Abstract
Severe hemodialysis access-induced distal ischemia is an uncommon complication after arteriovenous fistula creation. Finger amputation is rare and generally does not involve the entirety of the digit. The distal revascularization interval ligation procedure has become less commonly used for hemodialysis access-induced distal ischemia over the past decade. The procedure typically requires general anesthesia, greater saphenous vein harvest, and brachial artery ligation. We describe a 64-year-old female with hypertension, diabetes mellitus, and end-stage renal disease on hemodialysis via a well-functioning brachiocephalic arteriovenous fistula who developed rapid progression of finger gangrene. She underwent the distal revascularization interval ligation procedure, followed by finger amputations. The finger amputations healed within 6 months of the distal revascularization interval ligation procedure and the fistula was preserved at 2-year follow-up.
Introduction
The construction of a hemodialysis access in the upper extremity results in an alteration in perfusion with diversion of blood away from the high-resistance capillary bed of the hand into the low-resistance arteriovenous circuit. Up to 90% of hemodialysis accesses demonstrate flow reversal in the distal arteries (physiological steal phenomenon). However, only 1%–8% of these accesses will progress to pathological steal syndrome with significant malperfusion.1,2 Severe hemodialysis access-induced distal ischemia (HAIDI) can lead to irreversible hand ischemia and gangrene. Pathological factors include high flow rates through the access, arterial insufficiency from atherosclerotic disease, inadequate compensatory vasodilation, and/or insufficient collateral circulation. Finger amputation is a rare and devastating complication of hemodialysis access creation.
The distal revascularization interval ligation (DRIL) procedure is an effective approach to the management of HAIDI that offers both symptom relief and access salvage.3,4 The DRIL procedure involves creating a bypass from the artery proximal to the arteriovenous anastomosis to the distal artery and ligating the artery proximal to the new distal anastomosis. 5 The procedure is complex and requires general anesthesia which may be prohibitive in a frail patient. There is discomfort in ligating an axial artery as the blood supply to the hand becomes dependent on the bypass. The conduit of choice is the greater saphenous vein and wound complications can be high.6,7 The proximal anastomosis of the bypass graft should ideally be placed at least 10 cm proximal to the arteriovenous connection to avoid the anastomotic pressure sink, requiring a harvest incision spanning the length of the thigh.8,9 Furthermore, the arteries may be severely calcified and construction of the anastomoses may be difficult and risky. Because of these factors, the DRIL procedure has become less favored for the treatment of HAIDI. We describe a patient with a functioning brachiocephalic arteriovenous fistula who developed rapidly progressive finger gangrene. She was treated with the DRIL procedure and subsequent finger amputations. The finger amputations healed and the fistula was preserved.
Case description
A 64-year-old female with hypertension, diabetes mellitus, and end-stage renal disease on hemodialysis for 6 years via a left brachiocephalic arteriovenous fistula presented with worsening left third and fifth finger gangrene following minor nicks incurred during a manicure 4 months prior (Figure 1(a) and (b)). Past surgical history includes coronary bypass grafting 1 year prior and right below knee amputation 2 years ago. There was rapid progression of the third finger gangrene to the base of the digit and onto the palmar and dorsal aspects of the hand within 1 week (Figure 1(c) and (d)). There was atrophy of the intrinsic muscles of the hand with associated weakness. The brachial pulse was palpable, but wrist pulses were absent. A monophasic doppler signal in the radial artery was elicited with fistula compression. She had a well-developed left brachiocephalic arteriovenous fistula with a palpable thrill. No interventions had ever been performed on the fistula. The patient was afebrile with normal vital signs. Her white blood cell count was 21.3 bil/L. Her hemoglobin A1C was 10.6%. Blood cultures were negative. The patient was started on broad-spectrum intravenous antibiotics. Duplex ultrasound indicated a volume flow rate of 1952 ml/min. There was doubling of the radial artery waveform amplitude with compression of the fistula. Echocardiogram indicated a normal ejection fraction. Angiogram from the aorta to the palmar arch was obtained (Figure 1(e)–(h)). There were no arterial inflow lesions, a widely patent brachiocephalic anastomosis, and no cephalic vein nor central venous stenoses. There was no antegrade flow distal to the arteriovenous anastomosis which corrected with fistula compression. The radial artery was patent with terminal filling of the deep palmar arch. The ulnar artery was diminutive. A left DRIL procedure was performed (Figure 2(a)). General endotracheal anesthesia was used. The proximal brachial artery was exposed through a longitudinal incision below the axillary crease. The distal brachial and proximal radial arteries were exposed through a longitudinal incision below the antecubital crease. The greater saphenous vein was harvested from the right thigh. The vein was placed in a reversed configuration in an anteromedial subcutaneous tunnel along the upper arm. Systemic heparin was given. The proximal anastomosis was constructed end-to-side 18 cm proximal to the brachiocephalic anastomosis using 6-0 polypropylene suture. The distal anastomosis was created onto the distal brachial and proximal radial arteries in an end-to-side configuration. The distal brachial artery was ligated just distal to the arteriovenous anastomosis. Completion operative angiogram demonstrated patent proximal brachial to distal brachial artery vein bypass graft, patent brachiocephalic arteriovenous anastomosis, and brisk filling of the radial and ulnar arteries (Figure 2(b)). The patient underwent ray amputation of the third finger and amputation of the fifth finger distal phalanx 5 days following the DRIL procedure. The third finger amputation site was left open. Wound cultures were positive for Citrobacter freundii, Staphylococcus aureus, and Enterococcus faecalis and she completed a 2-week course of antibiotics. A split-thickness skin graft was used several weeks later to facilitate closure of the wound. At 2-year follow-up, the fistula was functioning well without the need for further interventions. The patient was free of hand pain with healed finger amputations and healed vein harvest incision (Figure 2(c)–(e)).
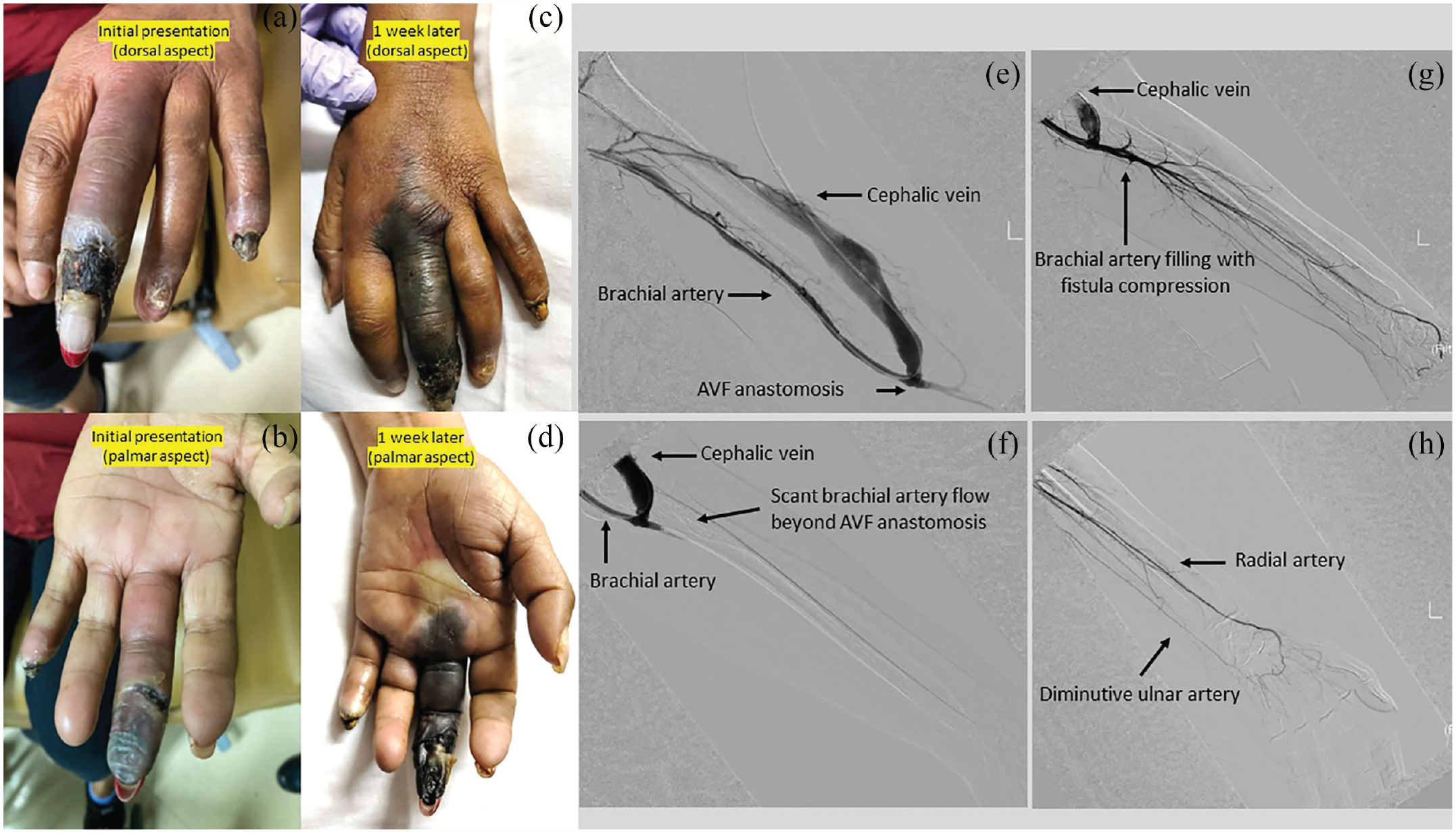
(a) and (b): Dorsal and palmar aspects of the left hand with gangrenous changes of the third and fifth fingers. (c) and (d) Progression of third finger gangrene 1 week later. (e) Left upper extremity angiogram demonstrating patent brachiocephalic anastomosis (f) and lack of opacification of the brachial artery beyond the arteriovenous anastomosis. (g) and (h) Filling of the brachial and radial arteries with manual compression of the fistula. Note that the ulnar artery is diminutive.

(a) Intraoperative photograph of the left upper extremity DRIL procedure. (b) Completion angiogram demonstrating patent brachiocephalic arteriovenous fistula and patent brachial artery to brachial artery vein bypass graft. (c) and (d) Dorsal and palmar aspects of the left hand 6 months following the DRIL procedure. (e) Right thigh greater saphenous vein harvest site 6 months following the DRIL procedure.
Discussion
Finger gangrene is a rare but bewildering complication of HAIDI. Risk factors for the development of HAIDI include diabetes, female gender, age >60 years, peripheral arterial disease, tobacco abuse, multiple previous ipsilateral access procedures, high-flow access, capacious outflow veins, use of the brachial artery for inflow, and prior history of HAIDI. 10 There is a wide spectrum of symptoms, including sensory loss, paresthesia, poikilothermia, pallor, diminished or absent distal pulses, muscle weakness, hand pain aggravated by hemodialysis, and tissue loss. The DRIL procedure has been considered the standard operation to manage HAIDI with high symptom resolution and access preservation rates. It adds a low-resistance collateral vessel to reduce the total peripheral resistance and simultaneously abolishes the suction retrograde flow, thereby augmenting flow to the distal extremity. However, it can be a technically challenging multistep procedure and other less aggressive treatment options including ligation, narrowing the efferent vein with banding or plication, percutaneous transluminal angioplasty (PTA) of proximal and distal arterial lesions, revision using distal inflow (RUDI), and proximalization of the arterial inflow (PAI) (Figure 3). Treatment options for HAIDI must consider if the flow in the access is high or normal/low. The optimal blood flow for hemodialysis access is 600–1500 ml/min. High-flow fistulas demonstrate flows greater than 2000 ml/min and may also present with excessive bleeding from puncture sites, aneurysmal dilatation, accelerated central venous stenosis, and high-output cardiac failure. 11 High-flow accesses with HAIDI require flow reduction. Normal-flow and low-flow accesses with HAIDI generally require revascularization procedures.

Treatment options for hemodialysis-access-induced distal ischemia.
Operations for high-flow fistulas include fistula ligation, venous side branch ligation, banding, plication, insertion of a tapered interposition graft, and RUDI. The most definitive treatment modality for HAIDI is access ligation. However, ligation eliminates a viable access and is used as a last resort. It is used in patients with exceptionally poor prognosis, fistula dysfunction, or extensive unsalvageable tissue loss. Endovascular ligation of fistulas with Amplatzer plugs and coils is particularly appealing in high-risk surgical patients.12–14 Our patient had a well-functioning brachiocephalic fistula with no prior interventions and fistula abandonment would be costly. Banding the access using nonabsorbable suture or a prosthetic cuff to constrict the juxta-anastomotic vein is imprecise and risks access thrombosis. Some have advocated precision banding or plication using intraoperative flow measurements to calibrate the degree of narrowing to the restoration of distal arterial perfusion. 15 The Miller (minimally invasive limited ligation endoluminal-assisted revision) procedure involves banding the juxta-anastomotic vein with monofilament suture over an inflated 3 to 5 mm balloon placed retrograde toward the arteriovenous anastomosis under fluoroscopic guidance. 16 RUDI extends the anastomosis further distally by dismantling the original brachiocephalic arteriovenous anastomosis above the elbow and interposing a vein or polytetrafluoroethylene (PTFE) graft from the outflow cephalic vein to a smaller-diameter lower-flow proximal forearm artery. 17 The selection of the inflow artery (radial or ulnar) is preferentially to the nondominant artery with the dominant artery reserved for hand perfusion. RUDI is not an option if there is significant forearm arterial occlusive disease (<2 mm, calcification) as in our patient who had a significantly diseased ulnar artery.
For normal-flow or low-flow fistulas, revascularization is generally necessary with PTA, PAI, or DRIL. For patients with severe disease in the radial or ulnar arteries, DRIL allows for optimal redistribution of blood flow. In our patient, there was an absence of proximal artery inflow disease. Isolated PTA of the diseased ulnar artery would have unlikely provided enough augmentation of blood flow to heal the digital gangrene. PAI converts the arterial supply of the brachiocephalic arteriovenous fistula to a more proximal level by first ligating the access close to the former arteriovenous anastomosis and implanting a small caliber 4–5 mm PTFE graft between the cephalic vein and proximal brachial or axillary artery. This new configuration adds length to the outflow segment (increased resistance) and the larger diameter of the artery increases perfusion to the hand. The more proximally located arterial anastomosis permits collateral recruitment at a higher point in the arm. 18 There is no need for axial artery ligation. However, the fistula does become partially prosthetic with its attendant risks of thrombosis and infection with compromise of fistula survival. PAI may be similar to performing the DRIL procedure without interval ligation as long as the insertion of the proximal anastomosis is at least 10 cm from the arteriovenous anastomosis.19,20 Zanow et al. 21 reported that interval ligation in DRIL increases the distal pressure by only 10%.
According to the United States Renal Data System, the expected remaining lifetime for a 60 to 70-year-old patient on hemodialysis is approximately 5 years. 22 Finger amputation portends an even poorer prognosis for survival, reflecting a greater atherosclerotic burden. Yeager et al. 23 reported a 27% 2-year survival rate once finger gangrene developed; finger gangrene was unrelated to a functional arteriovenous fistula in 52% of cases. Our patient had an uncomplicated postoperative course following the DRIL procedure with a well-functioning fistula at 2 years. However, Berman et al. 24 reported a 28.6% mortality rate from cardiovascular complications during a mean period of 7 months after DRIL. We posit that the increase in quality of life afforded by resolution of hand pain, healed finger amputations, and fistula survival justifies the use of the DRIL procedure despite mediocre life expectancy.
We prefer the DRIL procedure in younger patients with HAIDI who have reasonable life expectancies. Progression of finger gangrene can occur rapidly, particularly if there is superimposed infection and localized finger sepsis. Our patient had poorly controlled diabetes with an impaired immunologic response to combat infection. Expeditious finger amputation and redistribution of blood flow with the DRIL procedure can limit the extent of tissue loss and preserve fistula function even in the setting of forearm arterial occlusive disease.
Conclusion
Salvage of an arteriovenous fistula in a patient with finger gangrene and HAIDI can be formidable. The DRIL procedure effectively redistributes blood flow to optimize healing of gangrenous lesions in the hand while concurrently maintaining fistula function.
Footnotes
Acknowledgements
None.
Authors’ note
This case report was presented at the 7th Annual Meeting of the Society of Asian Academic Surgeons, Honolulu, Hawaii, September 16–19, 2022.
Author contributions
All authors contributed equally to the article and read and approved the final version of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
