Abstract
Diabetic striatopathy is a rare neurological complication of diabetes mellitus that presents with sudden onset hemichorea or hemiballismus and is associated with hyperglycemia and striatal abnormality, either by hyperdensity on non-contrast computer tomography or hyperintensity on T1-weighted magnetic resonance imaging. Here we report a 55-year-old female, from Sri Lanka, who presented with involuntary movements of the left upper and lower limbs. Her past medical history included diabetes mellitus and she was on warfarin 5 mg daily for a mechanical mitral and tricuspid valve replacement. The random blood sugar on admission was 462 mg/dL and the last INR was 3.03. While hemiballismus has multiple etiologies, intracranial hemorrhage would be the main differential in a patient on anticoagulation. Other differentials include drug-induced dyskinesia, metabolic abnormalities, and autoimmune etiologies. Hemiballismus in the presence of high blood glucose should always raise the suspicion of diabetic striatopathy. The non-contrast computed tomography of the brain showed hyperdensity in the right-side caudate nucleus, lentiform nucleus, and globus pallidus which is a characteristic of diabetic striatopathy but could have been mistaken for an intracranial hemorrhage. The involuntary movements improved with glucose control and treatment with clonazepam and tetrabenazine. This case highlights the potential for misdiagnosis of diabetic striatopathy as an intracranial hemorrhage in a patient on warfarin, which can lead to delays in appropriate management and erroneous omission of warfarin. Early recognition and treatment of diabetic striatopathy can lead to significant improvement in the quality of life.
Introduction
Diabetic striatopathy (DS) is a rare neurological complication of diabetes mellitus (DM) which presents an intriguing clinical challenge. The condition, characterized by involuntary movements primarily affecting the limbs, has been seldom reported in medical literature. DS has a prevalence of 1 in 100,000, which has been considered an underestimation due to underdiagnosis. 1 A systemic review showed that most of the cases were reported in Asia. 2 However, another study showed that DS was underdiagnosed in the Western populations. 3 DS usually occurs in older age, is more predominant in females, and may resolve over days with treatment of the underlying hyperglycemia or persist for years. 1
Its recognition is essential as it represents a unique manifestation of diabetes-related neurological dysfunction. In this case report, we present the clinical details of a patient with DS, which is often overlooked or misdiagnosed.
Case report
A 55-year-old female, from Sri Lanka presented with involuntary movements of the left upper limb and lower limb for 2-week duration. There was no weakness or paresthesia in between the episodes.
Past medical history
Her past medical history consisted of DM for 15 years for which she was on Gliclazide 40 mg bd and metformin 1 g bd. She had a mechanical mitral valve and tricuspid valve repair in 2010 and has been on warfarin 5 mg daily since then. The last two-dimensional echocardiogram showed an ejection fraction of 35% with a dilated left ventricle.
Examination
On examination, Glascow Coma Scale (GCS) was 15/15. Patient was afebrile. Blood pressure was 100/70 mmHg, pulse rate was 88 bpm, and there were no murmurs. SPO2 was 98% on room air with clear lungs. The abdomen was soft and non-tender with normal genitalia. There were no murmurs or pericardial rub and no femoral or renal bruit.
Investigations
Non-contrast computer tomography (NCCT) brain showed a hyperdensity in the right-side caudate nucleus, lentiform nucleus, and globus pallidus (Figures 1 and 2). The random blood sugar on admission was 462 mg/dL and the last INR was 3.03. Her full blood count, liver, and renal function tests were normal.

Non-contrast computed tomography scan of the brain shows hyperdensity in the right-side caudate nucleus, lentiform nucleus, and globus pallidus.
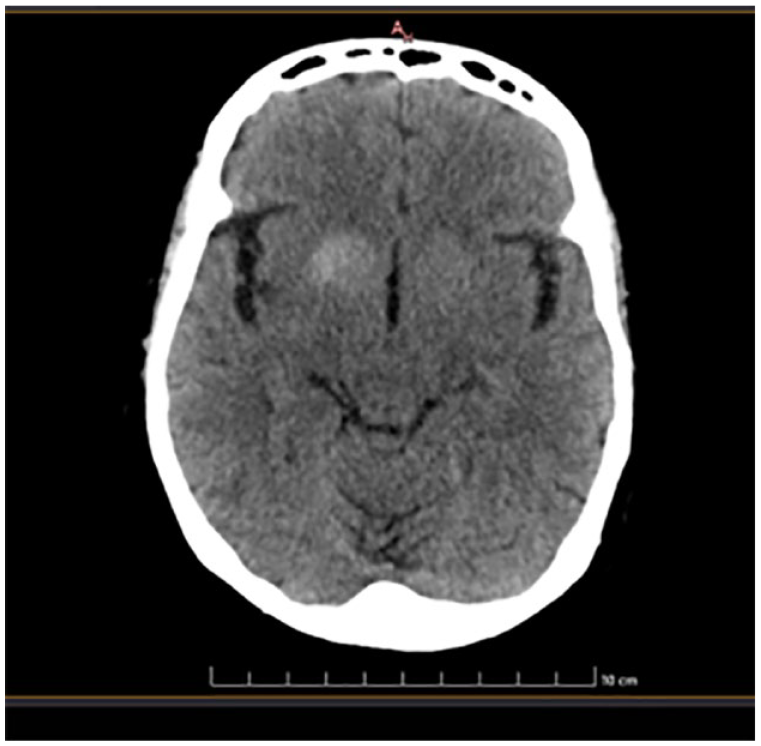
Non-contrast computed tomography scan of the brain shows hyperdensity in the right-side caudate nucleus, lentiform nucleus, and globus pallidus.
Discussion
The occurrence of chorea/ballism is mostly due to dysfunction of the basal ganglia and subthalamus. 4 Chorea consists of involuntary, rhythmic, purposeless, jerky movements involving distal limbs while proximal muscle involvement with larger amplitude is seen in ballism. 2
Etiologies of chorea or ballism include drug-induced dyskinesias, strokes or other vascular lesions in the basal ganglia, autoimmune conditions such as Sydenham’s chorea or anti-NMDA (N-methyl-D-aspartate) receptor encephalitis, metabolic disorders such as hyperthyroidism, Wilson’s disease, DS, hyperglycemia (without typical DS features); primary neurodegenerative disorders such as Huntington’s disease or neuroacanthocytosis syndromes and Parkinson’s disease.2,5 In this case, there is a high degree of suspicion for an intracranial hemorrhage (ICH) as the patient was on warfarin and therefore a hyperintensity on the NCCT brain could easily be misinterpreted as such.
DS is characterized by the sudden onset of hemichorea or hemiballism associated with hyperglycemic and striatal abnormality. 6 It is usually seen in the elderly and is more common in women. DS can present as the first manifestation of DM, though it is usually associated with long-standing, poorly controlled type 2 DM with an average hemoglobin A1c level of 13.1%. 2 DS is rarely seen in type 1 DM 7 and even after correction of hyperglycemia. 8
The pathophysiology is not entirely unraveled, but the main hypothesis is that in a non-ketotic hyperglycemic state, the brain metabolism shifts to the alternative anaerobic pathway in the Krebs cycle which leads to rapid depletion of gamma-aminobutyric acid (GABA). This results in disinhibition of the subthalamus and basal ganglia which causes hyperkinetic movements in DS patients. 2 However, in ketosis, GABA can be resynthesized using acetoacetate produced in the liver. This prevents the depletion of GABA and explains why DS is less common in diabetic ketoacidosis.5,9
The striatal abnormality includes hyperdensity on computer tomography or hyperintensity on T1-weighted magnetic resonance imaging. 6 This abnormality in the caudate nucleus and globus pallidus presumably results from petechial hemorrhage, mineral deposition (i.e., calcium or magnesium), myelin destruction, or infarction with astrocytosis.2,5
The mainstay of DS treatment is control of hyperglycemia and adequate hydration. However, only 25% of the patients will be successfully treated by glucose control alone while the majority may need pharmacological treatment which includes GABA receptor agonists, selective serotonin reuptake inhibitors, and dopamine-depleting agents. 9 Studies showed that the most common mono-therapeutic drug used was haloperidol. The other drugs used were tetrabenazine, risperidone, and clonazepam. 2 Out of all the combined regimens, the combination of haloperidol and diazepam was the most common, followed by haloperidol and clonazepam. 2
The doses of oral hypoglycemic drugs were optimized in our patient, and she was started on Clonazepam 0.25 mg bd and tetrabenazine 12.5 mg bd. The patient was reviewed weekly and involuntary movements gradually reduced in frequency and disappeared after 1 month of treatment.
This case report is unique as the patient was on anticoagulation, and any hyperdensity in the brain parenchyma in such a patient would be misinterpreted as ICH if the treating team is not aware of DS.
Conclusion
This case highlights the rare presentation of DS which should be considered in diabetic individuals presenting with movement disorders. It also shows the potential for misdiagnosis of DS as an ICH in patients on long-term anticoagulation, which can lead to delays in appropriate management and erroneous cessation of anticoagulation. Early recognition and treatment of DS can lead to significant improvement in the quality of life.
Footnotes
Acknowledgements
The authors were involved in the management of the patient.
Authors contribution
I.W. wrote and revised the manuscript. A.E., V.T.R., A.R., and S.M. reviewed the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
