Abstract
Atezolizumab plus bevacizumab is the recommended first-line treatment for unresectable hepatocellular carcinoma, based on guidelines from the Barcelona Clinic Liver Cancer prognosis and treatment strategy. However, atezolizumab plus bevacizumab may be used after administration of lenvatinib. Here, we present four patients who developed thyroid dysfunction after second-line treatment with atezolizumab plus bevacizumab, but not after lenvatinib alone. The patients were treated with lenvatinib and/or atezolizumab plus bevacizumab for unresectable hepatocellular carcinoma at Showa University Northern Yokohama Hospital. Of patients treated with only lenvatinib or atezolizumab plus bevacizumab, 2/18 (11%) and 4/15 (27%) developed thyroid dysfunction, respectively. All four patients treated with atezolizumab plus bevacizumab after lenvatinib developed hypothyroidism after 2–14 doses of atezolizumab plus bevacizumab. Three patients developed Grade 2 symptoms and were treated with levothyroxine sodium. In patients with hepatocellular carcinoma, the incidence of thyroid dysfunction may be higher among patients treated with atezolizumab plus bevacizumab after lenvatinib than those treated with lenvatinib or atezolizumab plus bevacizumab alone.
Introduction
Treatment for unresectable hepatocellular carcinoma (HCC) has evolved in recent years. Previously, tyrosine kinase inhibitors (TKIs), such as sorafenib1,2 and lenvatinib,3,4 were the only available options. A combination therapy of immune checkpoint inhibitors (ICIs) and humanized antivascular endothelial growth factor monoclonal antibodies, known as atezolizumab plus bevacizumab (Atezo/Bev), was approved in Japan in September 2020. 5 Based on guidelines from the Barcelona Clinic Liver Cancer prognosis and treatment strategy, updated in 2022, Atezo/Bev is recommended as first-line therapy. 6 Recently, Atezo/Bev has been widely administered as first-line therapy for HCC in Japan.
However, sorafenib and lenvatinib had long been administered prior to the approval of Atezo/Bev in September 2020. Therefore, in practice, a number of patients received TKIs (especially lenvatinib) as first-line therapy and Atezo/Bev as second-line therapy. In addition, due to comorbidities such as interstitial pneumonia and autoimmune disease, TKIs may have been selected as first-line therapy and Atezo/Bev as second-line therapy, if unavoidable. Furthermore, a third reason for administering TKI before Atezo/Bev is that administering lenvatinib after transcatheter arterial chemoembolization (TACE) has been reported to be beneficial. 7 Based on this report, some patients with HCC receive TACE, lenvatinib, and Atezo/Bev, in that order.
No cases of thyroid dysfunction were reported in the clinical trial1,2 on which the approval of sorafenib was based. In contrast, in the clinical trial on which the approval of lenvatinib was based, 3 the incidence of thyroid dysfunction was 16.0% for all grades according to the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. 8 The reported incidence of thyroid dysfunction in the clinical trial that led to the approval of Atezo/Bev for the treatment of unresectable HCC was 10.6%. 5 However, in our experience we have observed that thyroid dysfunction is more common in patients treated with Atezo/Bev after lenvatinib, than in those treated with lenvatinib or Atezo/Bev alone. In this report, we describe four patients who developed thyroid dysfunction after first-line treatment with lenvatinib and second-line treatment with Atezo/Bev.
Case presentation
We reviewed the medical records of patients treated for unresectable HCC with lenvatinib and Atezo/Bev at Showa University Northern Yokohama Hospital, between January 2018 to June 2022 and included follow-up until June 2022. Two of 18 (11%) patients on lenvatinib alone and 4 of 15 patients (27%) on Atezo/Bev alone developed thyroid dysfunction requiring levothyroxine sodium (Figure 1). In contrast, four patients (100%) who did not develop thyroid dysfunction after treatment with lenvatinib, but developed thyroid dysfunction after treatment with Atezo/Bev, were considered in this study (Figure 1). Data on patient characteristics—such as sex, age, staging, medical history, and treatment history—were obtained retrospectively (Table 1). The four patients were treated with first-line lenvatinib for 356–1251 days. All four patients developed hypothyroidism after 2–14 doses of Atezo/Bev as second-line treatment. The hypothyroidism was classified as Grade 2 in three patients with symptoms such as fatigue, and as Grade 1 in one patient. Those with Grade 2 symptoms were treated with levothyroxine sodium. All four patients were diagnosed with hypothyroidism by an endocrinologist. Details pertaining to thyroid dysfunction in each patient are shown in Table 2. The patients had no history of thyroid dysfunction prior to the first round of chemotherapy with lenvatinib. Data were also collected on adverse events other than thyroid dysfunction, such as hypertension, dry skin, eczema, and adrenal insufficiency. There were no deaths related to thyroid dysfunction.

Patient flow chart.
Patient characteristics.

Atezo/Bev: atezolizumab plus bevacizumab; HBV: hepatitis B virus; HCC: hepatocellular carcinoma; HCV: hepatitis C virus; RFA: radiofrequency ablation; TACE: transcatheter arterial chemoembolization; TAE: trans-arterial embolization; M: male; F: female.
Union for International Cancer Control (UICC) tumor-node-metastasis staging system, eighth edition.
Thyroid dysfunction after treatment of lenvatinib and atezolizumab plus bevacizumab.
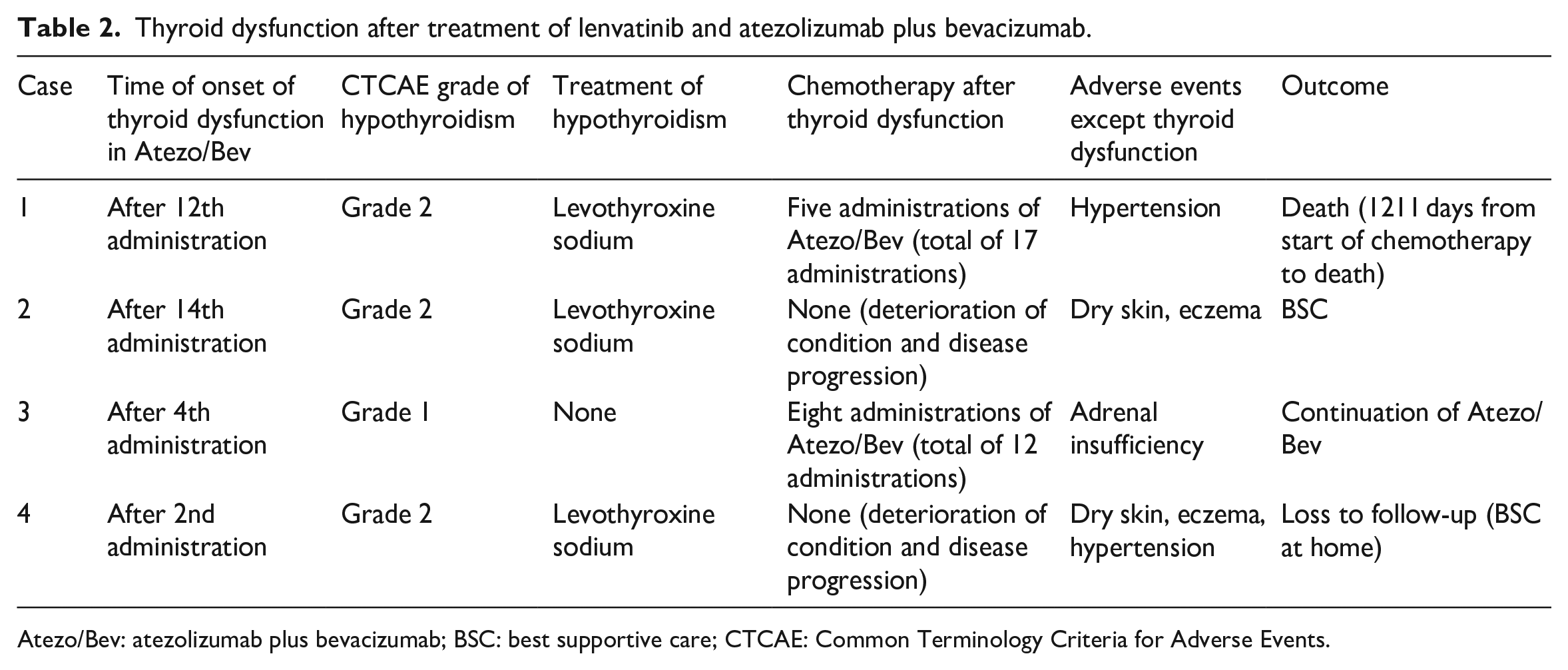
Atezo/Bev: atezolizumab plus bevacizumab; BSC: best supportive care; CTCAE: Common Terminology Criteria for Adverse Events.
Discussion
Four patients received Atezo/Bev following lenvatinib. All four patients developed thyroid dysfunction during second-line treatment with Atezo/Bev. A study based on health and medical service data (n = 665,445) found that the incidence of hypothyroidism was 4.4 times higher (odds ratio = 4.41, 95% confidence interval = 3.40–5.71) in cancer patients who receive ICIs with cytotoxic chemotherapy and angiogenesis inhibitors than in those who received ICIs alone. 9 TKIs such as lenvatinib have been reported to cause thyroid dysfunction by acting on the hypothalamus–pituitary–thyroid system.10,11 Thyroid dysfunction has been reported after treatment with epidermal growth factor receptor (EGFR)/anaplastic lymphoma kinase (ALK) TKIs, such as erlotinib, gefitinib, ceritinib, and crizotinib, in patients with non-small cell lung cancer 12 and after TKI administration in patients with renal cell carcinoma.13,14 Thyroid dysfunction can occur after first-line treatment with lenvatinib; however, the four patients in this report did not have thyroid dysfunction after first-line treatment with lenvatinib.
ICIs have been reported to cause thyroid dysfunction as an adverse event. The incidence of thyroid dysfunction with Atezo/Bev has been reported to be 10.6%. 5 The relationship between the number of doses of ICI administered and the incidence of thyroid dysfunction is unclear, as thyroid dysfunction may occur after a single dose of ICI. The four patients in this report developed thyroid dysfunction after 2–14 doses of second-line Atezo/Bev therapy.
Thyroid dysfunction has been reported as an adverse event of lenvatinib therapy alone 3 and Atezo/Bev therapy alone. 5 In this study, 2 of 18 (11%) patients on lenvatinib alone and 4 of 15 patients (27%) on Atezo/Bev alone developed thyroid dysfunction, requiring levothyroxine sodium. However, all four of the patients (100%) who we monitored did not develop thyroid dysfunction after first-line treatment with lenvatinib but developed thyroid dysfunction after second-line treatment with Atezo/Bev. This suggests that the frequency of thyroid dysfunction may be higher when ICIs are administered after TKI treatment.
This study describes a case series of patients treated at a single facility and includes a small number of cases. The thyroid dysfunction may have been due to immune-related factors or to anti-vascular endothelial growth factor (VEGF) therapy, but the limited sample size prevents us from making definitive conclusions. Another limitation is that all four case patients were not followed up for the entire period from the start of treatment until death. It is noteworthy that all four patients who developed thyroid dysfunction had HCC, as the incidence of thyroid dysfunction is relatively low in patients with HCC, compared with those with all cancers. However, the relationship between HCC and thyroid dysfunction is unclear. There may be factors other than treatment that influence thyroid dysfunction.
The administration of lenvatinib after TACE has been reported to be beneficial; 7 therefore, it is likely that more patients will receive lenvatinib after TACE followed by Atezo/Bev. We will conduct a larger study to assess the incidence of thyroid dysfunction in patients treated with lenvatinib after Atezo/Bev compared with patients treated with Atezo/Bev after lenvatinib. In some types of cancer, such as lung cancer, 15 patients with thyroid dysfunction have been reported to have a better prognosis. We would also like to assess the prognosis of patients with HCC and thyroid dysfunction.
Conclusion
Clinicians should be aware that there is a high risk of thyroid dysfunction in patients administered Atezo/Bev as second-line treatment after treatment with lenvatinib. However, further studies are required to confirm and clarify this relationship.
Footnotes
Author contributions
T.I., D.I., M.H., N.H., and T.B. contributed to the conception and design of study. T.I. contributed to the analysis and interpretation of data. T.I., D.I., M.H., N.H., and T.B. contributed to the acquisition of data. T.I. drafted the manuscript. T.I., D.I., M.H., N.H., and T.B. revised the manuscript critically for important intellectual content. T.I., D.I., M.H., N.H., and T. B. approved the manuscript version to be published.
Availability of data and material
All relevant data are within the manuscript and the supporting information files.
Code availability
Not applicable.
Consent for publication
Written informed consent was obtained from the patients for anonymized information to be published in this article.
Consent to participate
We obtained written informed consent from all the patients.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the institutional review board of Showa University, based on the ethical guidelines for medical and health research involving human subjects (approval no. 22-085-A).
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
