was very fortunate, that despite damaged leaflets and large vegetation, he did not suffer from an embolic stroke. The slow growth of this HACEK organism can further delay diagnosis, increasing the risk of harmful lasting effects including embolic strokes. Although antibiotic therapy may be effective and prevent further harm, early surgical intervention is critical to obtaining source control and appropriately managing endocarditis.
Footnotes
Acknowledgements
The authors of this publication would like to express their sincere gratitude to our mentors who have enabled us to succeed in this endeavor.
Author contribution statement
Nasrin Dhapa: writing original draft; Hannah Rodriguez: writing review and editing; Zachary Visinoni: writing review and editing; Neeladri Misra: supervision and writing – review and editing; Diwakar Lingam: supervision and writing – review and editing.
Data availability
Data available on request due to privacy/ethical reasons.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Consent to participate
The authors certify the patient signed an informed consent form, which is available upon request.
Consent for publication
The authors certify the patient signed an informed consent form indicating potential for publication of clinical case.
