Abstract
Objectives:
The clinical assessment of radioiodine-induced sialadenitis is relied on the observer-defined toxicity grading model. However, this model has significant limitations, the major one being the lack of systematic assessment based on objective criteria. The main aim of this study was the development and testing of an assessment tool which could examine the severity of post-irradiation sialadenitis.
Methods:
The development of the Sialadenitis Assessment Tool proceeded through three phases. The first and second phases included a literature review and the development of the tool which derived from the review, respectively. The third phase involved a pilot testing of the Assessment Tool to a sample of 34 patients undergoing I131 therapy. The assessment was carried out by two independent healthcare professionals, pre- and post-radioiodine therapy. The results of the assessment tool were compared with other scales, including the DIRIX and EORTC H&N35.
Results:
According to the Cohen’s kappa test, the Sialadenitis Assessment Tool is a reliable tool for the assessment of sialadenitis (Cohen’s κ = 1). The concurrent and internal validity tests showed a tendency of association with most variables (p < 0.001) in the DIRIX and EORTC QLQ-HN35 scales.
Conclusion:
Preliminary evidence show that Sialadenitis Assessment Tool is a valid and reliable tool to assess radioiodine-induced sialadenitis in patients undergoing I131 therapy post-thyroidectomy.
Keywords
Introduction
Differentiated thyroid cancer and the ablation of post-thyroidectomy remnants are commonly treated with radioiodine. 1 The mechanism of action of radioiodine (I131) includes the accumulation of thyroid tissue by sodium iodide symporters and the exertion of cytotoxic effects by b-irradiation. Sodium iodide symporters in salivary gland 2 lead to radioiodine uptake that is comparable with that of thyroid tissue.3,4 Salivary tissues are highly radiosensitive, with the parotid and the submandibular glands being more susceptible to radioiodine because of their size and location.5,6
Due to the glands radiosensitivity, frequent high-dose radioiodine therapy toxicities emerge such as sialadenitis and xerostomia, 7 leading to glandular damage and salivary dysfunction. 8 These oral toxicities are not unrelated since sialadenitis may also occur, with painful, swollen salivary glands, frequently leading to some degree of xerostomia. 6 Pain or swelling of the neck and parotid region, altered taste, and difficulty on swallowing may also occur. 9
Preceding studies reported the incidence of acute sialadenitis following I131 therapy, ranging from 24% to 67%, with chronic sialadenitis remaining in 11%–43% of those treated.1,7 The clinical assessment of I131-induced sialadenitis is relied on the observer-defined toxicity grading model. However, this model has significant limitations, the major one being the lack of systematic assessment based on objective criteria (i.e. measurable criteria). Furthermore, this grading model generates concerns as to the objectiveness of the person undertaking the assessment since this can be heavily influenced by the knowledge, expertise, and clinical experience (i.e. with sialadenitis cases) of the evaluator. Even in optimal conditions, the assessment does not provide a measurable outcome in relation to the actual severity grade of sialadenitis (e.g. whether dryness is mild or moderate or whether it is moderate or severe). This therefore limits the ability of the assessor to obtain an accurate assessment of the presence of sialadenitis that can be prospectively left untreated, leading to more severe symptoms and secondary complications. 7 In addition, the absence of a structured tool for the assessment of sialadenitis contributes to the lack of its integration into the routine assessments, despite its relatively high incidence, unless clinical symptoms are reported by the patient (e.g. presence of pain) or become visible (e.g. swelling), leading to inadequate preventing measures in patients likely to develop sialadenitis. Finally, the lack of such tools impedes the consistency of the patient’s assessment for sialadenitis over time (e.g. for comparison) and across settings (e.g. when the patient is transferred to another center).
From the above, it is apparent that the availability of a tool that includes a post-radiation sialadenitis grading system is of paramount importance, as part of a comprehensive management approach to its toxicity.
A preliminary preprint of the present manuscript prior to review is available with DOI:10.21203/rs.3.rs-191433/v1.
Methods
Aim
The aim of the present study was to develop and preliminarily validate a reliable grading tool that will assess the severity of post-irradiation sialadenitis.
Study design
A pretest–posttest design was implemented.
Method
The development and preliminary validation of the Sialadenitis Assessment Tool (SAT) was guided by international guidelines and methodologies and implemented through three distinct but interrelated phases. 10 In phase I, a systematic review of the literature was undertaken for the items and domains identification, followed by the synthesis of the Tool. Phase II included the testing of the content validity of the SAT by an experts’ panel.
Finally, in phase III, the pilot testing of the tool was undertaken in a sample of adult patients diagnosed with thyroid cancer, pre- and post-radioiodine treatment.
Phase I
The authors performed a systematic review of the literature targeted on tools related to the assessment of sialadenitis. PubMed, CINAHL, SCOPUS and COCHRANE scientific databases were searched for relevant research, published between 1 January 1955 (i.e. first case report of “radiation sialadenitis” from I131) and 31 December 2015. The following search terms were utilized: “sialadenitis” AND “sialitis” AND “sialadenitis” AND “treat” AND “assessment tool” OR “test” OR “instrument” AND “measure” OR “assessment” AND “H&N” AND “cancer” OR “oncology.” The inclusion criteria included clinical studies in humans that used tools for the assessment of sialadenitis in any context, and full text articles published in English. Grounds for exclusion included incomplete studies, case studies, and publications in a language other than English. Types of papers selected were those describing the assessment of sialadenitis in any context.
As shown in Figure 1, the initial search yielded a total of 91 titles, based on the predetermined key words and the inclusion criteria. A total of 34 articles were retrieved from PubMed, 57 from SCOPUS, 0 from CINAHL, and 0 from COCHRANE. After a review of the titles, abstract, and full text, 5 original studies were included in the systematic review. In addition, the references of the included articles were checked to identify relevant clinical studies. The systematic review findings are presented on Table 1.

Literature search flow chart.
Systematic review results.

SGS: salivary gland scintigraphy; GBI: Glasgow Benefit Inventory; COSS: Chronic Obstructive Sialadenitis Symptoms; RAIT: radioactive iodine treatment; SASDS: sialendoscopy-assisted salivary duct surgery.
A repeated search of the literature with the same inclusion and exclusion criteria was held between January 2017 and September 2019 through SCOPUS, CINAHL, COCHRANE, and PubMed in order to identify any newly emerged articles.
Based on the findings of the systematic review, the following assessment tools have been identified from the first study: sialendoscopy, symptom questionnaires (xerostomia), salivary flow rate, and scintigraphy. 11 The second study used salivary gland scintigraphy (SGS), sialometry, and a subjective open questionnaire. 12 The assessment tool that emerged from the third study 13 was the Oral Health Impact Profile-14 (OHIP-14), which intended to measure the self-reported QOL of subjects who had undergone salivary endoscopy as a treatment for chronic sialadenitis. Ianovski et al. 14 used the Glasgow Benefit Inventory (GBI) to assess patient-perceived treatment benefit after their procedure. The aforementioned second review of the literature yielded the Chronic Obstructive Sialadenitis Symptoms (COSS) Questionnaire, which was used to quantify and assess symptoms of invasive salivary duct surgery. The researchers implemented the use of 2 questionnaires: COSS and ShortForm8 Health Survey (SF-8) to measure the symptoms of chronic sialadenitis. 15
A thorough analysis was performed by the researchers in order to identify the means, the scales, and the items that were used for the assessment of sialadenitis. In most cases, sialadenitis was assessed clinically (e.g. by the presence of symptoms such as swollen, painful salivary glands) and often by the use of a VAS (Visual Analogue Scale) recording in order to recognize the presence of symptoms such as sensitivity, without assessing though the severity of the symptom.15,16
Studies in which an instrument was used to assess the condition of the salivary glands primarily involved the use of the Radiation Therapy Oncology Group (RTOG)/European Organization for Research and Treatment of Cancer (EORTC) classification criteria for early and late radiation reactions.11–15
The potential items of these instruments were extracted from the identified studies by implementing a parallel process between the researchers. Each researcher was requested to study these items, taking into account the known clinical manifestations of acute sialadenitis (e.g. swelling) and classify them accordingly as “essential,” “interesting but not essential,” and “irrelevant.” An agreement level of 75% among the researchers was decided as the threshold for inclusion in the pool of prospective items that would constitute the SAT. A pool of statements from the literature review was also integrated to achieve an adequate and comprehensive sample of items that corresponded to sialadenitis’ symptoms.17,18 These symptoms primarily included salivary gland pain, tenderness, swelling, limited saliva production, purulent exudate, and bloody exudate. The retrieved items were either used verbatim or shortened into affirmative and descriptive statements. Taking the above into consideration, and after a detailed review of signs and symptoms of sialadenitis, the four researchers designed the first version of the assessment tool, which included 14 potential items that specifically referred to the assessment of acute sialadenitis grouped under severity levels (i.e. grades). The potential items are illustrated on Table 2.
Potential items for inclusion in Sialadenitis Assessment Tool.

Phase II
Content validity specifies content relevance and content representations, that is, that the items capture the relevant experience of the target population being examined. The need for content adequacy is vital if the items are to measure what they are presumed to measure. 19
Following the initial retrieving and assessment of potential items, the authors consulted 8 healthcare professionals (HCPs) to evaluate the items and their relationship to the measured construct (e.g. to identify which symptoms indicate increased severity). The 8 HCPs group of experts consisted of 4 different specialties. Specifically, 3 medical oncologists, 1 nuclear physician, 3 cancer nurses, and 1 radiologist were involved. The aforementioned specialties were purposively selected because of their frequent involvement with patients (including the actual assessment process of acute sialadenitis) undergoing chemotherapy and radiotherapy, as well as their accompanying side effects, such as acute sialadenitis.
Specific questions were addressed to the experts that included the following:
a. Is the wording of the items correct for the respondent audience?
b. Should some item(s) be removed that do not apply to the construct domain?
c. Are there other items missing from the tool?
d. Are there (technical or specific) terms that can be misunderstood by the respondents?
e. Evaluate if each group of items belongs to each construct (i.e. severity level).
Lawshe 20 developed a quantitative measure for assessing content validity, called content validity ratio (CVR). The CVR offers information about item-level validity. 21 Items were retained in the tool if an agreement level of over 75% was achieved between the experts while the same agreement level was applied with regard to the severity-level constructs. A decision was made prior to this stage that any ambiguous items that could not be clearly categorized in one of the dimensions would be excluded. Three items were discarded by the experts as being irrelevant to the clinical assessment of sialadenitis. Once this process was completed, the experts were asked to provide evaluations in relation to the level of relevance of each item to the symptomatology and severity of acute sialadenitis. Depending on their relevance, the items were categorized as follows: 3 = “essential,” 2 = “interesting but not essential,” and 1 = “irrelevant.” For each item, the CVR was calculated with a decision to retain only items with over 87% agreement. The Content Validity Index (CVI) for the entire assessment tool was also calculated. At this stage, the assessment tool’s items were reduced to 11 and were categorized to normal, mild, moderate, and severe according to the symptoms. Upon completion of this process, the experts reached to a consensus that the scale was comprehensive, inclusive of all the relevant aspects of acute sialadenitis and without any ambiguous terms. The final SAT is demonstrated in Table 3.
Sialadenitis Assessment Tool (to be used in parallel with physical examination a ).

Physical examination should begin with the gland itself. The gland should be palpated for the presence of calculi. Examine the ductal opening for purulence. Palpation should extend into the floors of mouth as well as the soft tissue of the tongue, cheek, and neck. Lingual papillary atrophy should be looked for, as well as loss of enamel from the tooth surface. All of the major salivary glands should be examined for masses, symmetry, and the presence of discharge. The presence of lymphadenopathy should be noted.
Severe sialadenitis symptoms may intervene with patients’ therapy when not timely treated. 22 The SAT consists of 4 grades, staging from 0 to 3, and can guide the escalation of treatment. In particular, Grade 0 could be treated with preventive measures, namely, hydration and sialagogues, such as lemon candy, lemon juice, lemon slices, Vitamin C, and Vitamin E.23–26 Grade 1 could be treated with the above preventive measures, with the addition of local ice therapy for cases of sensitivity, 23 whereas Grade 2 patients could benefit from the use of the aforementioned measures and the use of cholinergic agents. 23 Sialendoscopy can be used when pharmacologic agents are ineffective,14,23 as well as to improve the symptoms of Grade 3 sialadenitis. 11 Sialadenectomy is performed as a last resort when conservative management has failed. 27
Acute sialadenitis has a good prognosis. Complete resolution is usually the expectation following conservative outpatient management. Most of the acute symptoms resolve in a week; however, edema takes a longer time to disappear. 28 Furthermore, despite the good prognosis of acute sialadenitis, rare serious complications may occur, including facial paresis/paralysis and development of salivary gland neoplasms. 27
Phase III
In a group pretest–posttest design (baseline vs 1 month after commencing I131 therapy), the assessment tool’s validity and reliability were tested. In order to assess its validity and reliability, the SAT was used for assessment of patients diagnosed with thyroid cancer, who underwent thyroidectomy and were currently on radioiodine treatment. Patients were assessed in terms of experiencing mucositis, salivary glands edema, and xerostomia additionally to sialadenitis.
Sample and setting
The study was conducted at the Bank of Cyprus Oncology Center (BOCOC), a major oncology center situated at Nicosia, Cyprus.
The study sample consisted of adult patients who were diagnosed with thyroid cancer, undergoing I131 therapy following total thyroidectomy. Inclusion criteria also involved the ability of patients to complete the aforementioned tool and be able to speak and write Greek or English. Patients with metastasis or/and with a history or active detriment of salivary glands were excluded from the pilot study.
The sample size for this validation study was calculated using power analysis for the expected interobserver agreement Kappa statistic.29,30 The minimum sample size, for an expected kappa statistic of 0.7, between 2 raters, for a multinomial scale of 4 categories (normal, mild, moderate, severe) with expected frequencies of 20%, 30%, 30%, 20% respectively, was 29 participants. Over the study period, we recruited 34 persons. The power analysis was conducted in R
31
with the package
Data collection tools
1.
2.
3.
Both of the aforementioned questionnaires (DIRIX and EORTC QLQ-Η&Ν35) were validated and used in the local language and same group of patients (i.e. head and neck malignancies) by Charalambous et al. 35
4.
The use of self-reported questionnaires was considered necessary in order to have subjective measurements to assess the involved patients’ view and also for the implementation of comparisons between objective and subjective measurements that would lead to the strengthening of the validity of the results.
After an extensive review of the literature, the tools used were selected aiming to achieve the objectives of the research. Particular attention was paid to the individuals who would complete the questionnaires and their general health status in order to avoid further inconvenience.
The entire research team carefully studied the questionnaires with emphasis on their validity and reliability and concluded that two widely used self-reported questionnaires were to be utilized. The use of the same questionnaires in a similar population in Cyprus comprised an important parameter. 35
Data collection process
During the pretest phase, which was held in hospital, the clinical evaluation for classification of the SAT was implemented by the examining radiation oncologist doctor and a cancer nurse of the oncology center prior to the I131 administration.
The clinical evaluation and the collection of the questionnaires of the posttest phase, took place 1 month after the first evaluation, in an outpatient setting. The evaluation was performed by an outpatient cancer nurse who had been appropriately prepared by the researchers.
The time period set in the study for the evaluation of sialadenitis that may occur after iodine administration was research-proven. The incidence of sialadenitis in the literature ranges from 24% to 67% of cases that received I131 1 month and 1 year after the therapy.36,37 By that time, salivary gland damage is possible, according to research.12,38,39
Statistical analysis
In order to examine the assessment tool’s concurrent and predictive validity, the DIRIX, the Oral Mucositis questionnaire, and the EORTC QLQ-HN35 assessment tools were used (see online supplemental).
The level of the questions of the Oral Mucositis questionnaire are presented as mean and standard deviation (SD). The quality-of-life level according to the DIRIX tool and the symptomology according to the EORTC QLQ-HN35 tool are presented as mean and SD for their individual dimensions.
The reliability of the SAT was assessed using the Cohen’s kappa index for the inter-rater reliability between the two independent assessments. Criterion validity of the SAT was assessed using two methods:
Concurrent validity: exploration of whether the Sialadenitis assessment levels at 1-month post-I131 assessment tool was associated with (1) the validated assessment tool DIRIX (xerostomia, pain, taste loss and dysphagia dimensions), and (2) the responses to the two questions regarding dry mouth and swollen salivary gland, causing edema. The association was explored using the chi-square test.
Predictive validity: the SAT was explored for whether it can be predictive of another assessment tool. Predictive validity was assessed by exploring the associations of the sialadenitis assessment levels at 1-month post-I131 with (1) the EORTC QLQ-HN35 dimensions that explicitly assess symptomatology regarding the area of the oral cavity: HN dry mouth, HN pain, HN social eating, HN sticky saliva, HN swallowing, and the level of QoL as measured by the DIRIX scale. The DIRIX QoL scale is an index of decreased quality of life, meaning that an increased score is associated with lower levels of quality of life. The association was explored using the analysis of variance (ANOVA) test.
The statistical analysis was performed using the SPSS v21. Figure 2 was produced in R v.3.6.1 40 using the package ggplot2. 41

Association of sialadenitis scale with the HN symptoms scale and QoL index by DIRIX.
Ethics approval and consent to participate
The study complied with all the principles of the Helsinki Declaration as these were developed by the World Medical Association (WMA). Approval was obtained by the Bioethics Committee in Cyprus and the Ethics Committee of the research site. A participant information sheet providing details of the study accompanied the questionnaires. Written and oral consent were assured by all the informants. Patients were informed that their participation in the study was strictly on a voluntary basis, and in the event they wished to decline their participation in the study, there would be no consequences in relation to their treatment or care.
Results
Thirty-four patients participated in the pilot study. Twenty-five (73.5%) were female. Mean age of participants was 45.2 (SD = 4.3) years. Twenty (58.8%) had a tertiary education and 24 (70.6%) were married.
At baseline, 25 (85.3%) patients were assessed with a normal grade, and 5 (14.7%) with a mild sialadenitis grade.
After 1 month, 19 (55.9%) patients were assessed with a normal grade, 6 (17.6%) with a mild, and 9 (26.5%) with a moderate sialadenitis grade. The demographic characteristics of the sample are presented in detail in Table 4.
Sample demographic characteristics.
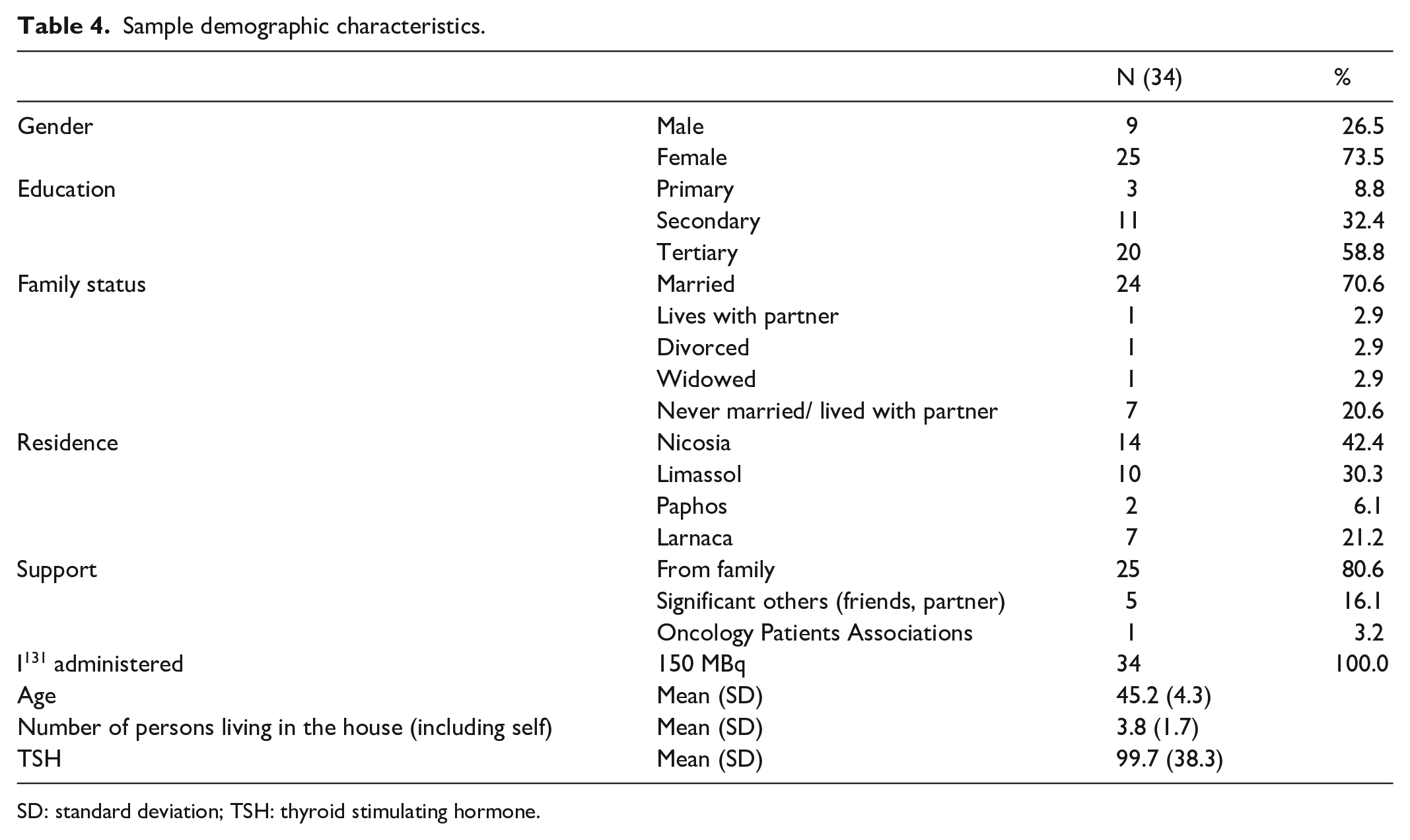
SD: standard deviation; TSH: thyroid stimulating hormone.
Inter-rater reliability
Two independent raters assessed the patients’ sialadenitis grade pretherapy and two raters posttherapy, with identical results. The raters consisted of one radiation oncologist doctor and one cancer nurse who was previously trained on the completion of the questionnaire. The first assessment took place prior to the patient’s therapy at the hospital. The radiation oncologist doctor assessed the patient at the outpatient department and the nurse assessed the patient at the oncology ward, during the admission process. The second assessment from the oncologist took place at the hospital 1-month post therapy, during the patient’s scheduled appointment, whereas the assessment from the trained nurse took place at a home visit, at the same day. The two raters had no interference between them. Both raters agreed on all ratings (Cohen’s κ = 1) with identical results.
Concurrent validity
As shown in Table 5, SAT results were associated with the responses in the two questions regarding dry mouth (p < 0.001) and swollen salivary gland (p < 0.001). Specifically, 90.5% of the patients who answered that they had not felt a dry mouth had a normal sialadenitis grade, while all patients who responded that they felt a dry mouth had a mild (38.5%) or moderate sialadenitis grade (61.5%). Similarly, in the second question regarding edema in the salivary glands, patients who felt the swelling were more likely to have a mild or moderate sialadenitis grade.
Association of Sialadenitis Assessment Tool results with questions on dry mouth, salivary glands edema, and DIRIX xerostomia grade measurements (N (%)).

Regarding the DIRIX xerostomia measurements, an association was shown between sialadenitis grade and dysphagia (p = 0.002). More specifically, patients with no dysphagia were more likely to have a normal sialadenitis grade (64.3%) compared with the patients with mild dysphagia where all of them had a moderate sialadenitis grade.
Differences were also associations. For example, differences with respect to QoL between the 3 sialadenitis levels (normal, mild, and moderate) could be also considered as associations between QoL assessment tool and SAT, as described next.
Predictive validity
Predictive validity was assessed by exploring the associations of the SAT results with the EORT Head & Neck assessment tools.
Table 6 presents the mean (SD) level of the HN symptom and the mean (SD) level of the QoL by DIRIX across the assessment level of the SAT. Patients who were assessed as having a moderate grade by the SAT had worst QoL (p = 0.035) and higher scores for: dry mouth symptoms (p = 0.001), pain (p = 0.002), symptomatology with respect to Social Eating (p = 0.052), symptoms with regards to sticky saliva and symptoms with regards to swallowing (p = 0.053). Figure 2 shows the distribution of scores of the patients across each assessment level of SAT, where the scores in each symptom were always higher in the patients with a moderate sialadenitis grade compared to the patients with normal and mild sialadenitis grades.
Association of Sialadenitis Assessment Tool results with HN symptoms scale.
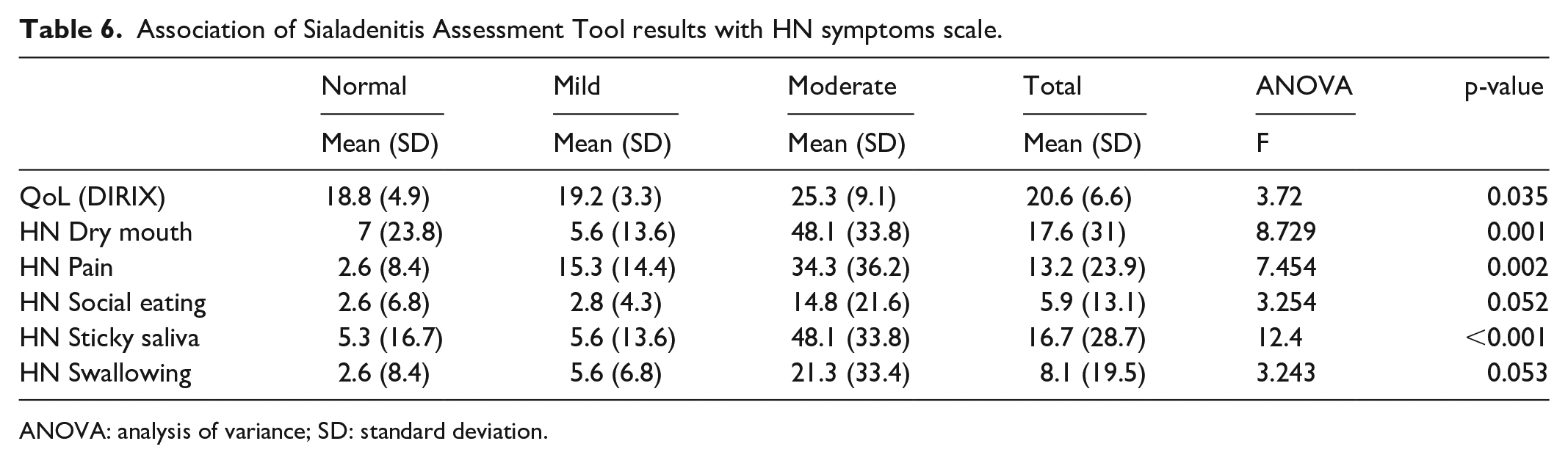
ANOVA: analysis of variance; SD: standard deviation.
Discussion
There are many tools for assessing complications after Iodine-131 treatment, following thyroidectomy.11–15 However, these mostly emphasize on the assessment of xerostomia and oral mucositis rather than sialadenitis. As a result, the presence and severity of sialadenitis are often underestimated and misdiagnosed, leading those patients to pain and discomfort, or even to secondary complications. 11 The aim of this study was to develop a reliable tool through a rigorous process for the assessment of post-irradiation sialadenitis and evaluate its preliminary validity and reliability.
The results of our study, and in particular Cohen’s kappa test, suggest that the SAT is a reliable tool for the assessment of sialadenitis in patients who underwent I131 therapy. The concurrent and internal validity of the tool were also tested with the use of dry mouth/salivary glands edema–relevant questions and the DIRIX and the EORTC QLQ-Η&Ν35 assessment tools, respectively. The results showed consistency with the SAT when compared with these widespread and validated tools.
The SAT is the first to recognize and categorize the presence of sialadenitis solely, following post-thyroidectomy I131 treatment. Other available tools that assess the presence of sialadenitis do so in the framework of general radiation reactions, 14 thus not focusing on the severity of sialadenitis within the acute context, an aspect that does not allow for any meaningful comparisons to be made.
The assessment tool consists of 4 grades, ranging from 0 to 3, with grade 0 denoting that the patient has no symptoms of sialadenitis and grade 3 that the patient has symptoms of severe sialadenitis. The grading of the assessment tool was based on clinical findings, which can guide the escalation of treatment, thereby reducing the probability of inadequate therapy, mistreatment, or even delayed treatment.
The uniqueness of the SAT derives from the fact that it provides a specific grading system that is correlated to the symptoms induced by sialadenitis and thereby allowing for the appropriate imaging diagnostic tests to be performed (i.e. non-enhanced computed tomography (CT) scan, magnetic resonance (MR) sialography, qualitative and quantitative salivary gland scintigraphy) and corresponding treatment to be initiated. This is in contrast with preceding studies where questionnaires had been used to assess sialadenitis, but these were limited in describing the symptoms of xerostomia. 42 Similarly, Ianovski et al. 14 utilized a different questionnaire to assess sialadenitis, but the majority of the questions dealt with xerostomia symptoms. Only a limited number of questions were included with regard to salivary glands which were not exhaustive of the clinical manifestations of sialadenitis. Gillespie et al. 13 used the OHIP-14 questionnaire (Oral Health–Related Quality of Life Appraised) to assess sialadenitis, but its main focus is on QoL and of general oral condition. Aubin-Pouliot et al. 15 dispensed 2 questionnaires (COSS and SF-8) to the participants in order to assess sialadenitis, but only focused on sialadenitis in the chronic obstructive stages, rather than the acute. The COSS is a self-reported 20-question survey based on sialadenitis symptoms, such as salivary gland pain, tenderness, and swelling during and in between meals. The COSS also examines the impact of these symptoms on daily functions (i.e. chewing, speaking, and swallowing), emotional symptoms (i.e. embarrassment), and QOL measures (i.e. sleeping and daily activities). In addition to COSS, the SF-8 revolved around questions regarding QoL. Ianovski et al. 14 in a prospective observational study, involving patients that underwent sialendoscopic procedures in New Zealand, completed the GBI. The GBI is a validated tool to measure the quality of life of patients after interventions and has been shown to be sensitive to otolaryngology interventions. 43 The GBI questionnaire consists of 18 items: 12 related to general improvement, 3 to social improvement, and 3 to physical improvement. Each question had a numeric rating scale for responses, which was further statistically analyzed. 12 As with preceding scales that were used in this context, the GBI also emphasizes on the general QoL improvement related to the treatment rather than the actual sialadenitis symptoms or the sialadenitis severity. Overall, the current available assessment tools do not provide a comprehensive assessment of sialadenitis within the acute context and this can negatively impact the appropriate assessment of this toxicity, the undertaking of appropriate diagnostic imaging tests, and the timely onset of the recommended treatment. In turn, this can negatively influence the overall QoL of the patient.
Although this study offers a preliminary validated tool, its use is restricted to patients undergoing I131 radiation and should only be used in conjunction with clinical examination, as instructed by the assessment tool notes. Another limitation of the study was the lack of pilot testing of the tool, since it was the outcome of the experts panel. As this study is the first one to provide evidence on the reliability and validity of the SAT, more robust prospective studies are needed to continue to assess the preliminary validity and reliability of the tool with more tests and a wider population. The assessment tool was used in Greek, thus should be validated for its use in other languages.
Conclusion
Radioiodine-induced sialadenitis is a frequent complication that affects up top 67% of thyroid cancer patients who have received I131 therapy to ablate remnant thyroid tissues after thyroidectomy. Despite its frequency and its negative effects on the patient’s QoL, there is no standardized and systematic assessment tool for sialadenitis adopted in clinical practice, giving prominence to the uniqueness of the SAT. Overall and despite its limitations, this study suggests that the referent tool is preliminary reliable and validated for the assessment of sialadenitis on the targeted population. As this tool is user friendly, it could be easily used at bedside as well as to help for documentation, research, and communication of acute sialadenitis severity and assess the impact of treatment options.
Supplemental Material
sj-docx-1-smo-10.1177_20503121211042211 – Supplemental material for A pretest–posttest pilot study for the development and preliminary validation of a tool for the clinical assessment of radioiodine induced sialadenitis
Supplemental material, sj-docx-1-smo-10.1177_20503121211042211 for A pretest–posttest pilot study for the development and preliminary validation of a tool for the clinical assessment of radioiodine induced sialadenitis by Andri Christou, Evridiki Papastavrou, Anastasios Merkouris and Andreas Charalambous in SAGE Open Medicine
Footnotes
Acknowledgements
The authors would like to express their gratitude to the healthcare personnel, as well as the patients who participated to the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from by the Bioethics Committee of Cyprus (approval number: 2012.01.113) and the Ethics Committee of the research site (approval number: 06.3.3013).
Informed consent
Written informed consent was obtained from all participants before the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
