Abstract
Congenital aortic arch anomalies are an uncommon diagnosis resulting from embryologic malformations during the fourth to eighth week of gestation. Asymptomatic variants frequently are overlooked in the perinatal period and diagnosed incidentally during adulthood. Symptomatic variants can present with steal syndrome or dysphagia lusoria. The right aortic arch is a rare anomaly that is usually associated with other congenital anomalies but can occur in isolation. The most common types of right aortic arches are mirror image branching or an aberrant left subclavian artery. Aortic arch anomalies are important to recognize as they may have important implications in management. We present a 74-year-old female with a right aortic arch and aberrant left subclavian artery that was discovered after a fall. An extensive evaluation and work-up revealed symptoms consistent with subclavian steal syndrome that resolved following a carotid-axillary bypass. Subclavian steal secondary to a right aortic arch is extremely rare. This report reviews the current literature of a right aortic arch with an aberrant left subclavian artery presenting as a subclavian steal syndrome.
Keywords
Introduction
The right aortic arch (RAA) is a rare variant of aortic arch anomalies that occurs in less than 0.1% of the population. 1 This results in an aorta that arcs over the right bronchus and passes to the right of the trachea and esophagus. Historically, RAA has been categorized into three distinct types: mirror-image branching, aberrant left subclavian artery (ALSA), and isolated left subclavian.2,3 The most common type is the mirror-image branching RAA. It is often diagnosed during the perinatal period due to a strong association with congenital cardiac defects, such as Tetralogy of Fallot or DiGeorge’s syndrome.3–5 Congenital anomalies are associated with only 5%–15% of ALSA, compared to 75%–85% with mirror-image branching and isolated left subclavian. 6 Most often ALSA is discovered incidentally on imaging during adulthood, but when symptomatic, can develop steal syndrome or dysphagia lusoria. 7 Duplex sonography, angiography, computed tomography (CT), or magnetic resonance imaging (MRI) are valuable for diagnostic evaluation and operative management.8,9 Management options include endovascular stenting or open surgical repair with transposition or bypass. 10 Our case presents a right aortic arch and aberrant left subclavian artery (RAA-ALSA) with an atretic origin and long-segment stenosis manifesting as subclavian steal syndrome. Currently, only 10 other cases of RAA-ALSA-associated subclavian steal syndrome are reported. 11
Case presentation
A 74-year-old female presented as a stroke alert due to multiple falls and confusion. For 6 years, she reported recurrent vertigo-induced ground-level falls and right-sided ataxia. She denied any presyncopal prodrome or other neurologic changes. Left upper extremity pressures were 27 mmHg less than the right. The arterial duplex demonstrated retrograde left vertebral artery flow and monophasic waveforms of the left subclavian artery (Table 1). Computed tomography angiogram (CTA) of the chest and neck showed a right-sided aortic arch with an aberrant left subclavian artery (Figure 1). A 3D reconstruction showed the origin of the left subclavian was hypoplastic with a 2.4-cm segment of stenosis extending to the left vertebral artery (Figure 2). CTA of the head and brain MRI showed no acute intracranial abnormality. The 3D reconstruction showed a dominant right vertebral artery (Figure 3). The posterior communicating arteries were not able to be identified. An electrocardiogram and echocardiogram were negative for any arrhythmias or cardiac defects. A 24-h continuous electroencephalogram was negative for seizure. A multidisciplinary team determined her symptoms were likely due to subclavian steal syndrome associated with RAA-ALSA. Due to atretic left subclavian origin, long segment stenosis, and retro-esophageal course, we performed a left carotid-axillary bypass using an externally supported 6 mm polytetrafluoroethylene graft (Figures 4 and 5). Postoperatively, her confusion resolved, and she had significant gait improvement with physical therapy. She was discharged on postoperative day (POD) 2 to a nursing facility. Her preoperative symptoms had near complete resolution during evaluation in the clinic on POD15.
Carotid duplex.

PSV = peak systolic velocity; EDV = end-diastolic velocity; CCA = Common carotid artery; ICA = internal carotid artery.
Duplex demonstrating less than 50% stenosis of the right internal carotid artery, no significant stenosis in the left internal carotid artery, and retrograde flow in the left vertebral artery with monophasic waveforms in the left subclavian artery.
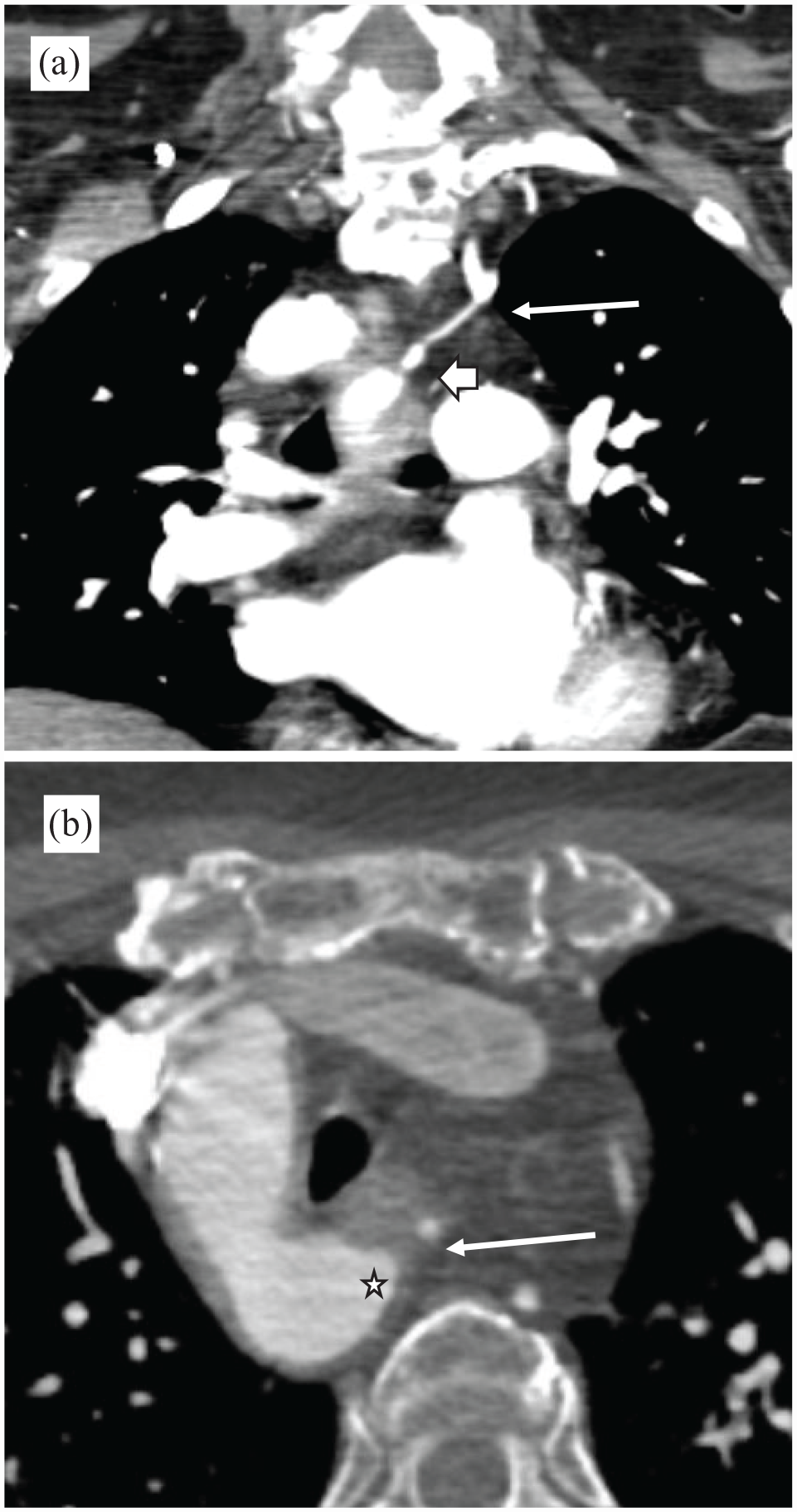
(a) Coronal view of computed contrasted angiography showing the atretic origin of the aberrant left subclavian artery (long arrow) with a distal bulbous segment and stenosis extending up to the origin of the left vertebral artery (short arrow). (b) Axial view showing the atretic segment of the aberrant left subclavian (arrow) and the Kommerell’s diverticulum (star).

(a) 3D reconstructed CT angiography of the aortic arch showing the origins of the great vessels. Beginning proximally, the left common carotid artery (LCC), right common carotid artery (RCC), right subclavian (RSC), aberrant left subclavian artery (LSC). (b) There is severe stenosis (arrow) of the entire proximal aberrant left subclavian artery (LSC) beginning at its origin from the aortic arch and extending to the origin of the left vertebral artery (V). Distal to the Kommerell’s diverticulum (star) is a completely atretic origin of the left subclavian artery.

3D reconstruction of the CT angiography of the head. The right vertebral artery (RV) is dominant compared to the left vertebral artery. There is no identifiable stenosis or lesion. Both anterior and inferior communicating arteries are normal. The posterior communicating arteries were not able to be identified.

Carotid artery (long arrow), sternocleidomastoid muscle retracted laterally (star), 6 mm PTFE graft (triangle), and internal jugular vein (small arrow) retracted medially.
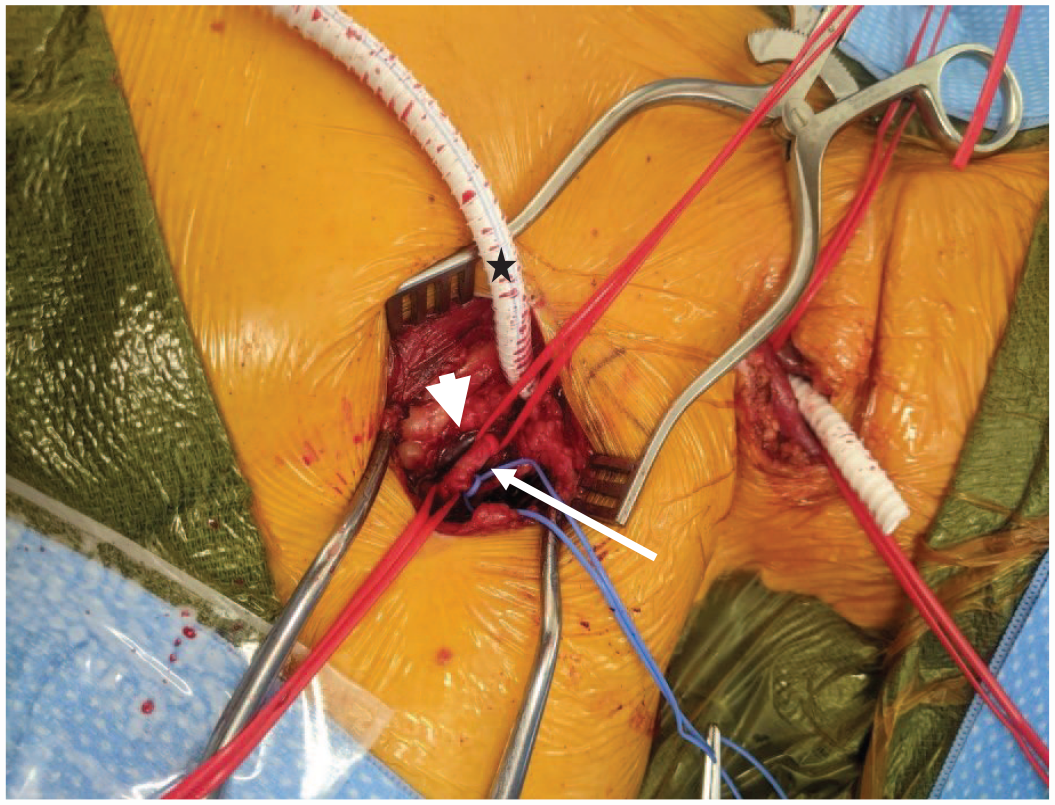
Axillary artery (long arrow), Axillary vein (short arrow), and PTFE graft (star).
Discussion
The aorta begins to develop during week four of gestation and gives rise to its mature pattern by week eight.7,10 A pharyngeal apparatus is formed by six paired pharyngeal arches and their associated arteries, called the aortic arches. The six aortic arches arise from the aortic sac and connect into two dorsal aortas. 12 As development occurs, the aortic arches undergo sequential maturation so that not all coexist at the same time. The first, second, and fifth aortic arches involute. The third arch contributes to form the internal carotid arteries. The right fourth arch becomes the proximal right subclavian artery and the left fourth arch becomes part of the adult aortic arch. The sixth arch forms the pulmonary arteries and ductus arteriosus. The two dorsal aortas fuse and give rise to intersegmental arteries which migrate to form other arterial derivatives. In particular, the seventh intersegmental arteries give rise to the subclavian arteries.10,12
Aortic arch anomalies occur due to the failure of these complex series of migrations, fusions, and involutions during fetal maturation. The congenital variants are thus categorized depending on the course and position of the arch and great vessels. In 1964, the Stewart et al. 13 classification organized aortic arch anomalies into four main groups: double aortic arch, left aortic arch, right aortic arch, and other anomalies of the aortic arch. RAA occurs due to the persistence of the right fourth pharyngeal arch vessel and right dorsal aorta with regression of the left arch. 7 This results in an aorta that arcs over the right bronchus and passes to the right of the trachea and esophagus.
RAA occurs in 0.1% of the population and is categorized into three types.1,2,14 Type I, mirror-image branching, is a direct reflection of a left aortic arch. The great vessels branch off the aorta in reverse order: left subclavian, left common carotid, right common carotid, right subclavian. Type II, aberrant left subclavian artery (ALSA), describes a left subclavian artery arising from a persistent outpouching of the aorta called Kommerell’s diverticulum. The branching order is left common carotid, right common carotid, right subclavian, then left subclavian. Type III, isolated left subclavian, involves the patent ductus arteriosus connecting the left subclavian to the pulmonary artery instead of the aorta. A more recent study has subclassified RAA into 10 distinct variants. 15 The most frequent variant is mirror-image branching (59.3%–84.5%), followed by ALSA (14.1%–39.9%), then isolated left subclavian (1%).3,14
Most diagnoses of RAA are made during the prenatal or neonatal period. 11 This is due to the strong association of RAA with congenital cardiac defects. Up to 24% of postnatal infants with RAA have a 22q11.2 microdeletion. 5 Mirror image RAA is frequently seen with Tetralogy of Fallot in 85%–90% of cases.3,6,15 Infants present with symptoms of cyanotic heart defects or respiratory distress and poor feeding/weight gain due to compression of the esophagus or trachea.5,6 Conversely, RAA-ALSA is associated with congenital cardiac anomalies in only 5%–15%, thus many with the ALSA variant will go undetected and are typically diagnosed incidentally on imaging during adulthood.3,4,6,7,15 The course of an ASLA can be divided into three pathways: retroesophageal, between the esophagus and trachea, or pre-tracheal. When symptomatic, the most common complaint is dysphagia. 6 Other symptoms include cough, dyspnea, chest discomfort, and in rare instances, syncope or subclavian steal syndrome. 8 Only 10 other cases of RAA-ALSA with subclavian steal have been reported in the current literature with five cases occurring in the pediatric population. 11 Six patients did not receive any stated treatment (Table 2). Takahashi et al. 16 had evidence of subclavian steal syndrome diagnosed by retrograde flow on angiography, however, no surgical treatment was pursued due to lack of symptoms. There were six patients 18 years or younger. Four had the presence of a congenital heart defect. One of which had complete repair of the Tetralogy of Fallot but did not undergo any correction for the aberrant left subclavian artery. Powell et al. 17 and Victorica et al. 18 reported the resolution of subclavian steal syndrome by ligation of the ligamentum arteriosum. There were four other cases that had presented during adulthood, three of which were symptomatic. Adult patients will more commonly present with neurologic symptoms secondary to subclavian steal.16,19–21 The most common treatment performed in the adult population was a bypass from the carotid artery to either the subclavian or axillary artery.
Summary of current literature on right aortic arch with aberrant left subclavian artery and evidence of subclavian steal syndrome.

GERD = gastroesophageal reflux disease; PA = pulmonary artery; VSD = ventricular septal defect.
Subclavian steal syndrome is the result of cerebral hypoperfusion due to the diversion of blood flow to the arm via the vertebral artery secondary to subclavian stenosis. 19 Symptoms resultant from vertebrobasilar insufficiency include dizziness, vertigo, confusion, headaches, ataxia, gait instability, and diplopia. The most common etiology is atherosclerosis of the proximal left subclavian artery. Takayasu arteritis, aortic coarctation repair, and thoracic outlet compression are other less common etiologies.6,11,19 Our patient had chronic vertebrobasilar insufficiency due to an atretic left subclavian artery leading to a prolonged history of multiple falls.
Management begins with careful evaluation of the thoracic vascular anatomy. Contrast-enhanced CT and MRI is the first and most significant imaging modality. 10 Barium esophagram is indicated in the presence of dysphagia to diagnose vascular rings leading to dysphagia lusoria. 12 Extracranial duplex sonogram will exhibit retrograde flow in subclavian steal. Echocardiography is necessary to rule out concomitant structural heart defects.8,9,12 Angiography can delineate anatomy, evaluate flow reversal, and allow therapeutic intervention. 6 Endovascular repair of subclavian steal-associated RAA-ALSA is a minimally invasive option for treatment if the non-atretic vessel can be traversed with a guidewire. However, most cases have severely stenotic or atretic origins, thus, are managed with an open carotid-subclavian transposition or bypass.10,19 Treatment is associated with low morbidity and excellent long-term results. 19
Conclusion
The diagnosis of the ALSA variant is frequently delayed until adulthood due to the absence of congenital defects and associated non-specific symptoms. Consequently, chronic vertebrobasilar insufficiency may lead to long-standing morbidity and poor quality of life. CTA with 3D reconstruction with evidence of retrograde flow from the vertebral artery provides the best method for the detection of the subclavian steal phenomenon. Once diagnosed, treatment achieves excellent results. Our patient had a complete recovery in gait stability, speech, and mental status after surgical bypass of her atretic ALSA.
Footnotes
Author contributions
Jonathan Nakata: Surgical resident involved in the patient’s case and manuscript.
Kurt Stahlfeld: Surgeon mentor involved with the manuscript.
Samuel Simone: Primary surgeon involved in the patient’s case.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
