Abstract
Mitral facies is a classical feature of chronic mitral stenosis (MS) that commonly associated with low cardiac output and pulmonary hypertension. A 44-year-old woman presented with 10-year history of refractory right heart failure. We noted distinctive malar rash appearance on her face known as “mitral facies.” An echocardiogram revealed severe MS and other significant valve involvement with typical characteristics of rheumatic valvular heart disease. Doppler measurement showed decreased cardiac output and severe pulmonary hypertension in this patient. The mitral facies could be an alarming sign of a more severe and advanced form of MS. It also can be a marker of impaired cardiac output and concomitant severe pulmonary hypertension.
Introduction
Mitral stenosis (MS) is characterized by the narrowing of mitral valve orifice due to structural abnormality of mitral valve apparatus. Rheumatic disease is the most common etiology of MS in the world. 1 The prevalence differs by geography, with fewer cases (0.2–0.3 per 1000) reported in industrialized than in the developing countries (5.5–5.7 per 1000). 2 The patient with MS may be asymptomatic for years and then present with a gradual decline in functional capacity. As the MS worsens and left atrial pressure elevates, the symptoms such as exertional dyspnea, orthopnea, and paroxysmal nocturnal dyspnea occur. 3 In more advanced stages, patient may complain symptoms of right heart failure such as abdominal discomfort, ascites, and leg swelling. 4 Patients who are symptomatic have a poorer prognosis with a 5-year survival rate of only 44% compared to asymptomatic or minimally symptomatic patients.4,5
Nowadays, echocardiography becomes the main modality used to confirm the diagnosis and to assess the severity and consequences of MS as well as the extent of anatomic lesions. 4 However, the physical examination of MS patient is usually characteristic and diagnostic such as the low-pitched rumbling diastolic murmur and opening snap. Several clinical signs are clues to more severe disease, one of which is the mitral facies, a condition related to impaired cardiac output and pulmonary hypertension. 3 Being able to recognize this sign not only can address appropriate treatment, but can also prevent more complications that increase mortality and morbidity.
Case description
A 44-year-old woman was referred from remote regional hospital to our cardiac center with 10-year history of progressive exertional dyspnea. In the last 6 months, she had refractory abdominal and leg swelling episodes in the previous hospital. She had already been known to have valvular heart disease for the last 5 years but had been managed with medical treatment only due to her low socioeconomic status. Her previous medications were furosemide, digoxin, spironolactone, ramipril, and warfarin, which had only been routinely consumed for the last 6 months.
The patient was fully alert with a blood pressure of 104/76 mmHg, a heart rate of 90–100 bpm, respiratory rates of 18 breaths per minute and peripheral oxygen saturation of 96%. Physical examination showed signs of right heart failure such as elevated jugular vein, hepatomegaly, ascites, and bilateral leg edema. Cardiac auscultation revealed irregular heart sounds and mid-diastolic and pansystolic murmurs at the region of the apex and left lower sternal border. Lung examination showed bibasilar crackles on both lungs. We noted reddish-purple rash and hyperpigmentation on bilateral cheeks, nose, nasal bridge, and perioral region (Figure 1). This distinctive malar rash is known as “mitral facies.” It had been noticed by the patient and her family since more than 2 years ago. There is no associated complaint of pruritus, pain or other symptoms related to this facial rash. She also denied any history of allergy or facial skin infection before.

Reddish-purple rash with hyperpigmentation on bilateral cheeks, nose, nasal bridge, and perioral regions.
Laboratory study showed an elevated NT pro-BNP level of 3724 pg/mL. She had mild microcytic anemia with a hemoglobin of 9.0 mg/dL. Renal function was mildly decreased with a creatinine level of 1.16 mg/dL and an estimated glomerular filtration rate of 53 mL/min/1.73 m2. Electrocardiogram (ECG) showed atrial fibrillation with rapid ventricular response, right axis deviation, incomplete right bundle branch block (RBBB), and right ventricular hypertrophy (RVH) (Figure 2). Chest X-ray showed marked cardiomegaly, widened right heart border, enlarged segment of main pulmonary artery and prominent cardiac waist (Figure 3). There was also mild bilateral pulmonary congestion.

ECG showed atrial fibrillation with incomplete RBBB and RVH.

Massive cardiomegaly with prominent pulmonary artery segment and cardiac waist.
Transthoracic echocardiogram demonstrated severe MS with concomitant severe mitral regurgitation (MR), moderate aortic regurgitation (AR), severe tricuspid regurgitation (TR) with significant tricuspid stenosis, and mild pulmonic regurgitation (Figure 4). There were typical characteristics of rheumatic valvular disease such as chordal and leaflet thickening, hockey stick deformities of mitral valves (MV), restricted leaflet motions and coaptation defect (Figure 4). Doppler measurement showed a mean MV gradient of 11–12 mmHg and a MV area of 0.8–0.9 cm2 by pressure half-time measurement, indicating severe stenosis (Figure 4). Left atrium (LA) was markedly dilated with a left atrial volume index of 296 ml/m2. There was enlargement and decreased contractility of right ventricle (RV) with tricuspid annular plane systolic excursion of 13 mm. Left ventricle (LV) systolic function was preserved with an ejection fraction of 73%.

(a) Parasternal long axis view showed typical rheumatic mitral disease such as chordal and leaflet thickening of MV, hockey stick deformity of anterior mitral leaflet, and an enlarged LA. (b) Doppler evaluation showed a mean mitral gradient of 10–12 mmHg and a MV area of 0.8 cm2 by pressure half-time measurement, indicating severe MS. (c) Two-chamber view showed severe MR indicated by a vena contracta width of 0.7 cm with eccentric regurgitation jet. (d) Four-chamber view showed severe TR. (e) Zoomed parasternal long axis view showed moderate AR jet.
There were multiple echocardiographic signs of pulmonary hypertension such as right atrium (RA) and RV dilatation with D-shaped LV, and distended inferior vena cava. Doppler measurement revealed a pulmonic valve acceleration time (PVaccT) of 95 m/s, and TR maximal velocity of 3.4 m/s with estimated pulmonary arterial systolic pressure (PASP) of 62 mmHg. Hemodynamic measurement by Doppler echocardiography showed left ventricular outflow tract velocity time integral (LVOT VTI) of 14 cm, stroke volume of 44 ml and cardiac output of 3.4 lpm (Figure 5).
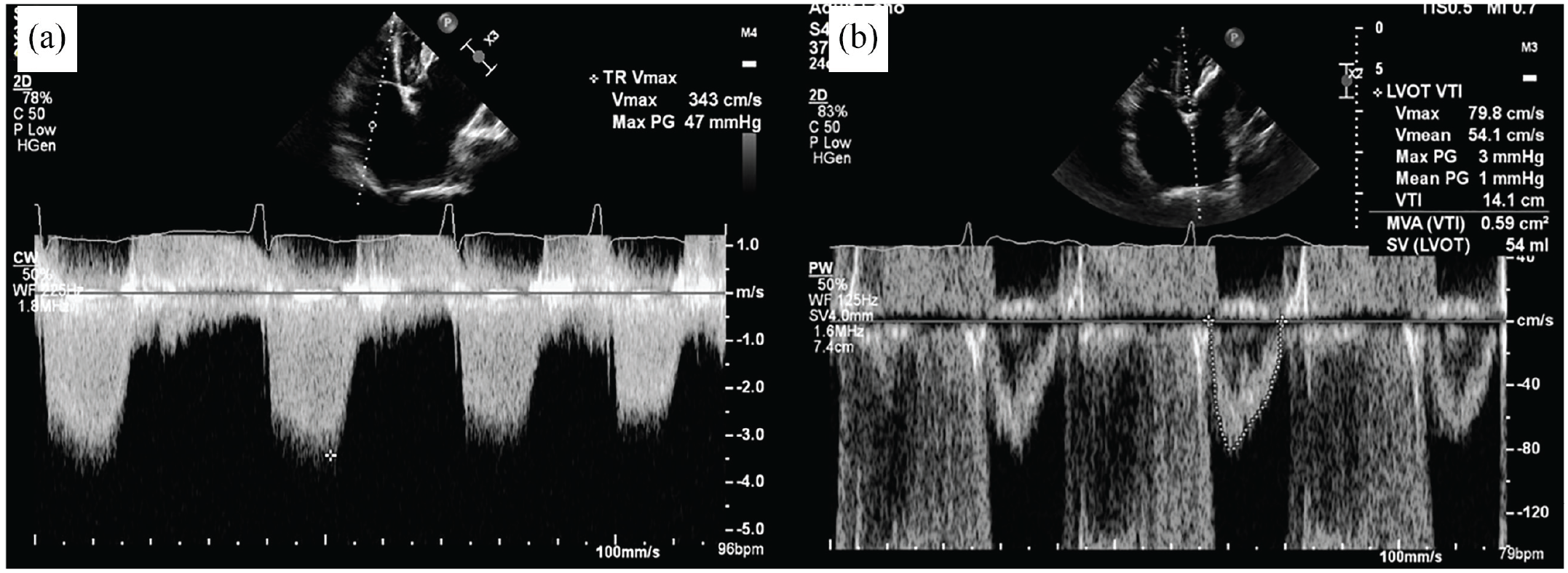
(a) Continuous wave Doppler measurement at TR jet showed a TR maximal velocity of 3.4 m/s. (b) Pulsed wave Doppler measurement at LV outflow tract showed LVOT VTI of 14 cm.
The patient was hospitalized for decongestion of the right heart failure condition with furosemide drip and spironolactone. Inotropic drug such as dobutamine was also given to improve the low cardiac output and RV contractility. After 14 days of hospitalization, acute decompensated heart failure was resolved and euvolemia was achieved. She was discharged with furosemide 40 mg, spironolactone 25 mg, ramipril 1.25 mg, digoxin 0.125 mg, warfarin 2 mg, and phenoxymethyl penicillin 250 mg. From the heart team’s discussion, it was planned for her to undergo elective surgery for MV and AV replacement, TV repair, and LA reduction. Unfortunately, 1 month after the hospitalization, the patient deteriorated and died at home during her preparation for the surgery.
Discussion
The “mitral facies” is not an uncommon finding in MS, yet this sign is often under recognized. There is very limited literature on this classical sign. It is typically described as plethoric cheeks punctuated by bluish patches. 3 Some also described it simply as pinkish-purple patches on the cheeks.6,7 “Mitral facies” has become rare in the developed world because it is usually encountered in patients with long-standing untreated MS. 8 This distinctive malar rash occurs in chronic severe MS with pulmonary hypertension.6,9 In this case, the patient’s symptomatic condition has occurred for the last 10 years and left untreated due to her socioeconomic status.
The “mitral facies” is quite similar to malar rash that can be caused by local and systemic disease such as systemic lupus erythematosus, erysipelas, rosacea, and pellagra.10,11 Systemic lupus erythematosus manifests as erythema, edematous, and sometimes purpuric rash with patchy macules, that usually precedes arthritis, fever, fatigue, and other debilitating autoimmune symptoms.12,13 In erysipelas, injury to blood vessels by streptococcal toxins produces a painful, well-circumscribed, erythematous, and shiny facial rash that is often accompanied by fever and malaise.11,12,14 Rosacea produces erythema, telangiectasia, and pustules or papules with episodes of flushing that can be triggered by hot drinks, stress, or alcohol.12,15 Dermatitis related to pellagra is a bilateral symmetric eruption on sun-exposed areas of the skin, that causes a painful, striking facial rash that usually involves the upper and lower extremities.11,12,16 In this patient, the facial rash is painless, more purplish in color, and less well demarcated with no triggering or precipitating factors. There are no accompanying systemic symptoms such as fever, malaise, or joint pain.
The pathophysiology of mitral facies alone is not commonly described. Wood stated in his article that “mitral facies” was attributed to peripheral vasoconstriction secondary to low cardiac output. This reflex vasoconstriction causes peripheral cyanosis of the face or hands. 17 He described that elevated pulmonary vascular resistance also contributed to make the cardiac output low and fixed. Atrial fibrillation (AF) and arterial oxygen saturation of less than 90% were also associated with this facial rash. 17 As MS severity worsens, flow restriction causing reduced diastolic filling, limits left ventricular output. 3 AF further impairs this limited diastolic filling. LV filling may also further be impaired by RV pressure or volume overload causing abnormal septal motion. 18 In severe MS, pulmonary vasoconstriction in addition to LA hypertension produces severe pulmonary hypertension. 3
In this patient, echocardiogram showed signs of severe pulmonary hypertension, including RA and RV dilatation with D-shaped LV, distended inferior vena cava, PVaccT of <105 ms, and an estimated PASP of >40 mmHg. Hemodynamic calculation using echocardiography showed a cardiac output of 3.4 lpm that is considered as low (<4 lpm). The reduced LV output is attributed to decreased preload from impaired LV filling caused by severe stenosis of the mitral inflow. This reduced LV preload is also aggravated by severe pulmonary hypertension and decreased RV function. The significant retrograde flow into LA also occurs due to concomitant severe MR in this patient, which further decreases the stroke volume into the systemic circulation.
Conclusion
In summary, we describe a case of 44-year-old woman with severe MS and regurgitation due to rheumatic heart disease. She came with clinical presentation of right heart failure. Echocardiogram showed severe pulmonary hypertension and low cardiac output state. We noticed on her face that there was a distinctive malar rash that had been occurring for a long time. The sign of mitral facies could be an alarming sign of more severe and advanced form of MS. It also can be a marker of long-standing impaired cardiac output and severe pulmonary hypertension condition.
Footnotes
Acknowledgements
The authors would like to acknowledge the patient and family for allowing this case report to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for anonymized information and images to be published in medical literature.
